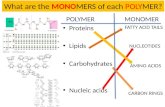
Basic Vocabulary Monomer – basic unit of a polymer Polymer – Large molecule composed of...
-
Upload
dominic-clark -
Category
Documents
-
view
223 -
download
2
Transcript of Basic Vocabulary Monomer – basic unit of a polymer Polymer – Large molecule composed of...


Basic Vocabulary
Monomer – basic unit of a polymer
Polymer – Large molecule composed of repeating basic units or monomers

Polymerization

1. CARBOHYDRATES1. CARBOHYDRATES
Contain the elements carbon, hydrogen, and oxygen Contain the elements carbon, hydrogen, and oxygen Hydrogen is in a 2:1 ratio to Oxygen – this means that Hydrogen is in a 2:1 ratio to Oxygen – this means that
for every 2 Hydrogens there is 1 Oxygenfor every 2 Hydrogens there is 1 Oxygen
key source of ENERGYkey source of ENERGY Ex. Glucose and StarchesEx. Glucose and Starches
Plants use carbohydrates for structurePlants use carbohydrates for structure Ex. CELLULOSEEx. CELLULOSE

MonosaccharidesMonosaccharides
all have the formula Call have the formula C66 H H1212 O O66
all have a Carbon Ring structure all have a Carbon Ring structure Ex. GlucoseEx. Glucose

DisaccharidesDisaccharides all have the formula Call have the formula C1212 H H2222 O O1111
Notice the number of atoms has increased Notice the number of atoms has increased in this formula! in this formula!
Ex. SucroseEx. Sucrose

Polysaccharides Polysaccharides
Formed of three or more simple sugar units Formed of three or more simple sugar units Glycogen - animal starch stored in liver & Glycogen - animal starch stored in liver &
muscles muscles Cellulose - indigestible in humans - forms Cellulose - indigestible in humans - forms
cell walls cell walls Starches - used as energy storage Starches - used as energy storage

How are complex How are complex carbohydrates formed carbohydrates formed
and broken down? and broken down?

Dehydration Synthesis Dehydration Synthesis ReactionReaction Combining simple molecules to form a more Combining simple molecules to form a more
complex one with the removal of water complex one with the removal of water
ex. monosaccharide + monosaccharide disaccharide + water
C6H12O6 + C6H12O6 C12H22O11 + H2O
Polysaccharides are formed from repeated Polysaccharides are formed from repeated dehydration syntheses of water dehydration syntheses of water
○ They are the stored extra sugars known as starchThey are the stored extra sugars known as starch


Hydrolysis Reaction Hydrolysis Reaction
Addition of WATER to a compound to Addition of WATER to a compound to SPLIT it into smaller monomersSPLIT it into smaller monomersEx. chemical digestionEx. chemical digestion
disaccharide + Hdisaccharide + H22O O monosaccharide + monosaccharide monosaccharide + monosaccharide
CC1212 H H2222 O O1111 + H + H22 O O C C6 6 HH1212 O O66 + C + C66 H H1212 O O66


2. LIPIDS2. LIPIDS Function in long-term energy storage, protection, and Function in long-term energy storage, protection, and
insulation insulation
Contain carbon, hydrogen, and oxygen Contain carbon, hydrogen, and oxygen butbut the H:O ratio is the H:O ratio is notnot in a 2:1 ratio in a 2:1 ratio
large moleculeslarge molecules Ex. Fats, oils, waxes, steroids Ex. Fats, oils, waxes, steroids

LIPIDS continued Lipids are formed from the union of 1 glycerol Lipids are formed from the union of 1 glycerol
molecule and 3 fatty acid moleculesmolecule and 3 fatty acid molecules
3 fatty acids + glycerol 3 fatty acids + glycerol lipid lipid
Fats – primarily in animalsFats – primarily in animals Oils and waxes – primarily in plantsOils and waxes – primarily in plants
Oils are liquid at room temperature, waxes are solids Oils are liquid at room temperature, waxes are solids
Ex. Steroids - used to build many reproductive Ex. Steroids - used to build many reproductive hormones and cholesterolhormones and cholesterol
Ex. Phospholipids – major component of cell membrane

3. PROTEINS3. PROTEINS Contain the elements carbon, hydrogen, Contain the elements carbon, hydrogen,
oxygen, and nitrogenoxygen, and nitrogen
Composed of MANY Composed of MANY amino acidamino acid monomers monomers
The arrangement of the amino acid that forms the The arrangement of the amino acid that forms the primary structure of proteins. primary structure of proteins.

PROTEINS continued Basic amino acid has a Basic amino acid has a
1.1. carboxyl group carboxyl group 2.2. methyl group – 1 methyl group – 1
hydrogen attachedhydrogen attached3.3. R Group attached to R Group attached to
the methyl groupthe methyl group4.4. amino group – amino group –
includes nitrogenincludes nitrogen
The R Group is a group of The R Group is a group of atoms that is different for atoms that is different for each amino acid. It each amino acid. It determines the protein’s determines the protein’s shape.shape.

Major Protein Functions Major Protein Functions
Growth and repair of tissuesGrowth and repair of tissues Helps organisms access energy Helps organisms access energy Buffers – helps keep pH levels of Buffers – helps keep pH levels of
body constant body constant

Dehydration Synthesis of Dehydration Synthesis of Dipeptide Dipeptide
Building a protein chainBuilding a protein chain Formed from two amino acid subunits Formed from two amino acid subunits
amino acid + amino acid amino acid + amino acid dipeptide + water dipeptide + water

Hydrolysis of a Dipeptide Hydrolysis of a Dipeptide Breaking down of a dipeptide into amino acidsBreaking down of a dipeptide into amino acids
dipeptide + Hdipeptide + H22O O amino acid + amino acid amino acid + amino acid

Polypeptide (protein) Polypeptide (protein) Composed of three or more amino acids linked Composed of three or more amino acids linked
by synthesis reactions by synthesis reactions
Ex. Insulin, hemoglobin, and enzymes. Ex. Insulin, hemoglobin, and enzymes.

Protein Structure Primary – sequence of
chain of amino acids
Secondary – occurs when the sequence of amino acids are linked by Hydrogen bonds
Tertiary – occurse when certain attractions are present between alpha helices and pleated sheets
Quaternary – is a protein consisting of more than one amino acid chain

4. NUCLEIC ACIDS4. NUCLEIC ACIDS Composed of nucleotidesComposed of nucleotides
NucleotidesNucleotides consist of 3 parts: consist of 3 parts:○ 5-Carbon Sugar5-Carbon Sugar○ Phosphate GroupPhosphate Group○ Nitrogenous BaseNitrogenous Base

NUCLEIC ACIDS continued Located in all cells
Functionsstore and transmit
hereditary/genetic information

DNA (DeoxyriboNucleic Acid) DNA (DeoxyriboNucleic Acid) contains the genetic code of instructions that contains the genetic code of instructions that
direct a cell's behavior through the synthesis direct a cell's behavior through the synthesis of proteins of proteins
found in the chromosomes of the nucleus found in the chromosomes of the nucleus (and a few other organelles) (and a few other organelles)

RNA (RiboNucleic Acid) RNA (RiboNucleic Acid) directs cellular protein synthesis directs cellular protein synthesis found in ribosomes, nucleoli, and cytoplasm found in ribosomes, nucleoli, and cytoplasm