Stratospheric Ozone Depletion Last time, we learned about natural ozone chemistry in the...
-
Upload
maximilian-edwards -
Category
Documents
-
view
218 -
download
0
Transcript of Stratospheric Ozone Depletion Last time, we learned about natural ozone chemistry in the...

Stratospheric Ozone Depletion
• Last time, we learned about natural ozone chemistry in the stratosphere.
• How do anthropogenic emissions of certain chemicals affect the ozone layer?
• Why does the ozone ‘hole’ form over Antarctica?

Anthropogenic Enhancement of the Stratospheric HOx Cycle
From: http://www.ipcc.ch/present/graphics.htm

From: IPCC report, 2007

Anthropogenic Enhancement of the Stratospheric NOx Cycle
From: http://www.ipcc.ch/present/graphics.htm

From: IPCC report, 2007

Chlorine Chemistry in the Stratosphere (ClOx family)
223
2
23
222
2210nm-185
3
OOOO :net
OClOClO
OClOO Cl
ClClCFhClCF
ClCFClhCFCl
• CFCs photodissociate in the stratosphere:

Reservoirs of ClOx
• Reservoirs are transported to the troposphere for removal, or:
MClONOMNOClO
HClCHCH Cl
22
34
ClNO3
3
22
2
NOCl
NOClOhClONO
OH ClOHHCl

Reservoirs of ClOx (Cly)
• ClOx includes:
– Cl + ClO
• Cly includes:
– Cl + ClO + ClONO2 + HCl + Cl2 + ClO2 + Cl2O2 + HOCl + BrCl
• About 99% of the chlorine in the stratosphere is in the form of Cly.
– This chlorine is not active towards O3.

The ClOx Cycle
http://www-as.harvard.edu/people/faculty/djj/book/powerpoints/

Sources of Stratospheric Chlorine• Natural sources:
– HCl: dechlorination of sea-salt, volcanic emissions, biomass burning• Very short lifetime in the troposphere
– CH3Cl: ocean and biomass burning emissions• Lifetime of about 1.5 years
• Anthropogenic sources:– Emissions of CFCs
• No tropospheric sink• Lifetimes on the order of 100 years

What is a CFC?
• Chlorofluorocarbon – contains only carbon, chlorine, and fluorine.
• ‘Rule of 90’ – take CFC-11 for example:– add 11+90 = 101
• First digit is the # of carbon = 1• Second digit is the # of hydrogen = 0• Third digit is the # of fluorine = 1• The remainder is chlorine
CCl3F

Why were CFCs Invented?
• Early refrigerators used toxic chemicals as refrigerants:
– NH3, CH3Cl, SO2.
• CFCs were invented by Thomas Midgley in 1928
– By 1935 over 8 million refrigerators had been sold containing Freon (the trade name for CFCs).

• CFCs were considered incredibly non-toxic:
– Expanded the use of refrigerators and air-conditioning in non industrial settings.
– Many additional uses were discovered:• aerosol propellant• blowing Styrofoam• fire retardant• electronics cleaning• etc.
Why did the use of CFCs Expand?

WMO/UNEPScientific Assessment of Ozone Depletion: 2006
The Rise and Fall of CFCs

The Rise and Fall of CFCs
WMO/UNEPScientific Assessment of Ozone Depletion: 2002

http://www-as.harvard.edu/people/faculty/djj/book/powerpoints/
CFCs in the Statosphere

Mixed Cycles• We have seen the coupling of NOx and HOx:
• NOx is coupled with ClOx:
• HOx is coupled with ClOx:
MHNOMNOOH 32
MClONOMNOClO 22
22
2
OHOClClOHO
ClOHHCl OH

Mixed Cycles• As HOx increases, active NOx decreases,
but active ClOx increases.
• As NOx increases, active ClOx and HOx decreases.
• As ClOx increases, active NOx and HOx decreases.

Mixed Cycles
22233
22
223
23
OOOOO
ClOHhHOCl
OHOClHOClO
OHOOOH
OClOOCl
HOx/ClOx HOx/NOx
OOhO
ONOhNO
OHNONOHO
OHOOOH
23
2
22
223
Ozone destruction Null cycle

Mixed Cycles
22233
23
32
2M
2
223
23
OOOOO
ONOhNO
NOClhClONO
ClONONOClO
ONOONO
OClOOCl
NOx/ClOx NOx/ClOx
OOhO
ONOhNO
NOClNOClO
OClOOCl
23
2
2
23
Ozone destruction Null cycle

BrOx in the Stratosphere
22233
23
23
2
2
OOOOO
OClOOCl
ClBrhBrCl
OBrOOBr
OBrCl
OClOBr
OBrClBrOClO
223
2
23
OOOO :net
OBrOBrO
OBrOOBr
Stratospheric bromine chemistry is analogous to chlorine chemistry.
BrOx/ClOx mixed cycle
BrOx is a more effective catalyst for O3 destruction because its reservoir species are more difficult to form.
y
x
y
x
Br
BrO
Cl
ClO

Sources of BrOx
• Natural:– CH3Br is naturally emitted from the ocean.
• Anthropogenic:– CH3Br is used as an agricultural fumigant.

Methylbromide
WMO/UNEP Scientific Assessment of Ozone Depletion: 2006

Non-Polar StratosphericOzone Depletion
WMO/UNEP Scientific Assessment of Ozone Depletion: 2002

Stratospheric Aerosol Layer• There is a persistent layer of sulfate
aerosols in the lower stratosphere.– The background source is carbonyl sulfide
(COS).– COS is naturally emitted from the ocean’s
surface
• tropospheric concentrations are about 500pptv.

COS in the Stratosphere
4(l)2223
2
SOHOHSOHO,OOH,HSSO,S,
HSCOOH COS
(22%) SOCOO COS
(71%) S COh COS
Since there is no precipitation in the stratosphere,the lifetime of the aerosol is 1-2 years.
The aerosols absorb water to their surface.

Volcanic Aerosols
• Major volcanic eruptions can inject H2SO4 aerosols directly into the stratosphere.
– normal stratospheric aerosol surface area:
0.5 – 1.0µm2 cm-3
– surface area after Mt. Pinatubo erupted:
20µm2 cm-3
• Since there is usually a major volcanic eruption every few years, it is uncommon for the stratospheric aerosol layer to not be influenced by volcanic activity.

Stratospheric Aerosol
WMO/UNEP Scientific Assessment of Ozone Depletion: 2002

• Remember the reaction:
– the lifetime of HNO3 is days
– the lifetime of N2O5 is hours
• The conversion of N2O5 to HNO3 represents a decrease in:
Heterogeneous Chemistry
3(s)252 2HNOOHON
y
x
NO
NO

• If the amount of active NOx is decreased:
– the amount of active ClOx is increased
Mixed Cycle
22 ClONONOClO

Stratospheric Aerosol
WMO/UNEP Scientific Assessment of Ozone Depletion: 2002

Increased UV Radiation
WMO/UNEPScientific Assessment of Ozone Depletion: 2006

CFC Replacements
http://www-as.harvard.edu/people/faculty/djj/book/powerpoints/

Ozone Depleting Chemicals
WMO/UNEPScientific Assessment of Ozone Depletion: 2004

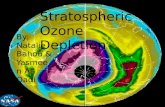
The Ozone Hole
http://www-as.harvard.edu/people/faculty/djj/book/powerpoints/

Antarctic Ozone BulletinNo 8/2005Winter/spring summaryJanuary 2006Global Atmosphere Watch
‘Density’ of Ozone Hole

Area of Ozone Hole
Antarctic Ozone BulletinNo 8/2005Winter/spring summaryJanuary 2006Global Atmosphere Watch

Seasonality of the Ozone Hole
http://www-as.harvard.edu/people/faculty/djj/book/powerpoints/

The Ozone Hole Area
From: NASA

The Ozone Hole
From: NASA

Vertical Structure of the Ozone Hole
Antarctic Ozone BulletinNo 3/2008World Meteorological Organization

The Antarctic Stratosphere
• What is special about the Antarctic stratosphere?
– Polar vortex• Extremely stable westerly circulation that
essentially cuts the Antarctic stratosphere off from the midlatitudes.
• The vortex persists until November (late spring)
– Extremely cold temperatures• As low as 180K
– Its totally dark during the winter (until September)

A New Catalytic Cycle
23
23
222
22
3OhO2
OClOOCl2
OClClhOCl
MOClMClOClO
This cycle does not depend on atomic O•Cl2O2 is easily photolyzed by hν, O2 is not.
•How could so much ClO be produced to account for the observed O3 loss? Since:
22 ClONONOClO

http://www.atm.damtp.cam.ac.uk/people/efs20/media/kiruna/PSCbest.jpg
Polar Stratospheric Clouds

PSCs
• Type I:– mixture of water and nitric acid– form at temperatures under 197K
• Type II:– pure water ice– form at temperatures under 188K

Formation of PSCs
WMO/UNEPScientific Assessment of Ozone Depletion: 2002

PSCs
• PSC help to activate ClOx
• At the same time removing NOx
– disabling the ClONO2 reservoir for ClOx

Heterogeneous Chemistryin the Dark
OHClHClHOCl
HNOHOClOHClONO
HNOClHClClONO
22PSC
3PSC
22
32PSC
2
•During the winter, all of the chlorine reservoirs are converted to Cl2 on the surface of PSCs.
•All of the NOy is removed from the Antarctic stratosphere. Much of the NOy settles out of the atmosphere altogether.

When the Sun Comes Up
23
23
222
22
3OhO2
OClOOCl2
OClClhOCl
MOClMClOClO
Cl2hCl2
•There is no NOx to sequester the ClOx, so O3 destruction goes on unabated.
•Remember, in the mid-latitude stratosphere, about 99% of Cl is in the form of Cly.•During Antarctic spring, almost all Cl is in the form of ClOx.

In the Summer
• The vortex breaks up– This allows mid-latitude NOx and O3 rich air
to mix over Antarctica.
– However, O3 depleted air mixes over populated areas of the southern hemisphere.
• The stratosphere warms and the PSCs evaporate

Ozone Hole Chemistry

Ozone Hole Calendar
http://www-as.harvard.edu/people/faculty/djj/book/powerpoints/

The Smoking Gun

Next Week’s Exam• The exam is on Wednesday, October 7
during the first 50 minutes of regular class time.
• You are allowed to use a calculator (no PDAs or cell phone calculators).– You may not use your calculator to look at
pre-recorded notes – this is considered cheating.
– Don’t forget your calculator, there will be no extras to hand out.

Next Week’s Exam• No aids in addition to a calculator are
allowed.• Do everything possible to avoid even the
suggestion of cheating:– no hats, sunglasses, wandering eyes, etc.
• Partial credit can be given only if your work is well organized and easily readable.
• All calculations need to be worked out on the exam to receive credit, even if you have the correct answer.

Next Week’s Exam
• The form of the exam:– Multiple choice:
• 20-30%
– Short answer• 30-40%
– Worked out calculations• 30-40%
• This exam is worth 22% of your grade.

Next Week’s Exam
• Priority of material on the exam:– Topics covered in lecture– Topics covered by the assigned reading
• You are responsible for material in the readings, even if it was not explicitly covered in lecture.
• Preparing for the exam:– Do the suggested problems– Study the lecture notes– Study text