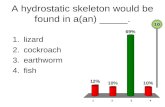
Animal Nervous System (Earthworm & Fish)
-
Upload
keth-laurel-catayas -
Category
Documents
-
view
224 -
download
0
Transcript of Animal Nervous System (Earthworm & Fish)
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
1/18
1
Animal Physiology lab Group 2
4B Benson, Jonah
Date Performed: (leader) Catayas, Keth Laurel
Date Submitted: Legarda,Criscire
Activity 3
Animal Nervous System
I. Earthworm Dissection
Materials and MethodsMaterials: Reagents:
Earthworm Ringers Solution
Dissection tray - 6g NaClDissection pins - 0.12g KClForceps - 0.2g CaCl2
Scalpel - 0.1g NaHCO3Scissors - bring to 1 liter distilled waterProbeWater bath
WaterIce
After the preparation of materials and reagents, the earthworm was euthanized in
the water bath with ice for 15 minutes. After 15 minutes, the body length and weight ofthe worm was obtained using a ruler and an analytical balance, respectively. Photos were
taken for documentation.
Figure 1. Dorsal side of the earthworm
The euthanized earthworm was placed on the dissecting pan, dorsal side up. The
dorsal side was determined using two ways: first, by locating the dorsal side of the
clitellum (without gonopores) and second, by finding the dorsal blood vessel which was a
very prominent dark line that ran throughout the worms body (Figure 1).
Dorsal blood vessel
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
2/18
2
Then, the worm was pinned first through the prostomium (first segment). This
step was done carefully since the cerebral ganglion is located in the third segment which
was very near to the prostomium. To completely put the worm in place, the anal (last
segment) was pinned (Figure 1).
With forceps, the dorsal skin was lifted. The scissors was inserted at the base of
the forceps to make a small slit on the skin (Figure 2). Beginning on the slit, the skin was
cut towards the anus (Figure 3). The separated body wall was pinned through the
dissecting pan to hold the worm firmly while continuously cutting the skin of the worm
towards the prostomium. After cutting through the entire worm, the septa was severed
using the scalpel, and the separated body wall was again pinned to the tray (Figure 4).
Once the internal organs were fully exposed, Ringers solution was poured to keep the
organism from dehydrating and free from dirt and blood (Figure 5).
Figure 2. Incision to the worms skin Figure 3. Incision towards the anus
Figure 5. Application of Ringers solutionFigure 4. Cutting of septa
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
3/18
3
After cleaning the worm, the cerebral ganglion (brain) was found near the anterior
and dorsal part of the pharynx. This structure was not that evident since it encircled the
pharynx. The brain was isolated by carefully cutting the pharynx and removing the excess
tissue trapped in the middle of it.
Next, the pharynx, esophagus, and other internal organs were pushed aside to
expose the ventral nerve cord and segmental ganglia which looked like white bulges
(Figure 6). Ringers solution was again poured to the worm. Using a scalpel, a thin layer
of film above the nerve cord was scraped to make the cord more visible, thus making the
isolation easier (Figure 7). The nerve cord and segmental ganglia were simultaneously
isolated by separating the nerve cord from the underlying ventral body wall using a probe
(Figure 8). This step was done until the last segmental ganglion was separated. The
isolated nervous system was placed in a petri dish with Ringers solution (Figure 9).
After the dissection, the materials and the laboratory area were cleaned. The
remains of the earthworm were buried on the area assigned previously by the teacher.
B. Results and Discussion
It was known that the body size of any organism is relatively 30-fold of that the
size of the brain. Due to this information, the length and weight of the earthworm were
Figure 7. Removal of the thin film
Figure 8. Isolation of ventral nerve cord Figure 9. Nerve cord in Ringers solution
Figure 6. Removal of internal organs
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
4/18
4
obtained. The model organism measured 11.5 cm long (Figure 10a) and weighed 1.23 g
(Figure10b).
After dissection, the nervous system of the earthworm was easily identified due to
its prominent white color. The major organs isolated were the cerebral ganglion and the
segmental ganglia along the ventral nerve cord. These segmental ganglia were
characterized due to their bulged appearance along the entire length of the nerve cord.
Peripheral nerves also arose on the sides of the nerve cord which looked like thin hair-
like projections (Figure 11).
The central nervous system is primarily composed of a bilobed cephalic ganglion
and a ventral nerve cord, with one ganglion per segment, extending through the whole
length of the body. The segmental ganglionic enlargements vary in size, shape, and
approximation at different parts (Clarke, 1856). The two lobes of the cerebral ganglion
are connected by a pair of circumpharyngeal connectives to the most anterior of the
ventral ganglia, the subpharyngeal ganglion (Mill, 1982).
The cephalic or cerebral ganglion rests on the beginning of the pharynx, beneath
the dorsal part of the third ring. Each lobe is a pyriform sac, which is very thick and
convex posteriorly. The convex portion is opaque-white in color and filled with oval,
round and pyriform cells, of various sizes. The anterior half, where the lobes are joined
are composed with a line of lamina cells. The interior of this portion is entirely fibrous.
The cephalic nerves are attached to the upper part of the ganglion. Roots of the nerves
immediately separate into two trunks; a lower and upper. The former runs above the
mouth, to the underside of the first segment, or upper lip (Clarke,1856)
a b
Figure 10. Measurements obtained from the earthworm (a) length (b) weight
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
5/18
5
.
Figure 11. Earthworm nervous system
Cerebral an lion
Circumpharyngeal
connective
Peri heral nerves
Segmental gangliaVentral nerve
cord
Subpharyngeal
ganglion
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
6/18
6
The peripheral nervous system, on the other hand, consists of the lateral
segmental nerves that project along the ventral nerve cord. Typically, each ventral
ganglion posterior to the subpharyngeal ganglion gives rise to three nerve pairs of lateral
nerves, in which the 2nd
and 3rd
segments are attached to each other via nerve to septum.
These lateral nerves supply filaments to the septa and muscular bands (longitudinal,
oblique, and circular muscles). In addition, a stomatogastric nervous system arises from
the circumpharyngeal connectives, which innervates the pharynx and the anterior region
of the intestine (Mill, 1982). The prostomial and prestomial nerves are distributed to the
muscular bands of the mouth, which after supplying the muscles of the anterior segments,
terminates in the integument of the lower lip (Clarke, 1856).
II. Fish Dissection
A. Materials and Methods
-Fish Phosphate Buffered Solution:-Dissecting tray - 6.7g NaCl
-Scissors - 0.1g KCl-Pins - 100ml 0.1 M PO4, pH 7.3
-Forceps - 150ml Distilled H20-Scalpel
Spinal Cord
To start the dissection proper, the body weight and length of the fish was measuredfirst using an analytical balance and a ruler, respectively. Photos were taken for
documentation purposes. The fish was placed on the dissecting tray with the head on the
left and using the scalpel, an incision was made through the body wall of the fish. Thefirst incision was at the belly of the fish from the front edge of the operculum through thetrunk to the anus (Figure 12).
Figure 12. Incision 1 Figure 13. Incision 2
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
7/18
7
The second incision was done along the side of the fish near the caudal fin to thebackbone (Figure 13). Along the dorsal side of the fish near the lateral line, the last
incision was completed through the ribs along the backbone to the tip of the operculum(Figure 14).
Figure 14. Incision 3 Figure 15. Removal of trunk flesh
The incisions were repeated at the adjacent side of the fish. The portion of the trunkand other organs were removed except the head and the backbone (Figure 15). Thebackbone was isolated from the head for easier isolation of the spinal cord (Figure 16).
Figure 16. Separation of the head Figure 17. Removal of spines
Before isolating the spinal cord, the spines of the vertebrae and other soft tissuescovering the vertebrae were removed using the scissors (Figure 17). The spinal cord was
then isolated gently by using freehand method, in which each segments of the vertebraewere gently separated (Figure 18). The spinal cord was placed immediately in the PBS in
preparation for staining (Figure 19).
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
8/18
8
Figure 18. Isolation of spinal cord Figure 19. Spinal cord in PBS
Brain
Using the scissors, the soft tissue along the ventral side of the skull was removed
(Figure 20). The skin and the tissue on the dorsal side of the skull were removed by usingthe scalpel (Figure 21).
Figure 20. Removal of ventral tissue Figure 21. Removal of dorsal tissue
The skull was carefully opened by removing the skull bone on dorsal side of the brainusing the forceps. The tissue surrounding the forebrain was removed using the forceps
(Figure22). The brain was the gently isolated from the braincase and immediately placedon the PBS for staining purposes (Figure 23).
Figure 22. Isolation of brain Figure 23. Brain in PBS
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
9/18
9
B. Results and Discussion
It is said that the any organisms body size is relatively 30-fold of that the size ofthe brain. With regards to this information, the weight and length of the fish were taken.
The organism measured 112.90 g in weight (Figure 24a) and 21.5 cm in length (Figure24b).
a b
Figure 24. Measurements obtained from the fish (a) weight (b) length
After the dissection, the nervous system was easily recognized because of its
distinct morphological characteristics. The nervous system of the fish was composed of aspinal cord and a brain (Figure 27). The brain was reddish-white in color, measuring up
to 1.3 cm long (Figure 25a) and 1.2 cm wide (Figure 25b). The brain was protectively
encased within the skull and had several clearly visible parts. The only parts identifiedwere the telencephalon, optic lobes and cerebellum. The telencephalon was small, oftenappeared to be as separate two lobes and located at the anterior part of the two optic
lobes. The optic lobes were the most prominent among the parts observed. The opticlobes occurred also in pairs and situated between the telencephalon and cerebellum. The
other optic lobe (at the left side facing the telencephalon) appeared to be indefinite andlightly distorted, possibly due to improper handling the skull bone during brain isolation.
Lastly, the cerebellum showed as a single-lobed structure, which was positioned at theposterior part of the brain. Furthermore, it was divided by various lobes; however, folds
were not present (Figure 26).
The spinal cord appeared to be as a thick, white, and cylindrical nervous material
that was attached from the base of the brain (posterior part) and extends along the fulllength of the fish's body. The spinal cord was thin, soft, and delicate, and was protected
by the spinal column or vertebrae. It was decided not to measure the spinal cord, sinceonly half of the spinal cord of the fish was isolated and the isolated parts were too
fragmentary (Figure 27). The spinal and cranial nerves were not evident along the wholesurfaces of the spinal cord and brain, respectively.
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
10/18
10
a b
Figure 25. Measurements obtained from the brain (a) length (b) width
Fish typically have quite small brains relative to body size compared with other
vertebrates, typically one-fifteenth the brain mass of a similarly sized bird or mammal,and is poorly developed compared to other vertebrates (Helfman et al., 1997).
Fish have highly developed nervous systems organized around a brain. A bonyfish's brain is divided into three sections: the forebrain, the midbrain, and the hindbrain.
The forebrain of fish is dominated by the olfactory lobes, a pair of structures that receiveand process signals from the nostrils via the two olfactory nerves that are responsible for
the bony fish's ability to smell. Behind the olfactory lobes is the two-lobed telencephalon,the structural equivalent to the cerebrum in higher vertebrates. In fish, the telencephalonis concerned mostly with olfaction. It also seems to control behaviors such as taking care
of the young and exploring the environment. Fishes that have an especial good of smelland primarily hunt by smell, such as eels, catfish, hagfish, and sharks have an enlarged
forebrain (Helfman et al., 1997). The teleosts, for which sight is often the most importantsense, have smaller olfactory lobes (Ramel, 2012).
Connecting the forebrain to the midbrain is the diencephalon which is locatedbelow the optic lobes and basically not visible. The diencephalon performs functions
coupled with hormones and homeostasis. The pineal body lies just above thediencephalon which involved in detecting light, maintaining circadian rhythms, and
controlling color changes (Helfman et al., 1997).
The midbrain or mesencephalon of a fish consists mostly of the two optic lobes,
which vary greatly in size between species in accordance with their dependence on sight.In fish, the mid-brain is important in sorting out incoming information and it is also the
main centre of learning (whereas in mammals it is the forebrain that is the main centre oflearning). The optic lobes may be so large that they completely cover the forebrain such
as those species that hunt by sight, like rainbow trout and cichlids. Blind bony fishes,such as blind cavefishes in the family Amblyopsidae, have a reduced midbrain (Helfman
et al., 1997; Ramel, 2012).
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
11/18
11
Figure 26. Earthworm brain
The hindbrain or metencephalon (medulla and cerebellum) is particularly
involved in swimming and balance. The cerebellum is a single-lobed structure thatcontrols motor coordination. This means that it controls the timing and interaction of
muscles once a muscular action has been initiated. The cerebellum is also important inmaintaining equilibrium. Fast-swimming bony fishes usually have an enlarged hindbrain.
Hagfish and lampreys have relatively small cerebellae, while the mormyrid cerebellum isbig and involved in their electrical senses. The medulla on the other hand, controls the
operations of the inner organs such as heart rate, blood pressure, digestion and wastedisposal. It is also a relay centre for many nerves sending messages to and from the mid
and forebrain. The brain stem or myelencephalon is the brain's posterior. As well as
controlling some muscles and body organs, in bony fish at least, the brain stem governsrespiration and osmoregulation (Helfman et al., 1997; Ramel, 2012).
Posterior to the brain is the spinal cord, which is the hollow dorsal nerve cord that
is characterized by chordates. It is a thick sheath of nervous material that extends to thefull length of the fish body, and protected by the neural canal of the vertebral column
(Monterey Bay Salmon and Trout Project, 2009). It serves as the basis of many simple
Optic lobes
Telencephalon
Cerebellum
Posterior Part
Anterior Part
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
12/18
12
responses and act as the major link to the brain for sensory input and brain responses(Ramel, 2012).
Apart from the brain and the spinal cord is a vast network of spinal nerves thatexits from the cord and connects to the internal organs and muscles. Nerves are built of
numerous neurons that travel the message to or from the brain or the spinal cord along aneuronal pathway (Ramel, 2012)
Figure 27. Fish nervous system
III. Staining of Nervous System
A. Materials and Methods
First, the Golgi-Cox stain was prepared by making three solutions; (solution A)
5% potassium bichromate solution, (solution B) 5% mercuric chloride solution, and 5 %
potassium chromate solution. In preparing the solutions, 500 g of potassium bichromate,
mercuric chloride, and potassium chromate were weighed individually using an analytical
balance. Five hundred grams of potassium bichromate was stirred into warm 1000 ml
Spinal Cord
Brain
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
13/18
13
a
Figure 28. Isolated nervous systems wrapped in gauze (a) earthworm (b) fish
a
Figure 29. Wrapped nervous systems in Golgi-cox stain (a) earthworm (b) fish
distilled water until dissolved. Five hundred grams of mercuric chloride was also stirred
into warm 1000 ml distilled water until dissolved. Five hundred grams of potassium
chromate was added into cold 1000 ml distilled water while being continuously stirred
until dissolved (Wright et al., 2011).
Then, 100 ml of solution A was added to solution B. Separately, 120 ml of
distilled water was added to 80 ml of solution C. The A/B solution was then slowly added
to the diluted solution C, while continuously stirring. An orange color was observed in
the final solution. The Golgi-cox solution was stored in the dark when not in use (Wright
et al., 2011).
Next, the newly isolated nervous systems of earthworm and fish were individually
wrapped in gauze and placed in separate vials (Figure 28a and 28b). The gauze was
completely immersed in the Golgi-cox stain (Figure 29a & 29b). Vials were sealed and
also kept in the dark room for 14 days (Wright et al., 2011).
After 14 days, the stained nervous systems were harvested. The parts were
removed from the gauze and placed in two separate petri dishes, one for the earthworm
and the other for the fish, containing distilled water. Fractions or slices of the nervous
system were immersed in diluted ammonium hydroxide (100ml of 20% ammonium
b
b
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
14/18
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
15/18
15
In Lumbricus terrestris, the segmental nerves are very important because they
serve as input centers from different sensory neurons. The tactile sensory axons enter the
central nervous system via the 1st
and 3rd
segmental nerves. Chemoreceptive inputs, on
the other hand, pass primarily through 1st
and 3rd
segmental nerves, but also to some
extent via the 2nd segmental nerves. Lastly, proprioceptive inputs (awareness of body
position) enter the CNS via all three segmental nerves (Mill, 1982).
Figure 31. Motor and sensory neurons (a) schematic diagram (b) actual ganglion stained
with nickel chloride (Mill, 1982)
The neurons were identified using the figures (Figure 31a and 31b) from the studyof Mill (1982). In which, the schematic diagram and actual stained ganglion showed the
distinct dark-pigmented cell bodies of both motor and sensory neurons lying on the sides
of the ganglion.
In Lumbricus, the sense organs are concentrated in two bands encircling the
animal in each segment. Each sense organ contains great number sensory cells. There are
Sensory
neurons
Motor
neurons
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
16/18
16
as many as 50 sensory neurons in each sense organ associated to supporting cells to
which they are attached by peripheral zonula adherens and desmosomes (Mill, 1982).
Furthermore, motor neurons are mostly found on the sides of the segmental
ganglia. There are 26 pairs of motor neurons with contralateral cell bodies in eachmidbody ganglion. The axons of these enter the CNS through the 1
stand 3
rdsegmental
nerves. There are also four pairs of giant motor neurons in each ganglion (Mil, 1982).
Unfortunately, these giant neurons were not located and identified.
Fish
a bFigure 32. Fish Neurons (a) Brain 100x (b) Spinal Cord 100x
After almost 3 weeks of staining, the neurons were examined and their structures
were identified under the microscope. The neurons from the brain and spinal cord were
easily identified from others because of their similarities in branching characteristics.
Only few cell bodies were evident and commonly distinguished because of their dark
pigment-like structures, with few (two to three) or lightly dense set of dendrites arising
from all over the surface. The cell bodies were almost asymmetrical and some were
Cell body
Axon
Cell body
Cell body
Axon
Dendrites
Dendrites
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
17/18
17
relatively circular in shape. The lightly visible dendrites were numerous, thin, short, and
highly branched from their origin (cell bodies). Axons were few and structurally thin and
long and frequently extend away from the soma or cell body. The axons appeared to be as
thin fragments of thread that were aligned in linear fashion. Each of these axons was
found connected with other cell bodies. Some dendrites and axons were superficially
similar in structure under the microscope which somehow made the identification of the
parts very complicated (Figure 32a & b).
The neurons of humans are similar to the neurons of other animals like the fish. A
typical neuron has an enlarged area called the cell body, which contains the nucleus.
Neurons have a large number of extensions called dendrites. They often look like
branches extending out from the cell body. The dendrites receive the information from
the neurons. Each neuron usually also has a longer tail-like structure called an axon,
which transmits information to other cells. This structure can easily be distinguished from
the dendrites because of its length. Longer axons are usually covered with a fatty,
segmented covering called the myelin sheath. This covering acts as an insulator which
improves the ability of axons to carry nervous system signals rapidly. At the very end of
the axon is the axon terminal also known as terminal button or synaptic knob which is
separated from the next cell by a tiny gap called a synapse (Moreno & Tharp, 2007).
-
7/30/2019 Animal Nervous System (Earthworm & Fish)
18/18
18
REFERENCES
Helfman, D., Collette, B., & Facey, A. (1997). The Diversity of Fishes. Blackwell
Publishing, pp. 4849, 191.
Lockhart, C. 1856. On the nervous system ofLumbricus terrestris.
Mill, P. J. 1982. Recent Developments in Earthworm Neurobiology. Comprehensive
Biochemistry and Physiology,73, 641-661.
Moreno, N. P. & Tharp, B. Z. 2007. What Are Neurons?. Baylor College of Medicine.
Monterey Bay Salmon and Trout Project. 2009. Fish Nervous System. Retrieved from
http://mbstp.org/Fish/thenervoussystem.html
Ramel, G. 2012. Fish Nervous System. Retrieved from http://www.earthlife.net/fish/nerves.html
Wright, K. A., Zimerman, E. L. & Harrington, M. E. 2007. A modified golgi-cox
procedure in undergraduate courses. The Journal of Undergraduate NeuroscienceEducation, 10, 85-87.