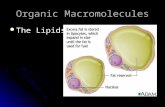
What are lipids? main component in every living cell. Organic have carbon chains with attached...
-
Upload
theodore-arnold -
Category
Documents
-
view
213 -
download
0
Transcript of What are lipids? main component in every living cell. Organic have carbon chains with attached...


What are lipids?What are lipids?
main component in main component in every living cell.every living cell.
OrganicOrganic have carbon chains have carbon chains
with attached with attached hydrogen atoms and hydrogen atoms and a a carboxyl groupcarboxyl group at at one end.one end.
FAT CELL

Made up of C, H, O, just like carbsMade up of C, H, O, just like carbs So what’s the So what’s the
difference?difference?
STRUCTURE!STRUCTURE!

Use of lipids in the bodyUse of lipids in the body
1. Energy- 2 x energy of Carbs1. Energy- 2 x energy of Carbs
2. Fat storage – energy for later 2. Fat storage – energy for later use use
3. Shock absorbers for the vital 3. Shock absorbers for the vital organsorgans

4. Satiety value – 4. Satiety value – makes us feel full makes us feel full longerlonger
5. converted to other 5. converted to other compounds- hormones, compounds- hormones, bile, Vitamin Dbile, Vitamin D
6. Insulates – a 6. Insulates – a fat blanket under fat blanket under the skinthe skin

Three categories of LipidsThree categories of Lipids
Triglycerides – fats & oilsTriglycerides – fats & oilsPhospholipids – emulsifiers, Phospholipids – emulsifiers,
component of cell component of cell membranemembrane
Sterols – bile, hormones, Sterols – bile, hormones, cholesterolcholesterol

TriglyceridesTriglycerides

Forms of TriglyceridesForms of Triglycerides
Animal sourcesAnimal sources Butter Butter LardLard
Eating different types Eating different types of triglycerides can of triglycerides can have different effects have different effects on the body.on the body.
Plant sourcesPlant sources MargarineMargarine Oils Oils shorteningshortening

Fat per dayFat per day 30 % of your calorie30 % of your calorie
intake or lessintake or less
9 calorie per gram9 calorie per gram

Women 19-49yrs 65 gms 16 tspWomen 19-49yrs 65 gms 16 tsp Men 19-49 90 gms 22 tspMen 19-49 90 gms 22 tsp 50+ 60 gms 15 50+ 60 gms 15
1 tsp = 4 gms fat 1 tsp = 4 gms fat

Fats and oils are Fats and oils are made from made from glycerol and fatty glycerol and fatty acids.acids.
Each glycerol is Each glycerol is attached to 3 fatty attached to 3 fatty acids.acids.
They contain They contain carbon , hydrogen carbon , hydrogen and oxygenand oxygen

Carboxyl group -COOHCarboxyl group -COOH

Each fatty acid consists of a hydrocarbon Each fatty acid consists of a hydrocarbon chain with a chain with a carboxyl groupcarboxyl group at one end. at one end.
The glycerol molecule has three The glycerol molecule has three hydroxyl groupshydroxyl groups (OH), each able to (OH), each able to interact with the carboxyl group of a fatty interact with the carboxyl group of a fatty acid.acid.


Essential Fatty Acids Essential Fatty Acids
Those the body Those the body must get through must get through food.food.
Can not make Can not make these in our these in our bodies.bodies.

Saturated fatsSaturated fats
Animal sourcesAnimal sources Fatty acids are Fatty acids are
saturated-hold all the saturated-hold all the hydrogen atoms they hydrogen atoms they can.can.
Solid at room Solid at room temperaturetemperature
We make our own.We make our own.


SourcesSources
Animal productsAnimal products

Unsaturated fatsUnsaturated fats
Most of the fatty acids are Most of the fatty acids are unsaturatedunsaturated
have one or more have one or more double double bondsbonds between their between their carbon atoms. carbon atoms.
2 carbon atoms missing a 2 carbon atoms missing a hydrogen bond join hydrogen bond join togethertogether
unsaturatedunsaturated because because they could hold more they could hold more hydrogen atoms than hydrogen atoms than they do.they do.

Monounsaturated - Monounsaturated - lacks 2 hydrogen- has lacks 2 hydrogen- has one double bond one double bond
Polyunsaturated- Polyunsaturated- lacks more than 2 lacks more than 2 hydrogen- 2 or more hydrogen- 2 or more double bondsdouble bonds

MonoMonounsaturated fatunsaturated fat

PolyPolyunsaturated fatunsaturated fat

This prevents the fatty acids This prevents the fatty acids from packing close together from packing close together and as a result, unsaturated and as a result, unsaturated fats have a lower melting point fats have a lower melting point than do saturated fats. (they are than do saturated fats. (they are healthier for you!)healthier for you!)

Trans fatty acidsTrans fatty acids
Unsaturated fatty acids - one of two shapes — "cis" and "trans."
refers to physical positioning of hydrogen atoms around carbon chain.
cis form is more common
found in small amounts in various animal products such as beef, pork, lamb and the butterfat in butter and milk.

Omega fatty acidsOmega fatty acids unsaturated fatty acids unsaturated fatty acids firstfirst double bond counting from double bond counting from
the opposite end from the the opposite end from the carboxyl group. carboxyl group.
terminal carbonterminal carbon atom (shown atom (shown here in blue) is called the here in blue) is called the omegaomega carbon atom. carbon atom.
monounsaturated fatty acid with monounsaturated fatty acid with its single double bond after its single double bond after carbon #3 (counting from and carbon #3 (counting from and including the omega carbon) is including the omega carbon) is called an omega-3 fatty acid. called an omega-3 fatty acid.
But so is a But so is a polyunsaturatedpolyunsaturated fatty acid, such as linolenic acid fatty acid, such as linolenic acid (shown here), if its first double (shown here), if its first double bond is in that position. bond is in that position.

Some studies have suggested that Some studies have suggested that omega-3 fatty acids help protect omega-3 fatty acids help protect against cardiovascular disease.against cardiovascular disease. This is known as This is known as Good FatGood Fat

Omega-6 Fatty acidsOmega-6 Fatty acids
Omega-6 fatty acids Omega-6 fatty acids belong to a group of belong to a group of "good" fats called "good" fats called polyunsaturated fatty polyunsaturated fatty acids. acids.
Help with immune Help with immune systemsystem
Relax Relax
Good dietary sourcesGood dietary sources of omega-3 fatty of omega-3 fatty acidsacids

Hydrogenated Vegetable oilHydrogenated Vegetable oil
In hydrogenation, plant oils are In hydrogenation, plant oils are exposed to hydrogen at a high exposed to hydrogen at a high temperature and in the temperature and in the presence of a catalyst, and presence of a catalyst, and turned from turned from liquid into solid!liquid into solid!

Occur when Occur when hydrogenation is done hydrogenation is done the unsaturated fats the unsaturated fats become saturatedbecome saturated
Hardened margarines Hardened margarines

GoodGood- Makes them resistant to oxidation- - Makes them resistant to oxidation- become rancid more slowlybecome rancid more slowly
GoodGood- makes them more stable- makes them more stable BadBad- makes them more saturated- makes them more saturated

Functions of Triglycerides in Food Functions of Triglycerides in Food Flavor – make foods Flavor – make foods
taste bettertaste better TenderizeTenderize EmulsifiersEmulsifiers Aeration – traps air in Aeration – traps air in
bubblesbubbles

RancidityRancidity
Oxidation of fatOxidation of fat Fatty acids combine with oxygen and lose Fatty acids combine with oxygen and lose
hydrogenhydrogen Unsaturated fats with more double bonds Unsaturated fats with more double bonds
are more prone to oxidationare more prone to oxidation heat, light and oxygen speeds up oxidationheat, light and oxygen speeds up oxidation

Rancid is the term Rancid is the term that describes the that describes the unpleasant flavors unpleasant flavors that develop as fats that develop as fats oxidizeoxidize
Different chemical Different chemical changes occur for changes occur for different types of fatsdifferent types of fats
Rancid nuts, meats Rancid nuts, meats and potato chips all and potato chips all taste differenttaste different

PhospholipidsPhospholipids Can dissolve in both fat Can dissolve in both fat
and waterand water Used as emulsifiersUsed as emulsifiers Found in eggs and Found in eggs and
peanutspeanuts Main component of cell Main component of cell
membranesmembranes


SterolsSterolsA fatty alcohol made from A fatty alcohol made from
glucose or saturated fatty glucose or saturated fatty acidsacids

strengthens cell membranestrengthens cell membraneprecursor of other steroids, precursor of other steroids,
including sex hormones.including sex hormones.Vital for producing Vitamin DVital for producing Vitamin D Used to make bileUsed to make bile

Your liver produces Your liver produces all the cholesterol all the cholesterol you need.you need.
Cholesterol in foods Cholesterol in foods is uselessis useless
It can make plaque It can make plaque which builds up on which builds up on the walls of blood the walls of blood vesselsvessels



LDL = low density lipoproteinLDL = low density lipoprotein(bad)(bad)
Carries cholesterol to Carries cholesterol to to the body tissues to to the body tissues to deposit theredeposit there
When elevated When elevated forecast heart and forecast heart and artery diseaseartery disease
Oxidation of LDL Oxidation of LDL causes damage to causes damage to arteriesarteries

AntioxidantsAntioxidants
Slows oxidation of Slows oxidation of LDLLDL
Vitamin C, Vitamin E, Vitamin C, Vitamin E, selenium are selenium are antioxidantsantioxidants

HDL = high density lipoproteinHDL = high density lipoprotein(good)(good)
HDL cholesterol- HDL cholesterol- carries cholesterol carries cholesterol away from tissueaway from tissue


Olestra- artificial fatOlestra- artificial fat
Remains undigestedRemains undigested Passes through the Passes through the
digestive tract intactdigestive tract intact Causes diahrea and Causes diahrea and
digestive upset digestive upset Does not allow Does not allow
vitamins to be vitamins to be absorbed by the bodyabsorbed by the body