ID of Diabetic Foot - ce.mayo.edu Diabetic Foot... · ID of Diabetic Foot . ... Diagnosing...
-
Upload
nguyencong -
Category
Documents
-
view
221 -
download
4
Transcript of ID of Diabetic Foot - ce.mayo.edu Diabetic Foot... · ID of Diabetic Foot . ... Diagnosing...

Division of INFECTIOUS DISEASES
Abinash Virk, MD, DTM&H, FIDSA
Associate Professor of Medicine
Member, Ortho ID Focus Group
Mayo Clinic College of Medicine
Mayo Wound Symposium 2017
February 23rd- 25th, 2017
ID of Diabetic Foot

DISCLOSURES
• Relevant Financial Relationship(s) • Travel and Wellness LLC
• Off Label Usage • None
©2011 MFMER | slide-2

OBJECTIVES
• Identify the best methods to make the diagnosis of diabetic foot infection (DFI)
• Describe the classification of DFI
• Understand the microbiology of DFI
• Explain the principles of DFI management

Age-adjusted Prevalence of Obesity and Diagnosed Diabetes
Among US Adults
Obesity (BMI ≥30 kg/m2)
Diabetes
1994
1994
2000
2000
No Data <14.0% 14.0%–17.9% 18.0%–21.9% 22.0%–25.9% > 26.0%
No Data <4.5% 4.5%–5.9% 6.0%–7.4% 7.5%–8.9% >9.0%
CDC’s Division of Diabetes Translation. United States Surveillance System available at
http://www.cdc.gov/diabetes/data
2014
2014
9.3% of US population or 29.1 million people have diabetes

Diabetes complications
• 15% lifetime risk of developing foot ulcers
• 66% of open ulcers result in infection
• Osteomyelitis prevalence in diabetic foot ulcers ~ 10-20%
• In 2010, ~73,000 non-traumatic lower-limb amputations (adults)
©2011 MFMER | slide-5 http://www.cdc.gov/diabetes
Lower
extremity
condition

Pathogenesis of diabetic foot infection
©2011 MFMER | slide-6
Neuropathy
Sensory Neuropathy
Decreased pain sensation
Clawing of toes
Decreased proprioception
Autonomic Neuropathy
Reduced sweating
Fissures / callus formation
Altered blood flow
Motor Neuropathy
Abnormal foot mechanics
Increased foot pressure
Vascular Insufficiency
ULCER
GANGRENE
Infection

Diabetic foot infection and osteomyelitis
©2011 MFMER | slide-7
Infection Bacterial virulence
Host immune response Reduced neutrophilic/macrophage response
Bacterial burden
Host glycemic control
Colonization

When should infection be suspected in patients with diabetic foot ulcers?
• Clinical signs of infection • Local:
– Usual signs: Erythema, swelling, purulent secretions
– Atypical signs: non-purulent secretions, friable or discolored granulation tissue, undermining of the wound edges, or foul odor
• Systemic signs of infection
• Presence of risk factors
• Classify diabetic foot ulcers / infection • Clinical classification
©2011 MFMER | slide-8

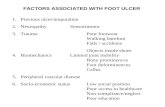
Risk factors for DFI
• Previous lower extremity amputation [OR 19.9]
• Probe-to-bone is positive [OR 6.7]
• Ulceration present for >30 days [OR 4.7]
• Peripheral neuropathy [OR 3.4]
• History of recurrent foot ulcers [OR 2.4]
• Traumatic foot wound [OR 2.4]
• Peripheral vascular disease [OR 1.9]
• Renal insufficiency
• Barefoot walking
©2011 MFMER | slide-9
Lipsky, B et al. CID 2012;54(12):132–173
Lavery LA eta al. Diabetes Care 29:1288–1293, 2006

Classification of DFI is helpful in management
• Why classify diabetic foot ulcers?
• Assist in
– Antimicrobial choice
– Urgency of evaluation and management
– Assessing need for involvement of other specialties
– Prognosis and predict outcomes
• Many classification systems in literature
• Most user friendly and validated • Infectious Diseases Society of America (2012)
• International Working Group on the Diabetic Foot (IWGDF) developed PEDIS (Perfusion, Extent/size, Depth/tissue loss, Infection, Sensation)
©2011 MFMER | slide-10
Lipsky, B et al. Clin Infect Dis 2012;54(12):132–173

IDSA and PEDIS DFI Classification Clinical Manifestation of Infection PEDIS
Grade
IDSA Infection
Severity
No symptoms or signs of infection
Infection defined by the presence of at least 2 of following:
• Local swelling or induration
• Erythema
• Local tenderness or pain
• Local warmth
• Purulent discharge (thick, opaque to white or sanguineous secretion
1 Uninfected
Local infection involving only the skin and the subcutaneous tissue
(without involvement of deeper tissues & without systemic signs)
• If erythema, must be >0.5 cm to ≤2 cm around the ulcer
• Exclude other causes of an inflammatory response of the skin
(eg, trauma, gout, acute Charcot neuro-osteoarthropathy,
fracture, thrombosis, venous stasis)
2 Mild
Local infection with erythema > 2 cm, or involving
structures deeper than skin and subcutaneous tissues
(eg, abscess, osteomyelitis, septic arthritis, fasciitis),
and
no systemic inflammatory response signs
3 Moderate
Local infection with the signs of SIRS
• SIRS manifested by ≥2 of the following: Temperature
>38°C or <36°C, HR >90 beats/min, RR >20
breaths/min or PaCO2 <32 mm Hg, WBC >12 000 or
<4000 cells/μL or ≥10% immature (band) forms
4 Severe
©2011 MFMER | slide-11 Lipsky, B et al. CID 2012;54(12):132–173

Hospitalization and amputation based on the Infectious Diseases Society of America and the
International Working Group on the Diabetic Foot infection severity classification.
Lawrence A. Lavery et al. Clin Infect Dis. 2007;44:562-565
© 2007 by the Infectious Diseases Society of America

PEDIS stage 2 or mild infection
©2011 MFMER | slide-13

PEDIS stage 3 or moderate infection
• Deeper structure involvement • Tendons
• Fascia
• Joints
• Bone
©2011 MFMER | slide-14

How do I assess a patient with DFI?
• Assess the person as a whole for • Signs of sepsis
• Metabolic status
• Cognitive and social function status
• …then the foot/limb – for biomechanics, neurological, vascular status
• Charcot foot neuroarthropathy
• Claw foot
• Calluses etc
• …then the ulcer – for infection
©2011 MFMER | slide-15

Initial assessment History
1. Chronic ( > 4 weeks) foot wound
2. Previous infection at same or nearby site
3. New pain in the wound (especially in a previously insensate foot)
4. Presence of immunosuppressive condition (beyond that related to diabetes)
©2011 MFMER | slide-16
Physical examination
1. Large wound ( > 2 cm2)
2. Deep wound ( > 3 mm)
3. Classic signs of inflammation (tenderness, pain, redness, warmth, induration)
4. Secondary signs of infection (foul odor, friable or discolored granulation tissue, undermining, purulent or non-purulent secretions)
5. Probe-to-bone or not

Key initial questions
• Need for hospitalization? • All severe
• Some moderate
• Social or cognitive limitations
• Need for urgent or emergent surgery? • Necrotizing fasciitis
• Gangrene
• Critical ischemia
• What grade DFI is it?
©2011 MFMER | slide-17

Diagnosing infection in the diabetic foot
• Clinical symptoms and signs
• Laboratory testing • CBC, Sed rate and CRP – not very helpful
– Leukocytosis present in 50% of patients with osteo
– Normal CBC does not preclude osteomyelitis diagnosis
• Procalcitonin
– Jury is still out.
– May help in diagnosing DFI or diabetic foot osteomyelitis
• Not helpful:
– Interleukin-6 (IL-6), interleukin-8 (IL-8), tumor necrosis factor alpha (TNFα), monocyte chemotactic protein-1 (MCP-1) and macrophage inflammatory protein-1 alpha (MIP1α)
©2011 MFMER | slide-18
Massara, M., et al. Int Wound J. 2015 Dec 3. doi: 10.1111/iwj.12536
Van Asten SA et al. Int Wound J. 2015 Dec 3. doi: 10.1111/iwj.12545
Jafari NJ et al. Int J Endocrinol Metab. 2014 January; 12(1): e13376

When to suspect osteomyelitis?
©2011 MFMER | slide-19
• Chronic ulcer > 4 weeks
• Deep ulcer > 3 mm
• Intermittently healing and draining wound
• Bone visible or probe-to-bone positive
• Sausage toe
• Osteomyelitis present • 10-20% of moderate infections
• 50-60% of severe infections

Diagnosis of Osteomyelitis in DFI
• Plain radiographs
• Radionuclide bone scans • Technetium-99 bone scan: more sensitive than X-rays, but non-specific
• Radionuclide white blood cell scans • Slightly less sensitive than bone scan, specificity is typically higher
• Generally done when MRI unavailable
• Magnetic resonance imaging (MRI) • Most useful imaging study for diagnosing DFO and for evaluation of
extent of both bone and soft-tissue involvement and surgery planning
• Positron emission tomography (PET) • May be helpful but not that well studied and more expensive
• Bone biopsy • For culture and histological examination of bone to both confirm the
diagnosis and potentially identify the pathogen(s)
©2011 MFMER | slide-20

60
65
70
75
80
85
ESR > 67 CRP > 14 mg/l Procalcitonin >0.3 ng/ml
Sensitivity
Specificity
©2011 MFMER | slide-21
Michail M, et al. Int J Low Extrem Wounds. 2013;12:94-99
Diagnosing osteomyelitis in the diabetic foot Using ESR, CRP and Procalcitonin
ESR (mm/hr) CRP mg/l PCT (ng/ml)
No osteomyelitis (n 34) 66 8.7 0.71
Osteomyelitis (n 27) 76 25 2.4
Gold standard
comparator:
probe to bone +
imaging

70
72
74
76
78
80
82
84
86
88
ESR > 70 Ulcer > 2.0 cm2 ESR + Ulcer
83
88
79
77 77
82
Sensitivity
Specificity
©2011 MFMER | slide-22
Ertugrul BM, et al. Med Sci Monit. 2009;15:CR307-CR312
Diagnosing osteomyelitis in the diabetic foot Using ulcer size, ESR or both
Gold standard
comparator:
MRI imaging
or histopath

0
10
20
30
40
50
60
70
80
90
100
Plain x-ray MRI Bone scan Probe tobone
Sensitivity
Specificity
©2011 MFMER | slide-23
Dinh MT et al. Clin Infect Dis . 2008;47:519-527
Diagnosing osteomyelitis in the diabetic foot Meta-analysis - 9 cohort trials Using x-ray, MRI, Bone scan and probe to bone
0
1
2
3
4
5
6
7
Plain x-ray MRI Bone scan Probe tobone
LR+
LR-
4 trials
n = 177
4 trials
n = 135
6 trials
n = 185
2 trials
n = 288
Gold standard
comparator:
histopath or
bone cultures

0
2
4
6
8
10
12
Ulcer > 2.0 cm Probe to bone Sed Rate > 70 Plain X-ray
LR+
LR-
©2011 MFMER | slide-24
Butalia S. et al. JAMA. 2008;299:806-813
Diagnosing osteomyelitis in the diabetic foot Meta-analysis - 21 cohort trials Using ulcer size, x-ray, ESR and probe to bone
1 trials
n = 35
3 trials
n = 75 4 trials
n = 108
16 trials
n = 567
Gold standard
comparator:
bone biopsy

International Working Group on the Diabetic Foot Osteomyelitis Diagnostic Criteria
©2011 MFMER | slide-25
Category Post-test
probability of
osteomyelitis
Management
advice
Criteria Comments
Definite (‘beyond
reasonable doubt’)
>90% Treat for
osteomyelitis
Bone sample with positive culture AND positive histology
OR Purulence in bone found at surgery
OR Atraumatically detached bone fragment removed from ulcer
by podiatrist/surgeon OR Intraosseous abscess found on MRI
OR Any two probable criteria OR one probable and two
possible criteria OR, any four possible criteria below
Probable (‘more
likely than not’);
51–90% Consider treating,
but further
investigation may
be needed
Visible cancellous bone in ulcer OR MRI showing bone edema
with other signs of osteomyelitis OR Bone sample with positive
culture but negative or absent histology OR Bone sample with
positive histology but negative or absent culture OR Any two
possible criteria below
Possible (but on
balance, less
rather than more
likely)
10–50% Treatment may
be justifiable, but
further
investigation
usually advised
Plain X-rays show cortical destruction OR MRI shows bone
edema OR cloaca, OR Probe to bone positive OR, Visible
cortical bone OR ESR >70mm/h with no other plausible
explanation OR Non-healing wound despite adequate
offloading and perfusion for >6 weeks OR ulcer of >2 weeks
duration with clinical evidence of infection
Unlikely <10% Usually no need
for further
investigation or
treatment
No signs or symptoms of inflammation AND normal X-rays AND
ulcer present for <2 weeks or absent AND any ulcer present is
superficial OR Normal MRI OR Normal bone scan
Berendt AR et al. Diabetes Metab Res Rev 2008; 24(Suppl 1): S145–S161.

©2011 MFMER | slide-26
Anurag Markanday. Open Forum Infect Dis (2014) 1 (2):doi: 10.1093/ofid/ofu060
Initial Clinical assessment Score
Visible cancellous bone in ulcer 2
Positive PTB test or visible cortical bone in ulcer 1
ESR >70 with no other plausible explanation 1
Cortical destruction on initial plain radiograph 1
Ulcer size more than 2 square cm 1
Clinical gestalt: nonhealing wound for >6 weeks despite perfusion or ulcer >2 weeks
duration with evidence of Infection
1
≥ 4
Radiology Scores: (add if initial score less than 4) Score
Positive MRI scan: +2
Interval change (minimum 2 weeks) on plain radiograph: +1
Positive leukocyte scan: +1
Negative MRI scan: −2
Negative bone scan: −2
< 4
< 4
Low probability of osteomyelitis. Treat as SSTI. Reassess.
≥ 4 Osteomyelitis
High
probability
2-Step Score-Based DF Osteomyelitis Diagnostic Pathway

©2011 MFMER | slide-27
PEDIS stage 3 or moderate infection
right fifth ray amputation

©2011 MFMER | slide-28
MSSA bloodstream infection

©2011 MFMER | slide-29

©2011 MFMER | slide-30

Obtaining samples for Microbiology
• All moderate or higher grade infections
• Mild if concerns for drug resistance
• Failing to respond to empiric treatment
Methodology of culture sampling: critical
©2011 MFMER | slide-31
Deeper tissue or bone cultures should be obtained
Lipsky, B et al. Clin Infect Dis 2012;54(12):132–173

How do the cultures help?
• Aim for continued clinical response with the narrowest spectrum antimicrobial covering organisms isolated
• If clinically responding • Select the narrowest spectrum based on cultures
• Not all organisms always need to be covered e.g. MRSA isolated but patient clinically responding to cephalexin
• If not responding • Broaden treatment to cover organisms not being covered
based on cultures
• Consider missed diagnosis – un-drained abscess, gout, underlying osteomyelitis, etc
• Consider need for surgical intervention
©2011 MFMER | slide-32

Microbiology of DFI
Staph aureus
MRSA (10-30%)
Enterococcus spp
Gram-negatives
ESBL GNB
Pseudomonas (<10%)
Anaerobes
Osteomyelitis
S. aureus most common
Infection with prior antibiotic use
Staph aureus Beta-hemolytic strep
such as S. agalactiae (GBS), S. pyogenes
Gram-negatives
In chronic or severe wounds
Anaerobes (~40%)
Infection
Polymicrobial colonization
Mostly aerobic gram-positive cocci such as
S. aureus, S. agalactiae (GBS), S.
pyogenes, and coagulase-negative
staphylococci
Ulcer
©2011 MFMER | slide-33
Use culture data to refine antimicrobial treatment

Antimicrobial selection
• Mild • Start narrow and broaden if no response
• Oral preferred, topical may help in very mild
• Aimed at aerobic Gram-positive organisms mainly
• Moderate • Start narrow and broaden if no response
• Oral preferred but on occasion IV may be needed
• Aimed at aerobic Gram-positive organisms and anaerobes
• Hold antibiotics if underlying osteomyelitis but no cellulitis or systemic toxicity
• Severe • Start broad and then narrow
• Parenteral treatment
©2011 MFMER | slide-34

MRSA antimicrobial coverage if
• Patient known to have MRSA infection or colonization within the past year
• Local MRSA prevalence is high • For mild infections: cover MRSA if local prevalence 50%
• For moderate infections: cover MRSA if local prevalence 30%
• Severe infection
©2011 MFMER | slide-35

Complexity of antimicrobial choice
Patient factors
Allergies
Renal function
GI absorption
Immune status
Infection
Severity
Recent Abx
Osteomyelitis
Vascular status
Antibiotics
Safety
Spectrum
Efficacy
Cost
Microbiology
Organisms present
Resistance
©2011 MFMER | slide-36

Specific antimicrobial choices Mild (usually treated with oral agent[s])
Probable
Pathogen(s)
Antibiotic Agent Comments
Staphylococcus
aureus(MSSA);Str
eptococcus spp
Dicloxacillin Requires QID dosing; narrow spectrum;
inexpensive
Amoxicillin-clavulanate Relatively broad-spectrum oral agent that
includes anaerobic coverage
Clindamycin Check for inducible resistance in MRSA by
confirming negative “D-test” in Microbiology
susceptibilities testing
Cephalexin Requires QID dosing; inexpensive
Levofloxacin Once-daily dosing; suboptimal against S. aureus
Methicillin-
resistant S.
aureus (MRSA)
Doxycycline Active against many MRSA & some gram-
negatives; not good against streptococcus
species
Trimethoprim/sulfametho
xazole
Active against many MRSA & some gram-
negatives; not good against streptococcus
species
©2011 MFMER | slide-37
Lipsky, B et al. Clin Infect Dis 2012;54(12):132–173

Specific antimicrobial choices Moderate to severe infections
Probable
Pathogen(s)
Antibiotic Agent Comments
MSSA;
Streptococcus
spp;
Enterobacteriac
eae;
obligate
anaerobes
Ceftriaxone Once-daily dosing, third-generation
cephalosporin but not active against P.
aeruginosa
Ampicillin-sulbactam Adequate for mild to moderate infections
Levofloxacin Once-daily dosing; suboptimal against S. aureus
Levofloxacin or
ciprofloxacin with
clindamycin or
metronidazole
Limited evidence supporting clindamycin for
severe S. aureus infections; PO & IV formulations
for both drugs
Moxifloxacin Once-daily oral dosing. Relatively broad-
spectrum, including most obligate anaerobic
organisms. Expensive.
Ertapenem Once-daily dosing. Expensive. Relatively broad
spectrum including anaerobes, not active P.
aeruginosa
Meropenem Very broad-spectrum (but not against MRSA);
use only when this is required. Consider when
ESBL producing pathogens suspected ©2011 MFMER | slide-38
Lipsky, B et al. Clin Infect Dis 2012;54(12):132–173

Specific antimicrobial choices Moderate to severe infections
Probable
Pathogen(s)
Antibiotic Agent Comments
MRSA
Vancomycin Nephrotoxicity risk
Ceftaroline BID dosing. Expensive.
Daptomycin Once-daily. Expensive. Requires CPK monitoring
Linezolid Expensive; increased toxicities when used >2 wk
Dalbavancin Expensive, 2 doses. Not studied for osteomyelitis
Oritavancin Expensive, 1 dose. Not studied for osteomyelitis
Pseudomonas
aeruginosa
Cefepime Adequate for Pseudomonas aeruginosa and other
common pathogens. P. aeruginosa is an uncommon
pathogen in diabetic foot infections except in special
circumstances
Piperacillin-tazobactam QID dosing. Useful for broad spectrum coverage.
Mixed infections:
MRSA,
Enterobacteriacae,
Pseudomonas, and
obligate anaerobes
Vancomycinc plus one of
the following: ceftazidime,
cefepime,
piperacillin/tazobactam,
aztreonam or a
carbapenem
Very broad-spectrum coverage; usually only used for
empiric therapy of severe infection.
Consider addition of anaerobic coverage with
metronidazole if not using piperacillin/tazobactam or
carbapenem.
©2011 MFMER | slide-39 Lipsky, B et al. Clin Infect Dis 2012;54(12):132–173

Multidisciplinary Diabetic Foot Care Team
Internal Medicine
Vascular
Medicine
Surgeons -
Orthopedic
Vascular
Podiatry
Infectious
Diseases Wound Care
Physical Medcine & Rehabilitation
Endocrinology
©2011 MFMER | slide-40
And more…. Nurses, social workers, pharmacists….

Which patients should undergo surgery?
• All severe infections posing risk to limb or life
• Necrotizing fasciitis • Suspected in severe deep pain in a previously insensate foot
• Necrosis, abscesses, gas gangrene
• Osteomyelitis
• Compartment syndrome
• Ischemia
©2011 MFMER | slide-41

Duration of antibiotics for DFI and osteomyelitis
©2011 MFMER | slide-42
Soft tissue infection only Mild 7 - 14 days
Moderate 14 – 28 days
Severe 14 – 28 days
Bone/joint infection Duration depends on extent of surgery and residual infection
No residual infected tissue 2 – 5 days
Residual infected soft tissue 14 – 28 days
Residual infected (debrided) bone 28 – 42 days
No surgery or residual dead bone ? months
Vascular status may
impact duration Lipsky, B et al. Clin Infect Dis 2012;54(12):132–173

Division of Infectious Diseases
Mayo Clinic, Rochester, MN
Thank you! Questions?