Management of Primary CNS Lymphoma (PCNSL)
-
Upload
narayan-adhikari -
Category
Health & Medicine
-
view
569 -
download
2
Transcript of Management of Primary CNS Lymphoma (PCNSL)

Management of Primary CNS Lymphoma
Presenter: Dr. Narayan Adhikari
Junior Resident
Moderator: Dr. Ahitagni Biswas
Department of Radiation Oncology
Dr. B. R. Ambedker Institute Rotary Cancer Hospital
AIIMS, New Delhi

Overview
• Introduction
• Management• Approach
• Investigations
• Treatment
• Review of literature
• Recent advances

Introduction
Primary Spinal Lymphoma
Primary Leptomeningeal Lymphoma
Primary Intraocular Lymphoma
Primary Cerebral Lymphoma

Epidemiology
1% of all intracranial tumours
4% of all intracranial tumours
Sarkar C, Sharma MC, Deb P, Singh R, Santosh V, Shankar SK. Primary central nervous system lymphoma--a hospital based study of incidence and clinicopathological features from India (1980-2003). J Neurooncol. 2005;71(2):199-204
Villano JL, Koshy M, Shaikh H, Dolecek TA, McCarthy BJ. Age, gender, and racial differences in incidence and survival in primary CNS lymphoma. Br J Cancer. 2011;105(9):1414-1418

Pathogenesis
Local malignant transformation
Systemic transformation?


• Focal deficits (70%)• Neuropsychiatric symptoms (43%)• Raised intracranial pressure (33%)• Seizures (14%)• Ocular Symptoms (4%)• Headache• Confusion• Lethargy
Clinical Features
Bataille B, Delwail V, Menet E, et al. Primary intracerebral malignant lymphoma: report of 248 cases. J Neurosurg 2000; 92:261

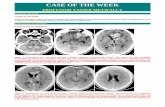
NeuroimagingSites of DiseaseBrain hemispheres (38%)Thalamus/basal ganglia (16%)Corpus Callosum (14%)Periventricular region(12%)Cerebellum (9%)Eyes (5-20%)Meninges (16%)Spinal cord (1%)Cranial and Spinal nerves (<1%)
Kuker W, et al: Primary central nervous system lymphomas (PCNSL): MRI features at presentation in 100 patients. J Neurooncol 72:169-177, 2005

• Tumefactive demyelination• Glial tumours• Brain Metastasis• Cerebral Abscess• Secondary CNS lymphoma
Differentials

Stereotactic biopsy followed by histopathological examination
Diagnosis
• 95% DLBCL• Molecular subtypes
Germinal center, Activated B cell and Type 3
• Overlapping state of differentiation
• EBV associated in immunocompromised
Histopathological findings in primary CNS lymphoma. (A) Hematoxylin and eosin stain demonstrates a population of mitotically active medium-to-large-sized lymphoid cells with large vesicular nuclei and prominent nucleoli mixed with small mature lymphocytes with a distinct propensity to cluster around medium-sized vessels. (B) Immunohistochemistry using a monoclonal anti-CD20 antibody reveals clusters of highly pleomorphic B lymphocytes. (C) Immunohistochemistry using an anti-CD3 antibody highlights the dense T-cell infiltrate within B-cell PCNSL. Tumor cells are indicated by black arrows. (D) The large fraction of mitotically active cells within the tumor can be visualized by immunohistochemistry with the Ki-67 antibody

Workup
Central review of pathologyImmunophenotypingEBV-LMP-1Bone Marrow Biopsy+ Aspirate CSF cytology+ Biochemistry
History and Clinical examinationOphthalmic examination (Slit lamp and indirect ophthalmoscopy)Neuropsychological evaluation
Complete blood countsLiver function Tests, Kidney Function Tests, ElectrolytesSerum LDHHIV serology and viral markers
Contrast enhanced MRI brainCECT neck+ chest+ abdomenUltrasonography of testis in male
Abrey LE, et al: Report of an international workshop to standardize baseline evaluation and response criteria for primary central nervous system lymphoma. J Clin Oncol 23:5034-5043, 2005

• Age ≤60 vs >60 years• PS 0-1 vs ≥2• Serum LDH (normal vs elevated)• CSF protein (normal vs elevated)• Deep regions (no vs yes)
Prognostic Factors
Ferreri AJ et al: Prognostic scoring system for primary CNS lymphomas: The International Extranodal Lymphoma Study Group experience. J Clin Oncol 21:266-272, 2003
Score OS at 2 years0 or 1 80%
2 or 3 48%
4 or 5 15%

• Exclusion of Systemic disease• Extension of the disease to eyes, CSF• Treatment feasibility in view of patient
characteristics• Prognostic factors• Recurrence
Therapeutic Decision

• The optimal treatment strategy is controversial• High dose methotrexate based polychemotherapy
is the mainstay of treatment• Role of radiation has been argued upon but it is
recommended in young patients• The role of rituximab is investigational
Treatment

Ferreri, how I treat PCNSL, bloodjournal, 2015

• Inj Methotrexate 2.5 gm/m 2 IV on weeks 1, 3, 5, 7 and 9• Inj Vincristine 1.4 mg/m 2 IV on weeks 1, 3, 5, 7, 9• Cap Procarbazine 100 mg/m 2 /day per oral for 7 days on
weeks 1, 5, 9• Inj Methotrexate 12 mg intrathecal on weeks 2, 4, 6, 8, 10• Tab Leucovorin 20 mg per oral every 6 h for 12 doses on
weeks 1, 3, 5, 7 and 9 following high dose methotrexate• Tab Dexamethasone per oral tapering dose for 7 days (16,
12, 8, 6, 4, 2 mg on weeks 1-6)• Whole brain radiotherapy (WBRT) 45 Gray in 25 fractions
over 5 weeks (weeks 11-15)• Inj Cytarabine 3 gm/m 2 /day IV for 2 days on weeks 16 and
19
DeAngelis Protocol

Radiation Therapy
German Helmet technique

• Whole Brain is treated due to multifocal and infiltrative nature of tumour
• Left and right opposed lateral fields with 6-10mv photons
• Anterior temporal lobe, cribiform plate, posterior aspect of eyes included
• Divergence beam to lens reduced by placing isocenter anteriorly between the lateral canthi or if anterior fields are made coplanar
• Inferior field edge in lower border of C2 vertebrae
• If ocular involvement both globes included for a portion of treatment to receive a dose of 30-36 Gy
• No role of CSI except for palliation in case of spinal involvement
Radiation Therapy

• Acute Adverse Effects- alopecia, erythema and dry desquamation of the scalp. Some experience fatigue, headache and inflammation of the external auditory canal or middle ear
• Patients requiring treatment of the eye are likely to experience conjunctival irritation and dry eye.
• These acute effects typically resolve within 6 – 8 weeks of completion of WBRT
Radiation Toxicity

• Late adverse effects: neurocognitive decline, sensorineural hearing loss, permanent alopecia
• Those whose eyes are treated- cataracts, chronic dry eye
• The risk of neurocognitive dysfunction increases with age, total RT dose and co-administration of chemotherapy
Radiation Toxicity

Review of literature

Review of literatureSN Study N Median
Age (years)
Chemotherapy WBRT/ Boost (Gy)
Response PFS OS Neurotoxicity
Results and Conclusions
1 RTOG 83-15, 1992
41 66 None 40/20 CR=62% NS 48% 1 year, 28% 2 years
NS KPS (p<0.001) and age (p=0.001) significant prognostic factors
2 RTOG 88-06, 1996
54 NS (>60) CHOD (2-3 cycles)
41.4/ 18
CR=19%, PR=48%, SD=6%, PD=15%
Median 9.2 months
42% 2 years
1 grade 5 encephalomalacia
Age (p=0.005 and KPS (p=0.057) were significant prognostic factors, no survival benefit of adding CHOD chemotherapy to RT, compared with RTOG 83-15, p=0.53)
3 Batchelor et al, 2003
23 60 MTX 8gm/m2, upto 8 cycles, maintenance upto 11 cycles
- CR=52%PR=22%
12.8 months
>23 months
modest Radiological response of 74% with grade 3 toxicity 8/25 and grade 4 toxicity 4/25
4 Abrey et al, 2000
52 65 MTX(3.5gm/m2) ,Vin(1.4mg/m2), Pro(100mg/m2), Cyt
45 vs none
Objective response rate to induction chemotherapy=90%
NS Median 60 months
13 In patients >60 yrs, OS similar with or without RT, late neurotoxicity more common in those who received RT (p=0.00004), young patients both OS and PFS not reached during publication but they fared well



SN Study N Median Age (years)
Chemotherapy WBRT/ Boost (Gy)
Response PFS OS Neurotoxicity Results and conclusions
5 RTOG 93-10, 2002
102 56.5 MTX (IV 2.5gm/m2 and IT 12mg), Vin,Pro, Cyt
45 later amended to 36 in CR group
CR=58%, PR=36% to chemotherapy Response rate=94%
Median 2 years
36.9 months 64% 2 years, 52% 3 years,32% 5 years
12 (15%) severe delayed neurotoxicity, 8Deaths
HDMTX- high response rate, plus RT- improved survival compared to previous studies of RT alone
6 Bessell et al, 2002
57
59 CHOD x 1, BVAM x 2
45 vs. 30.6
CR at the end of all t/t 68% and 77%
NS 36% 5 years
In 1-year survivors: 0/13 (30.6 Gy);1/12 (45 Gy and <60 years old);6/10 (45 Gy and ≥ 60 years old)
In <60 years who achieved CR, 3-year OS 92% v 60%, P =.04), 3 year relapse risk 25% vs 83% (p=0.01)
7 Ferreri et al, 2009
79 18-75 MTX 3.5g/m2 d1, Cyt 2g/m2 bd d2-3
36-45 CR 18% vs 46%, ORR 40% vs 69% in MTX only vs MTX+Cyt resp.
4 vs 8 months (median failure free survival)
10 vs 32 months
NS Combination of methotrexate with cytarabine produces better CR (0.006) ORR (p=0.009), with increased toxicity
8 RTOG 02-27, 2013Phase II results
53 57.5 HDMTX(3.5g/m2), TMZ, RTX
36hyperfractionated
ORR 37.7% post chemo
63.6% 2 years
80.8% 2 years
NS Compared to RTOG 93-10, significant benefit in OS(0.006) and PFS (p=0.03)

SN Study N Median Age (years)
Chemotherapy WBRT/ Boost (Gy)
Response PFS OS Neurotoxicity Results and conclusions
9 Rubenstein et al(CALGB50202), 2013
44 NS MTX (8g/m2), TMZ(150mg/m2 day7-11, odd cycles), RTX(375mg/m2, d3), only CR- consolidtn with EA
- CR 66% 52 m, 64%, 57% 47% at 1,2 and 4 years resp.
Estimated 75%, 70% and 65% at 1,2 and 4 years resp.
NS High Bcl6 expression correlated with shorter survival, patient above 60 did as well as younger patients
10 Ferreri et al, 2011 retrospective
33 55 MTX based Whole brain: 30-45 Gy, tumour bed: 36 to 54 Gy
5 year failure free survival 51%
5 year OS 54%
Significantly less neurologic impairment, as measured by MMSE, in patients treated with 30 – 36Gy vs. ≥ 40 Gy WBRT( p = 0.05)
WBRT doses ≥40 Gy not asso. with improved disease control compared to dose of 30 - 36 Gy, 5-year FFS, 51% vs. 50%; p = 0.26), WB and tumour bed RT doesnot have impact on survival
11 Omuro et al, 2011
64 47 (all <60, young), median KPS 70
MTX (3 g/m2), CCNU, pro, methylpred and IT MTX, cyt, and methylpred
CR-5 more cycles of CT, <CR, 27 WBRT, 29 HDC with SC rescue
ORR after induction chemo 87% (CR 54%, PR33%)
12 m 63 m 5 (none treated with chemo only)
Deferring WBRT in chemosensitiv pts compromises PFS not OS. As objective is cure in this age group, omitting WBRT may not be best strategy


• 280 receiving HDMTX (>1gm/m2) from 19 prospective series• No difference in OS between mono CHT and combination CHT
(p=0.38)• MTX ≥3gm/m2 (p=0.04), thiotepa (p=0.03) and intrathecal CHT
(p=0.03) improved survival and nitrosoureas (p=0.01) correlated with worse survival
• In multivariate analysis, limited to patient receiving ≥ 3 gm/m2 MTX, only the addition of cytarabine improved OS
• A RT dose ≥ 40 Gy to the whole brain or tumor bed did not improve OS• RT delay had no negative impact on survival• Similar findings were reported in another restrospective analysis by
Ferrari et al on 370 patients
Findings


• 81 patients analysed, 54 HDMTX only, 27 HDMTX+R• CR- 36% vs 73% (p=0.0145)• Median PFS 4.5 months vs 26.7 months (p=0.003)• Median OS 16.3 months vs not reached (p=0.01)• A retrospective study by Gregory et al in 120 patients
published in Neuro Oncol showed addition of rituximab is associated with increased OS.
• Care must be given to chances of Hepatitis B reactivation in HBsAg positive patients when given Rituximab
Findings





3

Limitations• Poor protocol adherence• 40% excluded from primary analysis• Low statistical power (60%)• Low accrual period• Suboptimal chemotherapy use• Inclusion of small centers with low experience in treating PCNSL• Lack of quality assurance• Insufficient neuropsychological evaluation• Dr. Lisa DeAngelis pointed out that competing risks analysis,
adjusting for the effect of relapse, demonstrates that approximately 24% of patients develop neurotoxicity at 5 years after WBRT, while the risk of PCNSL recurrence is twice the risk of neurotoxicity, so WBRT cannot be safely excluded from upfront management.








Study N Median Age (years)
Chemotherapy WBRT/ Boost (Gy)
Response PFS OS Neurotoxicity Results and conclusions
Shah et al, 2007
30 57 RTX, HDMTX, Pro,Vin, Cyt
23.4 vs. 45
ORR 93% 2 year PFS 57%, median PFS= 40 months
2 year OS 67%
None In CR patients with rdWBRT, 2 year PFS and 2 year OS 79% and 89% resp.


Study N Median Age (years)
Chemotherapy WBRT/ Boost (Gy)
Response PFS OS Neurotoxicity Results and conclusions
Morris et al, 2013
52 60 RTX, HDMTX, Pro,Vin, Cyt
23.4 vs. 45
CR 60%Objective response rate 95%
Median 3.3 years (7.7years for CR/23.4 Gy)
Median 6.6 years (notreached for CR/23.4 Gy)
None, except decreased motor speed(CR/23.4 Gy)
R-MPV with rdWBRT in CR is associated with high responserates, long-term disease control, and minimal neurotoxicity

HDC+ASCTStudySoussain et al
Soussain et al
Brevet et al
Colombat et al
Abrey et al
Montemurro et al
Cheng et al
Illerhaus et al
Illerhaus et al
araC indicates cytarabine; BCNU, carmustine; BEAM, carmustine, etoposide, cytarabine, and melphalan; Bu, busulfan; Cy, cyclophosphamide; IFO, ifosfamide; MBVP(regimen), methotrexate, carmustine, etoposide, and methylprednisolone; OS, overall survival; TRM, treatment-related mortality; TT, thiotepa; VP16, etoposide; and WBRT,whole-brain irradiation.*Only for patients not achieving a complete remission.†One patient received the treatment as salvage therapy

• Systemic high dose methotrexate based chemotherapy + Local therapy
• Local therapy either intravitreal chemotherapy with methotrexate or rituximab or ocular radiation upto 40 Gy
Primary Intraocular Lymphoma


Domain Test DescriptionAttention/Executive (Digits Forward
And Backward; WAIS-III)
Auditory attention
Trail Making Test (Parts A and B) Psychomotor speed (A); sequencing (B)-alternate form available(flexibility index = B-A)
Brief Test of Attention Auditory working memoryVerbal memory Hopkins Verbal Learning Test—Revised 12-word list:
Three learning/recall trialsDelayed recallRecognition (discrimination index)Six alternate forms
Motor Grooved Pegboard Test Motor speed and dexterity (dominant and nondominant hand)
Quality of life EORTC-QLQ 30 30-item self-report scale (physical, social, emotional, cognitive status)
BCM 20 20-item self-report scale (tumor and treatment-related symptoms)
Premorbid IQ Estimation Barona Index Weighted composite score on the basis of age, gender, race, residence, education, and occupation
Neurocognitive assessment
Correa DD, Maron L, Harder H, et al. Cognitive functions in primary central nervous system lymphoma: literature review and assessment guidelines. Ann Oncol Off J Eur Soc Med Oncol ESMO. 2007;18(7):1145-1151

• CR- complete disappearance and not receiving corticosteroids in last 2 weeks, no ocular lymphoma, negative CSF in previously positive patients
• CRu- Meets criteria of CR but continues to require corticosteroids
• PR- at least 50% decrease in contrast enhancing lesion, decrease in vitreal cell count, retina/optic nerve infiltration, CSF may show malignant disease
• SD- not meeting criteria of CR, CRu, PR, PD• PD- >25% increase in CE lesions, new site, increase in
vitreal cell count
Response Criteria
Abrey LE, Batchelor TT, Ferreri AJ, et al. Report of an international workshop to standardize baseline evaluation and response criteria for primary CNS lymphoma. J Clin Oncol 2005; 23:5034

• Leptomeningeal disease- MTX 12mg weekly intrathecally or through ommaya reserviour
• Older patients- dose adjustment, RT may be deffered till progression, proper monitoring of kidney functions
• In HIV positive: MTX based chemotherapy with anti retroviral therapy with or without radiation
Special scenarios


• Retreatment with methotrexate (ie, 3 to 8 g/m2 ) or methotrexate based combination chemotherapy if there has been a prior complete remission with this agent
• Alternative chemotherapy regimens (eg, topotecan, cytarabine, temozolomide, thiotepa, pemetrexed including high dose chemotherapy followed by autologous hematopoietic cell transplantation (HCT)
• Whole brain radiation therapy in previously nonirradiated patients, stereotactic radiotherapy may be an option for patients who have received whole brain radiation
Treatment of refractory or relapsed disease


Targeted therapies
Primary CNS
Lymphomapathways
B-cell receptor
NFkB
IRF4
PIM kinase
s
JAK/STAT
BCL-6
Ponzoni M, Issa S, Batchelor TT, Rubenstein JL. Beyond high-dose methotrexate and brain radiotherapy: novel targets and agents for primary CNS lymphoma. Ann Oncol Off J Eur Soc Med Oncol ESMO. 2014;25(2):316-322.

Conclusions• High dose methotrexate based chemotherapy with
consolidation cytarabine is the mainstay of treatment• WBRT should be included in the treatment especially in
young patients (<60 years), response adapted approach may be beneficial
• Rituximab may be tried in upfront management or can be reserved for recurrence
• Local therapy is recommended for Intraocular lymphoma, or leptomeningeal involvement along with systemic therapy
• Targeted therapies are investigational

Immuno-competent patients of newly diagnosed primary CNS lymphoma
Five 14-day cycles of induction chemotherapy with methotrexate, procarbazine, and vincristine (MVP)
Investigations
Response AssessmentNeuropsychological & QoL assessment
Complete Response Partial Response/Stable Disease/Progressive disease
Reduced dose WBRT 23.4Gy/13#/2.5 weeks
Standard dose WBRT 45Gy/25#/5 weeks
Consolidation ChemotherapyInj cytarabine (Ara-C), 2 cycles 1 month apart
Response assessment-CMRI brain every 3 monthsNeuropsychological & QoL assessment 6 monthly post T/t completion