1 II. Enzymes Proteins Organic catalysts that speed up the rate of a reaction, but are not used up...
-
Upload
randall-wright -
Category
Documents
-
view
212 -
download
0
Transcript of 1 II. Enzymes Proteins Organic catalysts that speed up the rate of a reaction, but are not used up...
1
II. Enzymes
• Proteins• Organic catalysts that speed up the rate of a
reaction, but are not used up• Lower energy of activation• Are specific in action, i.e., act on a specific
substrate
3
II. Enzymes, cont.
• 1. Substrate (S) combines at active (catalytic) site of enzyme (E).
• 2. Enzyme-substrate complex [ES] is formed.
• 3. The substrate is transformed into a product (P).
• 4. The products are released from enzyme.• 5. The enzyme is recovered.
6
II. Enzymes, cont.
• Naming of enzymes according to reaction:– oxidoreductase Involved in oxidation and
reduction reactions.– transferase Transfers functional groups.– hydrolase Hydrolysis.– lyase Removes atoms without hydrolysis.– isomerase Rearranges atoms in a molecule.– ligase Joins two molecules.
7
II. Enzymes, cont.
• Location of enzymes:– intracellular Endoenzymes. Act inside the cell.– extracellular Exoenzymes. Act outside the cell.
• Enzyme components:– apoprotein Protein portion.– cofactor Nonprotein portion. May be a metal ion
or an organic molecule which is called a coenzyme.
– holoprotein Apoprotein + cofactor.
9
II. Enzymes, cont.
• Coenzymes– Bind to an enzyme transiently– Are “carrier” molecules. They carry atoms (or
electrons) to the substrate or take atoms (or electrons) from the substrate.
– Examples • NAD+ is an electron carrier.• NADP is an electron carrier.• ATP is a phosphate and energy carrier.
10
II. Enzymes - Factors affecting activity
• 1.Temperature– Enzyme activity
increases with increasing temperature.
– Activity drops when heat denatures enzyme.
11
II. Enzymes - Factors affecting activity
• 2. pH– Enzymes have
an optimum pH.– Hydrogen ions
alter protein structure.
12
II. Enzymes - Factors affecting activity
• 3. Substrate Conc.– Rate of reaction
increases until all active sites are filled.
– At saturation, maximum rate is reached.
13
II. Enzymes - Factors affecting activity
4. Inhibitors– competitive -
react at the substrate site.
– noncompetitive -do not react at the substrate site.• e.g., allosteric
inhibitors
16
II. Enzymes, cont.
• Control of enzyme activity– genetic regulation The control of enzyme
synthesis. – metabolic regulation The control of enzyme
activity following enzyme synthesis. • Allosteric activators increase enzyme activity.• Allosteric inhibitors decrease enzyme activity, e.g., in
feedback inhibition, the end product of a pathway turns off the first enzyme of the pathway.
17
III. Energy productionIII. Energy production
• Carbohydrates, proteins, lipids are energy rich.• Each is a reduced molecule, i.e., has many
hydrogen atoms (an electron, e- , & a proton, H+)• Upon being oxidized, the molecule loses electrons.• The energy associated with the electrons is
ultimately conserved in ATP.• Are catabolic reactions
19
EnzymesEnzymes
• Biological catalysts produced by cells• Nearly all are proteins• Enormous catalytic power
– Reactions occur at lower temperatures and at higher rates
• Ordinarily highly specific
20
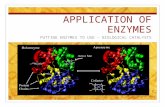
Induced-Fit Model of Enzymes
• Explains how enzyme works• Substrate: reacting substance• Active site: where chemical
reaction takes place and where substrate fits
21
CofactorsCofactors
• Something other than polypeptide chain required by enzyme
• May be metal– Iron in hemoglobin
• May be organic cofactor– Coenzyme
• Apoenzyme: does not have cofactor
22
Inhibition of EnzymesInhibition of Enzymes
• Lets cell control when an enzyme works
• Inhibitor binds to allosteric site
• Prevents substrate from binding
23
Enzyme Classification I
Systematic Names and Recommended Names are based on the reaction type and the molecular structure of the substrates
(But: they do not represent the holo-enzyme)
24
Enzyme Classification II
1. Oxidoreductases - catalyzing oxidation reduction reactions.
2. Transferases - catalyzing transfer of functional groups.
3. Hydrolases - catalyzing hydrolysis reactions.4. Lyases - catalyzing group elimination reactions to
form double bonds.5. Isomerases - catalyzing isomerizations (bond
rearrangements).6. Ligases - catalyzing bond formation reactions
couples with ATP hydrolysis.
25
1-Oxidoreductases
1.1 Acting on the CH-OH group of donors 1.1.1. With NAD or NADP as acceptor1.1.2. With a cytochrome as acceptor1.1.3.With oxygen as acceptor1.1.4.With a disulfide as acceptor1.1.5.With a quinone or similar compound as
acceptor1.1.99. With other acceptors
1.2 Acting on the aldehyde or oxo group of donors1.3 Acting on the CH-CH group of donors 1.4 Acting on the CH-NH2
group of donors
…
26
2. Transferases
2.1 Transferring one-carbon groups2.1.1. Methyltransferases2.1.2. Hydroxymethyl-, Formyl- and Related Transferases2.1.3. Carboxyl- and Carbamoyltransferases2.1.4. Amidinotransferases
2.2 Transferring aldehyde or ketonic groups2.3 Acyltransferases2.4 Glycosyltransferase...
27
3. Hydrolases
3.1 Acting on ester bonds3.1.1 Carboxylic Ester Hydrolases3.1.2 Thiolester Hydrolases3.1.3 Phosphoric Monoester Hydrolases3.1.4 Phosphoric Diester Hydrolases3.1.5 Triphosphoric Monoester Hydrolases
...3.2 Glycosylases3.3 Acting on ether bonds3.4 Acting on peptide bonds (peptidases)
28
4. Lyasen
4.1 Carbon-carbon lyases 4.1.1 Carboxy-lyases4.1.2 Aldehyde-lyases4.1.3 Oxo-acid-lyases4.1.99 Other Carbon-carbon lyases
4.2 Carbon-oxygen lyases4.3 Carbon-nitrogen lyases4.4 Carbon-sulfur lyases4.5 Carbon-halide lyases
29
5. Isomerases
5.1 Racemases and epimerases5.1.1. Acting on Amino Acids and Derivatives5.1.2. Acting on Hydroxy Acids and Derivatives5.1.3. Acting on Carbohydrates and Derivatives5.1.99. Acting on Other Compounds
5.2 cis-trans-Isomerases5.3 Intramolecular isomerases5.4 Intramolecular transferases (mutases)5.5 Intramolecular lyases5.99 Other isomerases