Pleural effusion
-
Upload
hiba-ashibany -
Category
Documents
-
view
178 -
download
3
Transcript of Pleural effusion


A pleural effusion is an abnormal collection
of fluid in the pleural space resulting from
excess fluid production or decreased
absorption or both. It is the most common
manifestation of pleural disease.
Hemothorax is blood in plueral space .
Chylothorax is chyle (lymph+fat) in pleural
spcae.
Empyema is pus in plueral space .

The pleural space is bordered by the parietal and visceral pleurae. The parietal pleura covers the inner surface of the thoracic cavity, including the mediastinum, diaphragm, and ribs. The visceral pleura envelops all lung surfaces, including the interlobar fissures. The right and left pleural spaces are separated by the mediastinum.

The pleural space plays an important role
in respiration by coupling the movement of
the chest wall with that of the lungs in 2
ways. First, a relative vacuum in the
space keeps the visceral and parietal
pleurae in close proximity. Second, the
small volume of pleural fluid, which has
been calculated at 0.13 mL/kg of body
weight under normal circumstances, serves
as a lubricant to facilitate movement of
the pleural surfaces against each other in
the course of respirations.

Pleural effusion is an indicator of an
underlying disease process that may be
pulmonary or nonpulmonary in origin and
may be acute or chronic. Although the
etiologic spectrum of pleural effusion is
extensive, most pleural effusions are
caused by congestive heart failure,
pneumonia, malignancy, or pulmonary
embolism

Reduction in intravascular oncotic pressure
(eg, hypoalbuminemia due to nephrotic
syndrome or cirrhosis)
Increased capillary permeability or vascular
disruption (eg, trauma, malignancy,
inflammation, infection, pulmonary
infarction, drug hypersensitivity, uremia,
pancreatitis)

Increased capillary hydrostatic pressure in
the systemic and/or pulmonary
circulation (eg, congestive heart failure)
Reduction of pressure in the pleural
space, preventing full lung expansion or
"trapped lung" (eg, extensive atelectasis,
mesothelioma)
Decreased lymphatic drainage or complete
blockage, including thoracic duct obstruction
or rupture (eg, malignancy, trauma)

Increased peritoneal fluid, with migration
across the diaphragm via the lymphatics or
structural defect (eg, cirrhosis, peritoneal
dialysis)
Movement of fluid from pulmonary edema across the visceral pleura
Altered permeability of the pleural membranes (eg, inflammation, malignancy, pulmonary embolus)

Pleural effusions are generally classified as
transudates or exudates, based on the
mechanism of fluid formation and pleural
fluid chemistry. Transudates result from an
imbalance in oncotic and hydrostatic
pressures, whereas exudates are the result
of inflammation of the pleura or decreased
lymphatic drainage.

Transudates causes include the following:
Congestive heart failure
Cirrhosis (hepatic hydrothorax)
Atelectasis - Which may be due to
malignancy or pulmonary embolism
Hypoalbuminemia
Nephrotic syndrome
Myxedema
Constrictive pericarditis

Urinothorax - Usually due to obstructive uropathy
Cerebrospinal fluid (CSF) leaks to the pleura -Generally in the setting of ventriculopleuralshunting or of trauma or surgery to the thoracic spine
Duropleural fistula - Rare, but may be a complication of spinal cord surgery
Extravascular migration of central venous catheter
Glycinothorax - A rare complication of bladder irrigation with 1.5% glycine solution following urologic surgery

common causes of exudates include the following:
Tuberculosis
Parapneumonic .
Malignancy (most commonly lung or breast cancer, lymphoma, and leukemia; less commonly ovarian carcinoma, stomach cancer, sarcomas, melanoma)
Pulmonary embolism
Collagen-vascular conditions (rheumatoid arthritis, systemic lupus erythematosus )
Pancreatitis

Trauma
Esophageal perforation
Radiation pleuritis
Sarcoidosis
Fungal infection
Intra-abdominal abscess
Meigs syndrome (benign pelvic neoplasm with
associated ascites and pleural effusion)
Yellow nail syndrome (yellow nails,
lymphedema, pleural effusions

Drug-induced pleural disease:
Isoniazide.
procainamide
hydralazine
quinidine
nitrofurantoin
methotrexate

The clinical manifestations of pleural
effusion are variable and often are related to
the underlying disease process:
Dyspnea :is the most common symptom
associated with pleural effusion.
Cough: in patients with pleural effusion is
often mild and nonproductive. More severe
cough or the production of purulent or
bloody sputum suggests an underlying
pneumonia or endobronchial lesions

Chest pain :which results from pleural irritation, raises the likelihood of an exudative etiology, such as pleural infection, mesothelioma, or pulmonary infarction.
Additional symptoms
Other symptoms in association with pleural effusions may suggest the underlying disease process. Increasing lower extremity edema, orthopnea, and paroxysmal nocturnal dyspneamay all occur with congestive heart failure.
Night sweats, fever, hemoptysis, and weight loss should suggest TB. Hemoptysis also raises the possibility of malignancy, other endotracheal or endobronchial pathology, or pulmonary infarction. An acute febrile episode, purulent sputum production, and pleuritic chest pain may occur in patients with an effusion associated with pneumonia

History.
Examination:
decreased expansion.
Stony dull percussion note.
Diminished breath sounds.
Decreased tactile vocal fremitus and
resonance.
Singns of underlying disease.

Investigatios:
Chest radiology and
ultrasonography
Diagnostic aspiration
Additional tests.

Effusions of more than 175 mL are usually
apparent as blunting of the costophrenic angle
on upright posteroanterior chest radiographs. On
supine chest radiographs, which are commonly
used in the intensive care setting, moderate to
large pleural effusions may appear as a
homogenous increase in density spread over the
lower lung fields. Apparent elevation of the
hemidiaphragm, lateral displacement of the
dome of the diaphragm, or increased distance
between the apparent left hemidiaphragm and
the gastric air bubble suggests subpulmonic
effusions.

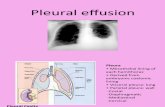
Anteroposterior, upright chest radiograph
shows bilateral pleural effusions and loss of
bilateral costophrenic angles (meniscus sign).

Posteroanterior, upright chest radiograph
shows isolated, left-sided pleural effusion
and loss of left, lateral costophrenic angle.

Lateral decubitus films more reliably detect
smaller pleural effusions. Layering of an
effusion on lateral decubitus films defines a
freely flowing effusion and, if the layering
fluid is 1 cm thick, indicates an effusion of
greater than 200 mL that is amenable to
thoracentesis. Failure of an effusion to layer
on lateral decubitus films indicates the
presence of loculated pleural fluid or some
other etiology causing the increased pleural
density.


Chest CT scanning with contrast should be performed in all patients with an undiagnosed pleural effusion, if it has not previously been performed, to detect thickened pleura or signs of invasion of underlying or adjacent structures. The two diagnostic imperatives in this situation are pulmonary embolism and tuberculouspleuritis. In both cases, the pleural effusion is a harbinger of potential future morbidity. In contrast, a short delay in diagnosing metastatic malignancy to the pleural space has less impact on future clinical outcomes. CT angiography should be ordered if pulmonary embolism is strongly suggested

A diagnostic thoracentesis should be performed
under ultrasound guide if the etiology of the
effusion is unclear or if the presumed cause of
the effusion does not respond to therapy as
expected. Relative Relative Contraindications:
small volume of fluid (< 1 cm thickness on a
lateral decubitus film)
bleeding diathesis or systemic anticoagulation
mechanical ventilation
cutaneous disease over the proposed puncture
site.

Reversal of coagulopathy or thrombocytopenia may not be necessary as long as the procedure is performed under ultrasound guidance by an experienced operator.Mechanical ventilation with positive end-expiratory pressure does not increase the risk of pneumothorax after thoracentesis, but it increases the likelihood of severe complications (tension pneumothorax or persistent bronchopleuralfistula) if the lung is punctured. An uncooperative patient is an absolute contraindication for this procedure.

pain at the puncture site
cutaneous or internal bleeding from laceration of an intercostal artery or spleen/liver puncture
pneumothorax, empyema, reexpansionpulmonary edema, malignant seeding of the thoracentesis tract, and adverse reactions to anesthetics used in the procedure.
Pneumothorax complicates approximately 6% of thoracenteses but requires treatment with a chest tube drainage of the pleural space in less than 2% of cases.
In addition, significant chronic obstructive or fibrotic lung disease increases the risk of a symptomatic pneumothorax complicating the thoracentesis

When a pleural fluid
sample is taken it
should be analysed as
follows:

the tests first proposed by Light have
become the criterion standards:
The fluid is considered an exudate if any of
the following are found:
Ratio of pleural fluid to serum protein
greater than 0.5
Ratio of pleural fluid to serum LDH greater
than 0.6
Pleural fluid LDH greater than two thirds of
the upper limits of normal serum value

The fluid is considered a transudate if all of the above are absent
alternative criteria:
Pleural fluid LDH value greater than 0.45 of the upper limit of normal serum values
Pleural fluid cholesterol level greater than 45 mg/dL
Pleural fluid protein level greater than 2.9 g/dL
Pleural fluid cosidered exudate if protein content is more than 35g/l and transudateif less than 25g/l.

The criteria from Light and these alternative
criteria identify nearly all exudates
correctly, but they misclassify approximately
20-25% of transudates as exudates, usually in
patients on long-term diuretic therapy for
congestive heart failure (because of the
concentrating effect of diuresis on protein
and LDH levels within the pleural space).

Using the criterion of serum minus pleural
protein concentration level of less than 3.1
g/dL, rather than a serum/pleural fluid ratio
of greater than 0.5, more correctly identifies
exudates in these patients.
A gradient of serum albumin to pleural fluid
albumin of less than 1.2 g/dL also identifies
an exudate in such patients.

Pleural fluid LDH levels greater than 1000
IU/L suggest empyema, malignant effusion,
rheumatoid effusion, or pleural
paragonimiasis.
Pleural fluid LDH levels are also increased in
effusions from Pneumocystis jiroveci
(formerly, P carinii) pneumonia. The
diagnosis is suggested by a pleural
fluid/serum LDH ratio of greater than 1, with
a pleural fluid/serum protein ratio of less
than 0.5. ???????????

Pleural fluid glucose and pH A low pleural glucose concentration (30-50 mg/dL) suggests malignant effusion, tuberculous pleuritis, esophageal rupture, or lupus pleuritis.
A very low pleural glucose concentration (ie, < 30 mg/dL) further restricts diagnostic possibilities, to rheumatoid pleurisy or empyema.
Pleural fluid pH is highly correlated with pleural fluid glucose levels. A pleural fluid pH of less than 7.30 with a normal arterial blood pH level is caused by the same diagnoses as listed above for low pleural fluid glucose. However, for parapneumonic effusions, a low pleural fluid pH level is more predictive of complicated effusions (that require drainage) than is a low pleural fluid glucose level. In such cases, a pleural fluid pH of less than 7.1-7.2 indicates the need for urgent drainage of the effusion, while a pleural fluid pH of more than 7.3 suggests that the effusion may be managed with systemic antibiotics alone.

Indications of thoracostomy in parapneumonic effusion:
a low pleural fluid glucose level (<40 mg/dL), a low. or
pleural fluid pH (<7.2) or
a positive Gram stain or
Positive culture of the pleural fluid are more likely to require tube thoracostomy.

In malignant effusions, a pleural fluid pH of
less than 7.3 has been associated in some
reports with more extensive pleural
involvement, higher yield on cytology,
decreased success of pleurodesis, and
shorter survival times.
Handle pleural fluid samples as carefully as
arterial samples for pH measurements, with
fluid collected in heparinized syringes and
ideally transported on ice for measurement
within six hours.

Pleural fluid lymphocytosis, with lymphocyte values greater than 85% of the total nucleated cells, suggests TB, lymphoma, sarcoidosis, chronic rheumatoid pleurisy.
Pleural lymphocyte values of 50-70% of the nucleated cells suggest malignancy
Pleural fluid eosinophilia with values greater than 10% of nucleated cells, is seen in approximately 10% of pleural effusions and is not correlated with peripheral blood eosinophilia.and is most often caused by air or blood in the pleural space.

Blood in the pleural space causing
eosinophilia may be the result of pulmonary
embolism with infarction or benign asbestos
pleural effusion or may be associated with
other nonmalignant diseases, including
parasitic disease (especially paragonimiasis),
fungal infection (coccidioidomycosis,
cryptococcosis, histoplasmosis), and a variety
of medications.
High neutrophil count in pleural fluid
suggests parapneumonic effusion.

Cytology findings(abnormal mesothelial cell)
are positive in 58% of effusions related to
mesothelioma.
The sensitivity of cytology is not highly
related to the volume of pleural fluid tested.
Sending more than 50-60 mL of pleural fluid
for cytology does not increase the yield of
analysis.

Additional specialized tests are warranted
when specific etiologies are suspected.
Pleural biopsy.
Measure pleural fluid amylase levels if a
pancreatic origin or ruptured esophagus is
suspected or if a unilateral, left-sided
pleural effusion remains undiagnosed after
initial testing. note, increased pleural fluid
amylase can also be seen with malignancy.
Measure triglyceride and cholesterol levels in
milky pleural fluids when chylothorax or
pseudochylothorax is suspected.

Tumor markers, such as carcinoembryonic
antigen, are suggestive of malignant
effusions (especially adenocarcinoma).
Consider immunologic studies, including
pleural fluid antinuclear antibody(SLE) and
rheumatoid factor (RA), when collagen-
vascular diseases are suspected.

Transudative effusions are managed by treating the underlying medical disorder. However, regardless of whether transudative or exudative, large, refractory pleural effusions causing severe respiratory symptoms can be drained to provide symptomatic relief.
The management of exudative effusions depends on the underlying etiology of the effusion. Pneumonia, malignancy, and TB cause most exudative pleural effusions. Complicated parapneumonic effusions and empyemas should be drained to prevent development of fibrosingpleuritis. Malignant effusions are usually drained to palliate symptoms and may require pleurodesis to prevent recurrence.

Surgery :
pesistanat collections
+pleural thickning on u/s
may require surgical
intervention.

THANK YOU