Lesson 15 Part II: Ionic Bonding & Intro to Lewis Structures › ... ›...
Transcript of Lesson 15 Part II: Ionic Bonding & Intro to Lewis Structures › ... ›...

Lesson 15 Part II:
Ionic Bonding &
Intro to Lewis Structures

Do Now: Follow all instructions to earn 5 points for participation grade.
• Take your CJs out – write info down from board.
• Keep CJs out and open on desk.
• Take out your HW – boxes 4 and 5 in Lesson 15 Part II notes.
• Answer the following question(s) in a box on your ‘do now’ sheet:
1. Brainstorm a list of things you know about polyatomic ions.
2. What is this separation technique shown below? What kind of mixtures are
separated with this technique? How?
Date: 1-10-18
7B, 7Pb

Check your HW – Boxes 4 & 5 in Lesson 15 Part II notes
2(+1) + 1(-2) = 0
1(+2) + 1(-2) = 0
3(+2) + 2(-3) = 0
1(+2) + 2(-1) = 0
Gold (I) sulfide
Mercury (II) sulfate
Manganese (II) phosphate
Cobalt (II) bromite
Ammonium fluoride

CaCO3
Mg(NO3)2
(NH4)2SO4
Na2SO4
Fe(OH)3
Cr3(PO4)2
NaClO
NaClO4

Review – halogen polyatomic ions
ClO-
hypochloriteClO2
-
chlorite
ClO3-
chlorate*most common
ClO4-
perchlorate
One MORE
oxygen
One LESS
oxygenOne LESS
oxygen
base name
for chlorine

Review – halogen polyatomic ions
F Cl Br I
One oxygen
“Hypo___ite”
Two oxygens
“___ite”
Three oxygens
“___ate”
Four oxygens
“per___ate”
ClO-
hypochlorite
ClO2-
chlorite
ClO3-
chlorate
ClO4-
perchlorate
FO-
hypofluorite
FO2-
fluorite
FO3-
fluorate
FO4-
perfluorate
BrO-
hypobromite
BrO2-
bromite
BrO3-
bromate
BrO4-
perbromate
IO-
hypoiodite
IO2-
iodite
IO3-
iodate
IO4-
periodate
All have an
overall charge
of 1-

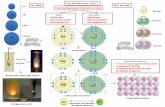
Review “Practice with Orbital Diagrams”:
Turn to p.5 in Lesson 15 Part II notes

Review “Practice with Orbital Diagrams”:

Review “Practice with Orbital Diagrams”:
Be will lose these
valence electrons
How many fluorine atoms
will accept Be’s valence
electrons?
F has one spot to fill

2s now empty.
1s2 is considered a full outer shell.
Two fluorine atoms each accepted one
electron. 2s22p6 is a full outer shell.
Review “Practice with Orbital Diagrams”: BeF2

Review “Practice with Orbital Diagrams”:

Review “Practice with Orbital Diagrams”:
Li will lose this
valence electron
O has two spots to fill

Review “Practice with Orbital Diagrams”:
2s now empty.
1s2 is considered a full outer shell.
One oxygen accepted two electrons, one
from each Li atom.
2s22p6 is a full outer shell.
Li2O

Intro to Lewis Dot Symbols:
Guidelines:
- dots are used to represent electrons
- only valence electrons are included
- cations no dots & and positive charge
- anions will have a filled valence shell (8) & a negative charge

STEPS:
1. Find the number of valence electrons (A group elements = group number)
2. Write the element symbol.
3. Draw dots starting at the top of the symbol and continue clockwise.
Intro to Lewis Dot Symbols:

EXAMPLES
1.Tin
Group # ____ How many valence electrons? ______4A 4
Sn
Now move
clockwise

EXAMPLES
2. Sulfur
Group # ____ How many valence electrons? ______6A 6
S

Cu Br B
Ne H Se
PRACTICE:

Cu Br BTransition metals have 1
or 2 valence electrons
PRACTICE:

Cu Br B
Ne H Se
PRACTICE:

Lewis Structures are also used to show ions and ionic compound
formation.

Lewis Structures are also used to show ions and ionic compound
formation.

Lewis Structures are also used to show ions and ionic compound
formation.

Lewis Structures are also used to show ions and ionic compound
formation.

Ffluorine
Ffluoride
-gains an
electron
#1
8 valence electrons =
full outer shell

Nasodium
Nasodium ion
+loses an
electron
#2
Cation has a full outer shell – we
don’t draw the electrons for cations

Ooxygen
Ooxide
2-gains two
electrons
#3
8 valence electrons =
full outer shell

PRACTICE:
Selenide Beryllium ion Barium ion
Potassium ion Nitride Phosphide

PRACTICE:
Selenide
Se2-
Beryllium ion
Be2+
Barium ion
Ba2+

PRACTICE:
Selenide
Se2-
Beryllium ion
Be2+
Barium ion
Ba2+
Potassium ion
K
Nitride
N
Phosphide
P+ 3- 3-

EXAMPLE: Draw how an ionic compound would from between Li and O to
form Li2O
Each Li atom will
donate their one
valence electron.
Li
Li

EXAMPLE: Draw how an ionic compound would from between Li and O to
form Li2O
Oxygen needs two
electrons to have
a full outer shell
Each Li atom will
donate their one
valence electron.
Li
Li
O

EXAMPLE: Draw how an ionic compound would from between Li and O to
form Li2O
Oxygen needs two
electrons to have
a full outer shell
Each Li atom will
donate their one
valence electron.
Li
Li
O1+
1+
2-
Li1+
Li1+
O....
.... 2-

#2 PRACTICE:
Draw how an ionic compound would from between Ca and F to form CaF2
Ca F2+
-
Ca2+
F....
.... -
F-
F.... .... -
Alkaline earth
ion
Fluoride ion

#3 PRACTICE:
Draw how an ionic compound would from between Na and Cl to form NaCl
Na Cl1+
-
Na1+
Cl....
....
-

Homework: Parts I, II, III under “Review & Practice” in Lesson 15 Part 2.

Element # of electron
shells
Group
number
# of valence
electrons
Predict the ion it
will form:
Metal, nonmetal,
metalloid?
Na 3 1A 1 Na+ metal
K
Sr
B
P
Cl
Kr