Chemical Reactions and Enzymes. I. WHAT ARE ENZYMES? Your body has lots of reactions! Enzymes:...
-
Upload
aaron-bowron -
Category
Documents
-
view
218 -
download
4
Transcript of Chemical Reactions and Enzymes. I. WHAT ARE ENZYMES? Your body has lots of reactions! Enzymes:...

Chemical Reactions and Enzymes

I. What are enzymes?
Y
our body has lots of reactions!
E
nzymes: special protein catalysts that speed
up specific reactions
E
nd in ASE • Ex. Lactase digest lactose

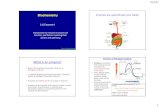
Activation Energy:The energy needed to get a reaction started

III. Lower the Activation Energy
• Activation energy is
the “start up” energy of a reaction
• They do not raise the temp!
• Increase the frequency that the reactants come together to react!

FreeEnergy
Progress of the reaction
Reactants
Products
Free energy of activationFree energy of activation
Without Enzyme
With Enzyme
EnzymesEnzymes

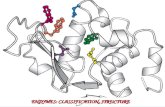
IV. Enzymes are Specific
The shape of each enzyme fits the
shape of what its acting on
SUBSTRATE:
the reactant that connects with
enzyme
ACTIVE SITE:
area where the substrate and
enzyme meet up


V. Lock and key model
L
ock is the enzyme
K
ey is the substrate
T
he lock hole is the
active site

Enzymes can only catalyze 1 reaction because they only
allow substrates with one particular shape to bond
oes not fit!

Factors that Affect the Enzyme Performance
Temperature
pH
Salinity
Concentration of enzyme or substrate

TemperatureE
nzymes are PROTEIN
H
eat too high they can denature

Enzymes work best at
an ‘optimal’ pH
Proteins they can
denature
pH

SalinityT
oo much salt will cause the enzyme
(protein) to denature, changing its shape
so it will
not function

Other factors
C
oncentration of enzyme and concentration of
substrate will affect how many reactions can
occur.
R
eactions depend on the limiting factor!

Enzymes are Reusable
Enzymes will speed the
reaction for a substrate
It will release the
product and will work on
another substrate

Examples L
ipase
P
rotease
L
actase
S
ucrase
A
mylase
L
ife is not possible without enzymes! !!!!!!!