2-1 The Macromolecules of Cells: Carbohydrates Lipids Proteins Nucleic Acid.
CARBOHYDRATES LIPIDS PROTEINS NUCLEIC ACIDS CHNOPS, CHNOPS, CHNOPS, CHNOPS, CHNOPS, CHNOPS…...
-
Upload
lambert-chandler -
Category
Documents
-
view
235 -
download
1
Transcript of CARBOHYDRATES LIPIDS PROTEINS NUCLEIC ACIDS CHNOPS, CHNOPS, CHNOPS, CHNOPS, CHNOPS, CHNOPS…...

CARBOHYDRATESLIPIDS
PROTEINSNUCLEIC ACIDS
CHNOPS, CHNOPS, CHNOPS, CHNOPS, CHNOPS, CHNOPS… EVERYBODY!
Macromolecules

What is a macromolecule?
Large molecules made up of smaller building blocks or subunits
They are polymers made up of monomers!
*Think back to that polymer you created in class that was made up of 3 subunits (monomers)

Carbohydrates
STRUCTURE: Contain mostly Carbon, Hydrogen, & Oxygen (CHO) Most carbohydrates have the composition of
1C:2H:1O ratio 1:2:1 ratio is seen in glucose C6H12O6
FUNCTION: Provide our cells with fuel (energy) to perform all
cellular activities Also provide strength and structure to cells and other
organisms (ex: insects)

Form follows function…
Form follows function Phrase often used in biology meaning that the structure
of an organism will evolve to best fit its function! Ex: Sharks are streamline in order to swim quickly
through the water to catch prey (Streamline form evolved as a predation advantage)
Carbohydrates main function is energy! How does this relate to the structure of a carbohydrate? Carbs have a lot of C-H bonds, & when they are broken,
a lot of energy is released! This chemical energy can be used to make ATP!

Monosaccharides – Simple Sugars
A single monomerContain 3-6 Carbon atoms & when they are
broken down, the products are usually NOT carbohydrates
Examples: Glucose, Fructose, Galactose What do you know about simple sugars?

Glucose Fructose
C6H12O6
Most important to living organisms
All other sugars are converted to glucose in the body
Found in sap & plants naturally Plants make this through
photosynthesis
C6H12O6
Found mostly in fruits, veggies, & honey
Sweetest of all sugars
Monosaccharides- Simple Sugars

If it ends in –ose, it’s a carbohydrate
Would sucrose be considered a
monosaccharide? Why or why not?

Glucose
The 3 fates of glucose:1. Fuel for cellular activity
Weak C-H bonds are broken in a glucose molecule & stronger bonds are formed, which releases ENERGY that can be used by the cell
Instant cellular energy
2. Stored temporarily as glycogen If blood glucose is high & the body does not need energy, glucose
can be stored primarily in the liver (& muscle) as glycogen Glycogen is a bunch of glucose molecules linked together and can
easily be broken down to release glucose into the blood when necessary
Remember what chemical causes glycogen to be converted into glucose?
3. Converted to fat for long-term storage

Dieting: Why does it work so well for the 1st week?
If you reduce the amount of calories you are consuming & are exerting more energy (exercise), your body must get energy from storage.
Glycogen stores will be raided due to lack of glucose in the blood stream.
Glycogen is bound to large amounts of water. 1 lb of glycogen = 4 lbs of water molecules bound to it
So as glycogen is removed from your tissues, so is a lot of water
Goodbye water weight. When your body begins to use fat as energy, weight loss
will slow dramatically because you will not be losing as much water

Disaccharides
2 monosaccharides bound together
Sucrose is a disaccharide Glucose and fructose
Lactose is a disaccharide Glucose and galactose

Polysaccharides- complex carbs
When large numbers of simple sugars (monosaccharides) are joined together
What is the polymer? What are the monomers?
Depending on what monosaccharides make up the polysaccharide, they can function as stores of energy or as structural material

Energy from disaccharides & polysaccharides is harder to obtain
First must break the bond between the individual sugar molecules & then those sugar molecules can be broken down (C-H bonds will release energy)
Starch is a polysaccharide consisting of 100+ glucose molecules joined together in a line Starch is the form of energy storage in plants (corn,
rice, grains, potatoes) Starch can still be used as energy, but it takes longer
to break it down… why?

Other Complex Carbohydrates
Chitin Forms outer shell of insects and crustaceans What is the function of chitin?
Cellulose Most prevalent compound on earth! Forms plant structures Indigestible to humans- but still important… Do you
know why? Roughage- actually scrapes your digestive tract walls to
rid them of any unwanted/harmful products of digestion

Lipids
Structure: Made primarily of CHO Just like carbs but in different proportions Come in a variety of structures, making them harder to
define
Function: Contain more stored energy than carbohydrates
Characteristics of lipids: Not soluble in water… what does this mean? Greasy to touch Nonpolar

Lipids
Nonpolar due to long chains of C-H atoms Charge is shared equally by C and H
Since lipids are nonpolar, they cluster together rather than interact with water molecules Remember that water is polar; lipids are not! Lipids are often called hydrophobic… what does this
mean?
Let’s try an experiment!

3 Types of Lipids
Fats Long term energy storage & insulation
Sterols Regulate growth & development Ex: cholesterol, sex hormones
Phospholipids Form cell membranes that enclose cells, providing a
barrier

Fats
Glycerol head region and 2-3 long fatty acid tails
The fatty acid tails are hydrocarbons Hydrocarbons = chain of a dozen or so carbons
bonded together with 1 or 2 hydrogen atoms on each carbon atom
Triglycerides are fats that have 3 fatty acid hydrocarbon tails
The chemical breakdown of fat molecules release more energy than carbohydrates

Saturated Fats Unsaturated Fats
Each C atom in the fatty acid chains has 2 H bonded to it; no double bonds
Found in animal fats, meat, eggs, etc.
Accumulate in your bloodstream & narrow vessels
Fatty acids line up tightly
Some of the C atoms in the fatty acid chains only have 1 H atom, thus have a double bond to another C
Found in avocados, peanuts, plant oils
Kinks in the tail due to C=C
Fats

Saturated vs. Unsaturated Fats
Which is solid at room temperature? Which is liquid? Why?

What does “partially hydrogenated” mean?
A liquid unsaturated fat (like vegetable oil) has had H added to it to make it more saturated.
Changes the texture and consistency of the food
Gives chocolate that melt in your mouth texture
It is less healthy though because saturated fats are more likely to accumulate in your blood vessels since they’re less reactive

Sterols
Regulate growth & developmentBasic structure = 4 interlinked rings of C
atomsCholesterol is necessary for cell membranes,
but if you ingest too much cholesterol, it will attach to blood vessels, thickening them Increases blood pressure, making the heart pump
harder Steroid hormones (testosterone & estrogen)
regulate sexual development and sperm & egg production A variation of cholesterol

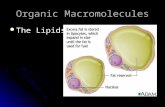
Phospholipids
Make up the cell membrane that surrounds cells & controls the flow of chemicals in and out of the cell
Have a phosphorus head & 2 fatty acid tailsPhosphate head is hydrophilic & always faces
outwards on the lipid bilayerThe fatty acid hydrocarbon tails are
hydrophobic and face inwards

Proteins
Function: Many functions; they are the building blocks of life! Make up bones, muscles, skin, feathers; fight
microorganisms; control chemicals in your blood stream; carry oxygen; act as enzymes Structural, protective, regulatory, contractile, transport
Enzymes- start and assist in chemical reactionsStructure:
Made from CHO & N and some have S Made up of 20 different amino acids (monomers)

Amino Acid Basic Structure
Central C covalently bonded to a carboxyl group, amino group, hydrogen, & R group
The R group is the only thing that differs among the 20 amino acids & is called a side chain Determines the amino
acids properties


Proteins in our Diets
When we eat proteins, our body recycles the amino acids to make more proteins
Proteins can also store energy in their bondsOur bodies can make some amino acids, but
about half of the 2o cannot be made… where do we get them? These a.a. that we can’t make are called the essential
amino acids

Protein Structure
Proteins are formed by linking amino acids together with a peptide bond
DipeptidePolypeptide
Dehydration Synthesis Reaction

Primary Protein Structure
The sequence of amino acids in the polypeptide chain
Example: met hionine-lysine-guanine-
cysteine-proline-valine

Secondary Protein Structure
Amino acids in a polypeptide chain do not remain in a simple line, but there are corkscrew twists and pleated folds formed by hydrogen bonds between the amino acids in the chain Alpha helix Beta pleated sheets

Tertiary Protein Structure
The protein folds and bends on itself forming a complex 3D shape based on the side chain interactions with each other and with the aqueous surrounding environment What types of amino acids
are found in the middle of the protein? Which on the outside? Why?

Quaternary Protein Structure
Some proteins have a quaternary structure if 2 different polypeptide chains are held together by amino acids from each
Hemoglobin is made of 4 polypeptide chainsProtein structure is essential to proper
function

Enzymes
Proteins help initiate and speed up specific chemical reactions in our bodies
They remain unchanged at the end of the reaction, thus can be reused again and again
Active site- a groove in the protein that provides a place for the participants (reactants or substrate) in the chemical reaction to nestle
Enzymes bind only with their substrate… how?

Nucleic Acids
Function: Store information & play a role in the
production of proteinsStructure:
CHNOP Subunits called nucleotides that are
made up of a sugar, phosphate, & nitrogen base
The backbone is alternating sugar and phosphate groups and the nitrogen bases are like rungs to the ladder

DNA- deoxyribonucleic acid
RNA- ribonucleic acid
Double stranded helix
Base pairs: Adenine & Thymine Guanine & Cytosine
Much longer
Single strandedBase pairs:
Adenine & Uracil Guanine & Cytosine
Much shorter
Types of Nucleic Acids


Review
What are the functions of each of the 4 macromolecules?
What are the basic structures of each of the 4 macromolecules?
Give an example of each of the 4 macromolecules.