Examples of different random coil conformations Conformation = stereostructure of the molecule...
-
Upload
suzanna-fields -
Category
Documents
-
view
225 -
download
8
Transcript of Examples of different random coil conformations Conformation = stereostructure of the molecule...


Examples of different random coil conformations
Conformation = stereostructure of the molecule defined by its sequence of bonds and torsion angles
C
H
CC
CC
CC
CC
H
H
H
H
H
H
H
H
HH H H H
HHHH
Conformation of polymers

22
6
1rRg
r
Rg
1) Average of end-to-end distance (mean square end-to-end distance) 2r
Note: if the Contour lenght of the chain is L maximum of rmax = L.
denotes the average calculated from a large number of conformations
2) Radius of gyration Rg Root mean square distance of the collection of atoms from their common centre of gravity
Gaussian coil
n
ii
n
iii
g
m
rmR
1
1
2
2
ir is the vector from the centre of the gravity to atom i
Mean square end-to-end distance and Radius of gyration Rg
are related
Polymer coil size

The essential message …a) Polymer chains tend to retract to coils (unless there exist opposing factors..) and the chain conformation can be characterized as a random coil (solution, polymer melt and also glassy amorphous state)
The polymer chain size can be evaluated based on average end-to-end distance r and radius of gyration Rg .
r
Rg
b) Individual polymer chains can be studied as dissolved in solvents. Unperturbed state is characterized by the absence of long-range interactions ( -q solvent)
c) Models for polymer chain dimensions are developed. Mean square of end-to-end distance scales like
22 Cnlr
C = 1 freely jointed chainC 2 freely rotating chainC 3.4 hindered rotationC 6.7 Advanced models (statistical weight matrices) Experimental value for polyethylene 6.7 C 4 – 10 Experiments for different polymers

Different conformations
Example 1: individual C-C bonds, simplest case ethane CH3-CH3
CH
HH
CH H
H
Both end form tetrahedrons.What is the relative orientation of these two tetrahedrions ?

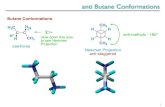
Example 2: n-butane 4 carbons
H H
HH H
H H
H H
H
H H
HH H
H H
H H
H
60 o
H H
HH H
HH
H
HH
60 o
Trans-conformation
All carbon atoms in the same plane
Gauche-conformation
G
G’
CH3 CH2 CH2 CH3

TT
TG
TG'
GT
GG
GG'
G'T
G'G
G'G'
9 potential combination: but only six are independentTG = GT, TG ’= G’T, and GG’ = G’G
33 nConformations … if the polymer have 10 000 carbons thus
onsconformati103 47709997 In general
(number of distingguishable conformations is less due to symmetry and also some comformations have low probability e.g. GG’)
Statistical treatment:Flory: Rotation isomeric state approximation. Each repeat unit can only be in T, G, or G’. Fluctuations around minima are ignores
Example 3: pentane 5 carbons

We have demostrated that the allowed conformations of the consequtive C-C bonds are T, G, G’. We can start constructing chains just by taking random orders of T, G, G’
T T
G
T
G
{TTGGTG’T….}= polymer chain conformation
Random coil model ideal ”phantom” chain
But long range interactions..
Polymer chain tends to revisit a spatial point that is already occupied. We have to take also account the chain segments that are ”far apart” - long range interactions.
Self avoiding walks – more open coils
Models for polymers

Individual polymer chains
• We want to eliminate interactions between the polymer chains • Analogy with the idal gas model: ideal gas = no interactions between the gas molecules - real
gas (interactions: repulsion and attractions)
Gas:
Low concentration:No interactions – like ideal gas
High concentration:Interaction between the molecules ”real gas”
Polymer solutions:
Low concentration – no interaction between the different chains
High concentration:Entanglemets etc..
But:In low concentration there is still internal interactions within the single polymer chain and interactions with the solvent molecules...
Dilute solutions

Example
Good solvent (T > 35 oC)”open coil” Rg large
CH2 CH n
Rg
But if solvent is changed to ethanol, which is ”bad” solvent for polystyrene ”compact coil’’ Rg small
Rg
Good solvent
”open coil””extended coil”
”Bad” solvent
”compact coil”
In general: -q solvent
Polymer-polymer and polymer-solvent interactions compensate each others...
1.
2. 1.
2.
Polymer repeat unit
Solvent molecule
polymer-polymer 1) interaction
>> polymer-solvent 2) interactionpolymer-polymer 1) interaction
<< polymer-solvent 2) interaction
CH2 CH2
CH2
CH2 CH2
CH2Low concentration 1% polystyre 99% cyklohexane

T > -q Temperature
”open coil””extended coil”
T < -q Temperature
”compact coil”
One can define for polymer-solvent system -q Temperature
T = -q Temperature
”in between” where Polymer-polymer and polymer-solvent interactions compensate each others
Polymer solubility depents typically also on Temperature .. In general solubility is better in high Temperatures (typical case but not always..)
Compare ideal gas Boyle-Temperature where attractive interaction between the gas molecules are equal to repulsive ”hard core” interactions. In this temperature gas molecules behave like ideal gas
q-Temperature

r
r1
r2 r3 ri
rn
General model:
Assume that the each mainchain bond (or repeat unit) in the polymer is a vectore segment ri
CH2 CH2
ri
Chain consist of these segments:n = total number of segmentsl = lenght of each segment
End-to-end vector:
n
iirr
1
vector: magnitude and direction
Vector magnitude
n
ij
n
ji
n
i
n
jji rrrrrrr
1 11 1
2 )()(
n
jiji
n
iii rrrr
1
cosABBA
Remember: where q is the angle between vectors
n
jiji
n
jiji
n
i
rrnlrrlr 2
1
22
Models for statistical chains

…Take large ensemble of different conformations
mean square end-to-end distance
n
jiji
n
jiji
n
i
rrnlrrlr 2
1
22
and end-to-end distance
n
jiji rrnlr22
ijji lrr cos2
This result is still a general formulation and is valid for any continuos polymer chain. (Unperturbed phantom chains, q-solvents and also polymer melt)
Average value of <r2>

1) The freely jointed chainThe freely jointed chain consist of a chain of bonds: the orientation of the different bonds is completely uncorrelated and no direction is preferred.
i.e. bond angle t [0, 180] and torsion angle f [0, 360] can have any value
n
jiij
n
jiji lnlrrnlr cos2222
There is no correlation between the segments: angle between the two bond vector qij (= 180-t) can have all values
0cos jiij
22 nlr
mean square end-to-end distance
average end-to-end distance 212 nlr
22 Cnlr now C = 1 C 4 – 10 Experiments for different polymers …

i.e. bond angle t is fixed but torsion angle f [0, 360] can have any value.
Carbon-carbon bond angle 110o
ii-1 i+1
jiforalsoij ,0cos
)180cos(21 lrr ii
222 )180cos( lrr ii
323 )180cos( lrr ii
.
.
. ijji lrr )180cos(2
ri ri+1
180-
ri ri+1
180-
ri+2
180-
n
jiji rrnlr22
ijk and180cos( inserting
summation can be performed over single variable (k) by
1
1
222 )(2n
k
kknlnlr
1
1
1
1
2 221
n
k
kn
k
k kn
nl
2) The freely rotating chain

1
1
1
1
22 221
n
k
kn
k
k kn
nlr m
m
k
km xxxxxS
...321
1
113211
11 ...
m
mmm
m
k
km xSmxxxxxS
On the other hnd)1()...1( 321
1 mm
m SxxxxxxS
mmm
m xSxSxxS )1(1
x
xxxSxxxSS
mm
k
km
mmm
1
1
1
1
Differentiate previous
)1(*)1(1
)1(12
1
1
1
x
xx
x
xmkx
mmm
k
k
2
1
1
1
)1(1
)1(1
x
xx
x
xmkx
mmm
k
k
Multibly by x
2
2
2
221
1 )1(
)1(
1
)1(1
)1(1
)1(
x
xx
x
xmx
x
xx
x
xmxkx
mmmmm
k
k
Geometrical series sum

1
1
1
1
2 221
n
k
kn
k
k kn
nl
))1(1
(2
121
2
122
nnn n
nnl
)11)1(
(2
121
2
122
nnn n
nnl
)1)1(
)(
2
121
2
2122
nnn n
nnl
)1)1(
(2
121
2
12
nnn n
nnl
1
2
)1(
2
121
2
12
nnn
nnl
2
2
)1(
)1(2
1
21
n
nnl
infinitely long chains (n=)
1
1
1
21 222 nlnlr
)180cos(1
)180cos(12
nl
if t=110o 222 2)70180cos(1
)70180cos(1nlnlr
freely rotating model gives C 2, for polyethylene chain good approximation at high T when T,G, and G’states are almost ”equally” populated.At low T trans is more populated and chain conformation is more extended
x
xxx
mm
k
k
1
1
1
2
221
1 )1(1
)1(
x
xx
x
xmxkx
mmm
k
k

ii-1 i+1
i.e. bond angle t is fixed and also torsion angle f can only have three values T, G, G’.
Relative population of T, G and G’ states depends on the energy levels (figure) and they are temperature dependent
n
jiji
n
jiji
n
i
rrnlrrlr 2
1
22
Again we can use same equation:
but this case it means a lot of work … see Example Gedde pages 26-27
cos(1
cos(1
)180cos(1
)180cos(122 nlr.. result
First term is the freely rotation result and second term is the correction due to hindered rotation
)cos( is the average value for cosf, where f is the torsion angle. And it is temperature dependend
3) The hindered rotation model

How to calculate average values?
i = 1 i =2 i = 3
Now we have a system which have a fixed number of states (3: G, T, and G’).System partition function Z measures the number of different states the system can adopt at the tremperature of interest
i
RTEieZ / At T = 0 this equals 1 and it increases with increasing temperature
Take any parameter f, which takes a value fi for the state i.
i
RTE
i
RTEi
i
i
e
eff
/
/
AverageNow for polyethylene 3 states:
i = 1 (Gauche) f1 = -120, E1 = Eg
i = 2 (Trans) f2 = 0 E2 = E t=0i = 3 (Gauhe’) f3 = 120, E3 = Eg
RTERTERTE
i
RTE gggi eeeeeZ //0/3
1
/ 21
Averages in statistical mechanics

cos(1
cos(1
)180cos(1
)180cos(122 nlr
121
121
)180cos(1
)180cos(1
211
1
211
1
)180cos(1
)180cos(1 22 nlnl
21
1
21
)120cos()0cos()120cos(/
/0/
RTE
RTERTE
g
gg
e
eee
3
2
)180cos(1
)180cos(12nl
RTEge /
3
1
/
3
1
/)(coscos
i
RTE
i
RTEi
i
i
e
e
For hindered rotation square end-to-end average
For example polyethylene at 140 oC, Et =0 and Eg 2.1,
54.0/ RTEge
2222 14.354.0*3
54.022
3
2
)180cos(1
)180cos(1nlnlnlr
Note that in this case the constant C is higher than freely rotating model- hindered rotation includes some rigidity to the chain.. but still less than experimentally found C = 6.7 for polyethylene
where
Average for cos(f), polyethylene

i.e. bond angle t is fixed and also torsion angle f can only have three values T, G, G’.
And potential energy of a given bond i depends on the states of the adjacent bonds i-1 and i+1
The conformation of n bond each having three possible torsion angles may specified by n-2 componenets. For example heptane 7 bonds: one possible conformation
TGTGG’ Total conformation energy
'GGTGGTTGT EEEEEE
4) The chains with interdependent bonds: statistical weight matrices
ii-1 i+1

The chains with interdependent bonds
1
1
1
'
'
G
G
T
uU
GGT
ii
…average square end-to-end distance …

Statistical problem - What is the propability that the chain displacement vector reaches from origin to the point r and lies within the volume element dV=dxdydz ?
Similar random process problems - Brownian particles
Previous models for chain conformation predicts quite well the polymer chain end-to-end distance scaling
21
nlr
But they does not lead any further analysis – for example what is the distribution of end-to-end distances r ?
Random flight analyzis – The Gaussian Chain

Also similar random process problem – rifle shots on a target

The function which models random processes is the Gaussian function
2)/(
)(xe
xp
1D The probability that polymer end-to-end lenght lying between x and x+dx is the product of p(x) and dx
dx
edxxp
x
2)/(
)(
correspondingly in 3D
dxdydz
edxdydz
e
dxdxdyzpypxpdxdydzzyxPrzyx
3
/
3
/)(
)()(
)()()(),,(222222
The probability that chain end-to-end lenght lies between r and r+dr is the product of P(r) and dV
drrdV 24
(4pr2 is the area of the sphere)
drr
edVrP
r2
3
/
4)(
)(22

Mean square value of r ?
1
4)(
1
)(
)(2/
30
2
0
0
2
2
22
drrer
dVrP
dVrPrr
r
drer r 22 /
0
43
)(
4
aa
ndrex
nnaxn
10
2
2
)12...(5312
definite integral from mathematics tables
now n = 2 and a = 1/r2
2
22
33
/
0
43 /1)
1(2
31
)(
4
)(
4 22
drer r
2
38
34
4
2/1
2/332 r
And from random freely jointed chain 22 nlr
ln
nlr2/1
22
2
3
2
3

Crystallization:High density polyethylene HDPE
•Crystallization
•Liquid crystals or stiff chains
•helices
-Chains with preferred conformations

nC CF
F
F
F
If hydrogen atoms (from PE) are replaced by fluorine atoms with van der Waals diameter 270 pm – to accommodate the fluorine atoms a rotation around each C-C bond of about 20o is induced. This is accompanied by a slight opening of the C-C chain bond angle to about 116o
poly(tetrafluoroethylene) (PTFE)
13:1 helix

Vinyl polymers: example Isotactic PP (i-PP)


Dense Polymer Systems: Entanglements
How can we sketch a melt of polymers ? Given the red polymer with N monomers, we call N’ the number of monomers in between two entanglements
In a polymer melt where N>>N’, a chain will see N/N’ obstacles or entanglements . It is the number of entanglements (or N/N’) which rules motion of macromolecules in melt.
We now aim at describing a theory which captures sufficient details of a polymer motion, but with a picture as simple as possible:
Problems should be made as simple as possible….but not any simpler !!! (A. Einstein)
From Mezzenga lecture

Reptation Model (in simple linear chains)
Suppose to Sketch a chain in a melt of chains constrained within obstacles O1, O2. These obstacles represent the entanglements, and the chain is not allowed to cross them.
How can the chain move within this network ? The obstacles are fixed. The chain can only move tangentially to its countour, since transversal motion is impeeded.
The chain moves as a snake, a reptile and the movement is called reptation (after DeGennes)
From Mezzenga lecture

From Rubinstein talk

From Rubinstein talk

Diffusion of Branched Chains We study now the diffusion of branched chains. Lets start with a chain with 1 side group having Ns monomers.
If N2 monomers now carry a side chain with having Ns monomers (and N1 are free) we can work out the total force f to apply to the chain to have a contour velocity v: f=v(N1μ1
-1 + N2μ2-1)
In order to be mobile a branch unit with Ns monomers needs to refold back. If z is the
coordination of the lattice, this may happen with zNs possibilities.
The probability for the branch chain to get back to the branch starting point is then P≈e-αNs with α≈1. Thus we can write μ2=μ1 e-αNs and f=vμ1
-1(N1+ N2 eαNs ).
Ns
tubeNstube eNN
D
N
T
L
eNN
211
22
t21
1 and
The effect of branching is exponential. Suppose N2=1.
τis dominated by branching as soon as Ns>1/α*ln(N1)
The motion of the main red chains is identical as before. However, the mobility of J, the monomer to which the side chain is attached is not longer μ1 but μ2
From Mezzenga lecture

From Rubinstein talk