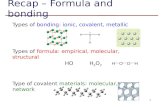
Types of Bonding. There are two types of bonds: Ionic bonds Covalent bonds.
BONDING TYPES: IONIC BONDING
description
Transcript of BONDING TYPES: IONIC BONDING

BONDING TYPES:IONIC BONDING
What is an ionic bond?How do I name ionic compounds?
How do I write the chemical formula for ionic compounds?

IONIC BONDING• Ionic Bonding: I, selfish, stealing, transfer of e-• attraction that bonds charged ions together• force behind opposites attract
++++++CATION
- - - - - -ANION

NAMING BINARY COMPOUNDS:• Binary Compounds two elements joined
together, can be ionic or covalent• IONIC = name of cation + name of anion

Cation + Anion = Compound

Bond with a Classmate!
Is this right?
Cation + AnionMg +2 S -2
Mg 2 S 2
REDUCE!
MgS
Magnesium Sulfide

REVIEW POLYATOMICS
• http://www.chemfiles.com/flash/polyatomic_ions.html• http://www.chemfiles.com/flash/polyions.html• http://www.chemfiles.com/flash/polyions1.html• http://www2.pvc.maricopa.edu/tutor/chem/chem130/no
menclature/polyatomicion.html
• REFERENCE TABLE LIST on Page 7

NAMING COMPOUNDS WITH POLYATOMIC IONS

Naming Ionic Compounds with Transition Metals
• Roman Numeral Naming System – we will use this methodex. Manganese (II) Oxide MnO
• Latin Naming System exists – we will not use this methodex. Stannous Oxide SnO

Transition Metal Charges

Naming Ionics with Transition Metals

Writing Chemical Formulas• Assign Charges then criss x cross the numbers• DO NOT criss x cross the charges, just the #s
Ex. Magnesium ChlorideAssign Charges Mg +2 Cl-1
Criss-Cross Mg Cl2
Chemical Formula MgCl2