The Benefits of Early and Aggressive Lowering of LDL- Cholesterol › wp-content › uploads ›...
Transcript of The Benefits of Early and Aggressive Lowering of LDL- Cholesterol › wp-content › uploads ›...

The Benefits of Early and
Aggressive Lowering of LDL-
CholesterolJune 18, 2020 at 2:00 PM EST
Presenter: Nihar Desai, MDModerator: Andrea Baer, MS, BCPA

Mended Hearts’ mission is “to inspire hope and improve the quality of life of heart patients and their families through ongoing peer-to-peer support, education and advocacy.”
The National Lipid Association’s (NLA) mission is “to enhance the practice of lipid management in clinical medicine.”
The Foundation of the NLA’s mission is “to improve the welfare of patients and families affected by cholesterol and triglyceride problems.”

Andrea Baer, MS, BCPAExecutive DirectorMended Hearts
Nihar R. Desai, MD, MPHAssociate Professor of Medicine, Yale School of MedicineAssociate Chief, Section of Cardiovascular MedicineInvestigator, Center for Outcomes Research and Evaluation

Beyond the Numbers:The Benefits of Early and Aggressive
Lowering of LDL Cholesterol
Nihar R. Desai, MD, MPHAssociate Professor of Medicine, Yale School of Medicine
Associate Chief, Section of Cardiovascular MedicineInvestigator, Center for Outcomes Research and Evaluation

Disclosures
• Grant support: Agency for Healthcare Research and Quality.
• Support from the Centers for Medicare and Medicaid Services to develop and maintain performance measures that are used for public reporting and payment programs.
• Research support and consulting from Amgen, Boehringer Ingelheim, Novartis, SC Pharmaceuticals.
• Research support from Amgen for several clinical studies of evolocumab.
• Medical review committee for Anthem.

Roadmap
• Review the relationship between LDL-C, atherosclerotic cardiovascular disease and adverse events.
• Emphasize the importance of early management of hypercholesterolemia for preventing heart disease and aggressive LDL-C control for avoiding any further events.
• Discuss recent clinical trial results that have informed our understanding of benefits of lowering LDL-C with statin and non-statin therapies and how they have shaped practice guidelines.

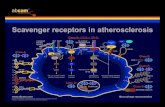
The Central Role of Lipids in Atherosclerosis

The Central Role of Lipids in Atherosclerosis
0
10
20
30
40
50
60
Smoking Lack ofExercise
Fruit/Vegetable
Consumption*
Alcohol All Psychosocial
Factors†
PA
R (
%)
Lifestyle Factors
Population Attributable Risk From Various Modifiable Risk Factors on Acute MI
(Overall Population)1
Hypertension Diabetes
RISK FACTORS
Lipids‡Abdominal Obesity
PAR=population attributable risk, which indicates the number or proportion of cases that would not occur in a population if the risk factor were eliminated.2
PARs from individual risk factors are reported. Note that the sum of individual PARs is greater than 100% because “cases” can simultaneously be attributed to more than one risk factor and be
counted twice. PAR percentages reflected here do not indicate the amount of risk that would decrease by addressing the identified risk factors.1
*Irregular consumption of fruits and vegetables; †A model-dependent index combining positive exposure to depression, perceived stress at home or work (general stress), low focus of control,
and major life events, all referenced against non-exposure for all 5 factors. ‡ApoB/ApoA1 ratio; INTERHEART study; n=15,152 patients and 14,820 controls in 52 countries.1
Apo=apolipoprotein; MI=myocardial infarction.
1. Yusuf S, et al. Lancet. 2004;364:937-952.. 2. Rockhill B, et al. Am J Public Health. 1998;38:15-19.

The Cumulative Effect of Hypercholesterolemia

ASCVD Risk Estimator

2019 ACC/AHA Guideline on 1⚬
Prevention of CV Disease

2019 ACC/AHA Guideline on 1⚬
Prevention of CV Disease

Reducing LDL-C Reduces CV Events
Primary prevention trials
Secondary prevention trials
50 70 110 130 150 170 19090 210
% P
atie
nts
wit
h C
HD
Eve
nt
LDL-C achieved mg/dL
CARE-Rx
4S-Rx
LIPID-PL
4S-PL
CARE-PL
LIPID-Rx
AFCAPS-Rx
WOSCOPS-Rx
WOSCOPS-PL
AFCAPS-PL
25
20
15
10
5
0
ASCOT-PL
ASCOT-Rx
HPS-Rx
HPS-PLHPS
LRC-PLLRC-Rx
POSCH-PL
POSCH-Rx
TNT-80A
TNT-10A
Ballantyne CM. Am J Cardiol. 1998;82:737-743. O’Keefe JH, et al, J Am Coll Cardiol. 2004;43:2142-2146.
PL = placeboRx = active treatment
30
Prove-IT TIMI 22 – 80A

Statin v. Control
0.4 0.6 0.8 1 1.2 1.4
Nonfatal MI
CHD death
Any major coronary event
CABG
PTCA
Unspecified
Any coronary revascularisation
Ischaemic stroke
Haemorrhagic stroke
Unknown stroke
Any stroke
Any major vascular event
2310 (0.9%)
1242 (0.5%)
3380 (1.3%)
816 (0.3%)
601 (0.2%)
1686 (0.6%)
3103 (1.2%)
987 (0.4%)
188 (0.1%)
555 (0.2%)
1730 (0.7%)
7136 (2.8%)
3213 (1.2%)
1587 (0.6%)
4539 (1.7%)
1126 (0.4%)
775 (0.3%)
2165 (0.8%)
4066 (1.6%)
1225 (0.5%)
163 (0.1%)
629 (0.2%)
2017 (0.8%)
8934 (3.6%)
0.74 (0.69 - 0.78)
0.80 (0.73 - 0.86)
0.76 (0.73 - 0.79)
0.76 (0.69 - 0.83)
0.78 (0.69 - 0.89)
0.76 (0.70 - 0.83)
0.76 (0.73 - 0.80)
0.80 (0.73 - 0.88)
1.10 (0.86 - 1.42)
0.88 (0.76 - 1.02)
0.85 (0.80 - 0.90)
0.79 (0.77 - 0.81)
No. of events (% pa)
Statin ControlRelative risk (CI) per
mmol/L LDL-C reduction
Statin better Control better
99% or 95% CI
Reducing LDL-C Reduces CV Events

Primary prevention trials
Secondary prevention trials
50 70 110 130 150 170 19090 210
% P
atie
nts
wit
h C
HD
Eve
nt
LDL-C achieved mg/dL
CARE-Rx
4S-Rx
LIPID-PL
4S-PL
CARE-PL
LIPID-Rx
AFCAPS-Rx
WOSCOPS-Rx
WOSCOPS-PL
AFCAPS-PL
25
20
15
10
5
0
ASCOT-PL
ASCOT-Rx
HPS-Rx
HPS-PL
HPS
LRC-PLLRC-Rx
POSCH-PL
POSCH-Rx
TNT-80A
TNT-10A
Ballantyne CM. Am J Cardiol. 1998;82:737-743. O’Keefe JH, et al, J Am Coll Cardiol. 2004;43:2142-2146.
PL = placeboRx = active treatment
30
Prove-IT TIMI 22 – 80A
The Big Questions…

Some Answers…IMPROVE-IT Trial

Some Answers…IMPROVE-IT Trial

Some Answers…IMPROVE-IT Trial

Monoclonal Antibody Against PCSK9 Blocks The PCSK9/LDL-R Interaction
1. Chan JC, Piper DE, Cao Q, et al. Proc Natl Acad Sci U S A. 2009;106:9820-9825.

GLAGOV Trial
Global Assessment of Plaque Regression with a PCSK9 Antibody as Measured by Intravascular Ultrasound
• Prior intravascular ultrasound (IVUS) trials have shown that statins slow progression or induce regression of coronary disease in proportion to the magnitude of LDL-C reduction.
• No other LDL-lowering therapy has shown regression in an IVUS trial.
• GLAGOV examines whether the addition of evolocumab, to a background of statin therapy, can reduce the burden of atherosclerosis as assessed by IVUS.
Nicholls, SJ et al. JAMA. 2016;316(22):2373-2384.

GLAGOV Trial: Study Design
423 statin completers 423 evolocumab completers
61 patients did not complete
Follow-up IVUS of originally imaged “target” vessel (n=846)
Stable, optimized statin dose for 4 weeks with LDL-C >80 mg/dLor 60-80 mg with additional high risk features
Intravascular ultrasound via motorized pullbackat 0.5 mm/sec through >40 mm segment
968 patients at 197 global centers with symptomatic CAD and other high riskfeatures and coronary angiography showing 20-50% stenosis in a target vessel
Statinmonotherapy
Statin plus monthly SCevolocumab 420 mg
18 monthstreatment
61 patients did not complete
Nicholls, SJ et al. JAMA. 2016;316(22):2373-2384.

GLAGOV Trial: LDL-C Effect
-80
-70
-60
-50
-40
-30
-20
-10
0
10
20
0 8 16 24 32 40 48 56 64 72 80 88
LD
L-C
Ch
an
ge f
rom
Bas
eli
ne (
mg
/dL
)
Study Week
Mean LDL-C 93.0 mg/dL
Mean LDL-C 36.6 mg/dL
Change from baseline 3.9%
Change from baseline -59.8%
Nicholls, SJ et al. JAMA. 2016;316(22):2373-2384.

GLAGOV Trial: Primary Endpoint
0.05
-0.95
-1.2
-1
-0.8
-0.6
-0.4
-0.2
0
0.2
Changein Percent Atheroma
Volume(%)
Statinmonotherapy
Statin-evolocumab
P < 0.0001
P = NS
P <0.0001
Nicholls, SJ et al. JAMA. 2016;316(22):2373-2384.

GLAGOV Trial: Achieved LDL and Plaque Regression
P = NS
P <0.0001
Ch
ange
Pe
rce
nt
Ath
ero
ma
Vo
lum
e (
%)
On-Treatment LDL-C (mg/dL)
Nicholls, SJ et al. JAMA. 2016;316(22):2373-2384.

FOURIER Trial: Study Design27,564 high-risk, stable patients with established CV disease
(prior MI, prior stroke, or symptomatic PAD)
Screening, Lipid Stabilization, and Placebo Run-in
High or moderate intensity statin therapy (± ezetimibe)
LDL-C ≥70 mg/dL or
non-HDL-C ≥100 mg/dL
RANDOMIZED
DOUBLE BLINDEvolocumab
140 mg Q2W or 420 mg QM
Placebo
Q2W or QM
Follow-up Q 12 weeks
Sabatine MS et al. Am Heart J 2016;173:94-101

FOURIER: LDL-C Reductions
100Placebo
70
90
80L
DL
Ch
ole
ste
rol(
mg
/dl)
60
Absolute reduction: 56 mg/dl (95%CI 55-57)50
40
30
59% mean reduction (95%CI 58-60), P<0.00001
10
0
0 12 24 36 48 60 72 84 96 108 120 132 144 156 168
Weeks
20Evolocumab
(median 30 mg/dl, IQR 19-46 mg/dl)
Sabatine MS et al. NEJM 2017;177.

FOURIER Trial:Clinical Outcomes
9%
10%
Hazard ratio 0.80
9.9%
7%
6%
5%
8%C
VD
ea
th,
MI,
or
Str
ok
e(95% CI, 0.73-0.88)
P<0.00001Placebo
7.9%
3%
2%
1%
0%
4% Evolocumab
12 18 24
Months from Randomization
0 6 30 36
Sabatine MS et al. NEJM 2017;177.

ODYSSEY Outcomes: Study Design
Post-ACS patients (1 to 12 months)
Run-in period of 2−16 weeks on high-intensity or maximum-tolerated dose of atorvastatin or rosuvastatin
At least one lipid entry criterion met
Placebo SC Q2WAlirocumab SC Q2W
Randomization
Schwartz GG, et al. Am Heart J 2014;168:682-689.e1.
Patient and investigators remained blinded to treatment and lipid levels for the entire duration of the study

37.642.3
53.3
93.3
55.7mg/dL
Approximately 75% of months of active treatment were at the 75 mg dose
96.4101.4
48.1mg/dL
105
90
75
60
45
30
15
00 4 8 12 16 20 24 28 32 36 40
Months Since Randomization
Excludes LDL-C values after premature treatment discontinuation or blinded switch to placebo
44 48
Me
an
LD
L-C
(mg
/dL)
–62.7%
54.1mg/dL
–61.0%–54.7%
Placebo
Alirocumab
ODYSSEY Outcomes: LDL-C Reductions
Schwartz, GC et al. NEJM 2018; Nov. 29 [Epub ahead of print]

ARR 1.6%MACE: CHD death,
non-fatal MI,
ischemic stroke, or
unstable angina requiring
hospitalization
11.1%
9.5%
ODYSSEY Outcomes: Clinical Outcomes
Schwartz, GC et al. NEJM 2018; Nov. 29 [Epub ahead of print]

Putting It All TogetherFOURIER Trial: Achieved LDL-C
0%
3%
6%
9%
1 4 7 10 13 16 19 22 25 28
≥100
70-99
50-69
20-49
<20
LDL-C (mg/dL)
at 4 wks10%13%
31%
25%
Adj RRR Ref.
Months after Randomization
Ka
pla
n-M
eie
r E
ven
t R
ate
LDL-C=low-density lipoprotein cholesterol; RRR=relative risk ratio. Giugliano RP et al. Lancet. 2017;390:1962-71.

LDL‐
C(m
g/d
L)200
180
160
140
120
100
80
60
40
20
01994 1996‐2002 2004‐2005 2015 2017
Evolving Paradigm of LDL-C Management

They Knew It All Along…
Adapted from Nobel Prize Lecture, Stockholm, Sweden, 1985.
Science 1986;232:34.

*Progressive ASCVD, including UA that persists after achieving an LDL-C < 70 mg/dL (1.8 mmol/L), or established clinical ASCVD in individuals with diabetes, CKD stage 3 or 4, and/or HeFH, or in individuals with a history of premature ASCVD (< 55 years of age for males or < 65 years of age for females). †In very high risk ASCVD,* use an LDL-C threshold of 70 mg/dL (1.8 mmol/L) to consider the addition of nonstatins to statin therapy. A
threshold is the point/trigger at which intensification of therapy may be considered. Additional AHA/ACC guidelines were published in 2013 but did not provide a recommendation for target LDL-C levels to reduce the ASCVD risk.14
1. Goodman DS, et al. Arch Intern Med. 1988;148:36-69. 2. Grundy SM, et al. JAMA. 1993;269:3015-3023. 3. NCEP. Circulation. 2002;106:3143-3421. 4. Jellinger PS, et al. Endocr Pract. 2017;23(suppl 2):1-87. 5. Grundy SM, et al. J Am College Cardiol. 2019;73:e285- e350. 6. Reiner Z, et al. Eur Heart J. 2011;32:1769-1818. 7. Catapano AL, et al. Eur Heart J. 2016;37:2999-3058. 8. Mach F, et al. Eur Heart J. 2020;41:111-188. 9. Wood D, et al. Eur J Gen Pract. 1999;5:154-161. 10. De Backer G, et al. Atherosclerosis. 2004;173:381-391. 11. Graham I, et al. Eur Heart J. 2007;28:2375-2414. 12. Jellinger PS, et al. Endocr Pract. 2012;18(suppl 1):1-78. 13. Jacobson TA, et al. J Clin Lipidol. 2014;8:473-488. 14. Stone NJ, et al. J Am Coll Cardiol. 2014;63(25 pt B):2889-2934.
1985 1990 1995 2000 2005 2010 2015 2020 2025
NCEP ATP1-3
AACE/ACE (extreme* ASCVD risk)4
AHA/ACC5,†
ESC/EAS (very high CV risk)6-8
ESC/EAS (high CV risk)6-11
AACE (very high CV risk)12
NLA (very high risk)13
0
20 (0.5)
40 (1.0)
60 (1.5)
80 (2.0)
100 (2.5)
120 (3.0)
140 (3.5)
Historical Perspective of LDL-C Targets/Thresholds as Recommended by Globally Recognized Guidelines1-13
LD
L-C
, m
g/d
L (
mm
ol/
L)
Year
Evolving Paradigm of LDL-C Management

2018 ACC/AHA Lipid Guidelines

2018 ACC/AHA Guidelines
Table 4. Very High-Risk* of Future ASCVD Events
Major ASCVD Events
Recent ACS (within the past 12 mo)
History of MI (other than recent ACS event listed above)
History of ischemic stroke
Symptomatic peripheral arterial disease (history of claudication with ABI
<0.85, or previous revascularization or amputation)

2018 ACC/AHA Guidelines
High-Risk Conditions
Age ≥65 y
Heterozygous familial hypercholesterolemia
History of prior coronary artery bypass surgery or percutaneous coronary
intervention outside of the major ASCVD event(s)
Diabetes mellitus
Hypertension
CKD (eGFR 15-59 mL/min/1.73 m2)
Current smoking
Persistently elevated LDL-C (LDL-C ≥100 mg/dL [≥2.6 mmol/L]) despite
maximally tolerated statin therapy and ezetimibe
History of congestive HF
Table 4. Very High-Risk* of Future ASCVD Events

Summary and Conclusions
• Reducing LDL-C with statin therapy is the cornerstone of hyperlipidemia management.
• It is important to start early to prevent heart disease and critical to aggressively lower LDL-C to prevent any further events.
• Clinical trials of ezetimibe and PCSK9 inhibitors have demonstrated additional reductions in adverse events with additional reductions in LDL-C.
• The ACC/AHA guidelines recommend additional lipid lowering therapy in patients with very high risk ASCVD who have LDL > 70mg/dL.

Beyond the Numbers:The Benefits of Early and Aggressive
Lowering of LDL Cholesterol
Nihar R. Desai, MD, MPHAssociate Professor of Medicine, Yale School of Medicine
Associate Chief, Section of Cardiovascular MedicineInvestigator, Center for Outcomes Research and Evaluation

For additional questions, please email:[email protected]
Join us for the next session of the series:Understanding and Interpreting Your Cholesterol Blood TestJune 23, 2020 at 5:00 PM ET
Presenter: Joseph DeBoe, DNPModerator: Andrea Baer, MS, BCPA
This webinar series is brought to you by Mended Hearts, The National Lipid Association (NLA), and The Foundation of the NLA.This activity is supported by Amgen and Esperion