Water and Aqueous Systems Chemistry Mrs. Coyle. The Water Molecule Bent Two lone electron pairs...
-
Upload
joy-pierce -
Category
Documents
-
view
213 -
download
0
Transcript of Water and Aqueous Systems Chemistry Mrs. Coyle. The Water Molecule Bent Two lone electron pairs...

Water and Aqueous Systems
ChemistryMrs. Coyle

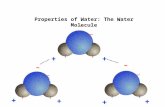
The Water Molecule Bent Two lone electron
pairs Polar molecule

Hydrogen Bonding:
The intermolecular forces in which hydrogen that is covalently bonded to a very electronegative atom is also weakly bonded to an unshared electron pair of another atom (N, O, F).


Hydrogen bonding is responsible for many of the unique properties of water such as:
high surface tension
having a low vapor pressure
having a lower density in the solid form than in the liquid form.

Surface tension causes water to form nearly spherical droplets.

http://www.chemistryland.com/CHM107/Water/WaterBeadsOnPlantPlusWaterCharge.jpg


Surface Tension
Surface tension is an inward force that causes the surface to behave as a skin.
The higher the intermolecular forces the greater the surface tension.

Surface Tension of Water
http://quest.nasa.gov/space/teachers/microgravity/image/66.gif

Water – Mercury

Surfactants:
Substances that interfere with the hydrogen bonding between molecules and reduce the surface tension.
Cause spreading or wetting. Examples of surfactants are soaps.

Water’s Low Vapor Pressure
Because of the hydrogen bonds holding the water molecules together, the molecules have a low tendency to break free from the surface into the vapor phase.

Water has a relatively high boiling point.
Why?

Density of Water
Ice has a lower density than liquid water. Maximum density of water happens at
4OC.

Density of Water



Ice- (Honeycomb Shape)