U.S. Food and Drug Administration, CDER, Silver Spring...
Transcript of U.S. Food and Drug Administration, CDER, Silver Spring...
Over-the-Counter (OTC) Topical Acne Drug Products and Hypersensitivity-Related Drug Events:
Post-market Data from the FDA Adverse Event Reporting System A. Ferguson, J. Swann , J. Tonning, T. Pham, P. Diak, A. Brinker, H. Mehta, S. Proestel†
U.S. Food and Drug Administration, CDER, Silver Spring, Maryland
† The authors of this poster have no financial interest to disclose.
¥ Acknowledgements: M. Chen, L. Governale, J. Mathew, G. Chai
TABLE 1. Demographic and Clinical Characteristics of the Serious Cases associated with Hypersensitivity Events for the
OTC topical Acne Products (N=131) FAERS search period: 1969 through 01-28-2013
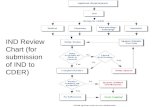
FIGURE 1.
Hypersensitivity-Specific Case Selection
BACKGROUND
We searched the FAERS database for reports of OTC topical acne products associated
with hypersensitivity reactions submitted from the database inception (1969) to January
28, 2013. To provide context for the adverse event reports submitted to the FAERS
database, the retail sales of these products were assessed using the OTC International
Market Tracking database.
Because of the clinical significance of anaphylaxis, each hypersensitivity case-report was
further evaluated to identify cases of anaphylaxis. Each case of anaphylaxis met the
following criteria: 1) Reporting of one of the following adverse events: anaphylaxis,
anaphylactoid reaction, anaphylactic shock, anaphylactoid shock, angioedema, or shock
OR 2) Involvement of skin or mucosal tissue, AND compromise of at least one of the
following organ systems: respiratory or cardiovascular. The remaining cases were
considered non-anaphylaxis in nature and classified as a non-anaphylaxis hypersensitivity
reaction.
METHODS
Benzoyl peroxide, salicylic acid, sulfur and resorcinol/sulfur are marketed, active
ingredients for over-the-counter (OTC) topical acne monograph products for the treatment
of acne vulgaris. Both single-ingredient and combination OTC topical acne products are
regulated under the OTC monograph in 21 CFR 333 and are considered GRAS/E
(Generally Recognized as Safe and Effective). In 2006, the Federal Food, Drug and
Cosmetic Act was amended, by Public Law 109-462, to require that all serious adverse
events be reported for dietary supplements and nonprescription OTC drugs marketed
without an approved application when used in the US. This analysis was initiated due to
the identification of an increasing number of hypersensitivity-related reports associated
with the use of OTC topical acne monograph products.
We assessed data from the FDA Adverse Event Reporting System (FAERS) to evaluate
the postmarket adverse event experience for topical OTC acne monograph products in
association with serious hypersensitivity reactions, with benzoyl peroxide, salicylic acid,
sulfur or resorcinol/sulfur as active ingredients.
PURPOSE
LIMITATIONS OF FAERS
Limitations of FAERS data include: underreporting, stimulated reporting, biased
reporting, variable quality and content, and no certainty that the reported event is due to
the product.
TABLE 1. Demographic and Clinical Characteristics of the Serious Cases associated
with Hypersensitivity
Events for the OTC topical Acne Products (N=131)
FAERS search period: 1969 through 01-28-2013
FAERS SEARCH RESULTS n = 141
EXCLUDED REPORTS (n= 19)
Reasons for exclusion:
Not a product of interest
Not used for acne
A result of accidental exposure
CASE SERIES
N = 131
Benzoyl Peroxide (n= 110)
Salicylic Acid (n= 21)
Resorcinol-Sulfur (n= 0)
Sulfur (n= 0)
DISCUSSION AND CONCLUSION
FDA postmarket data demonstrate that there may be an association of serious
hypersensitivity reactions, including anaphylaxis, with use of OTC topical acne
monograph products containing benzoyl peroxide and salicylic acid. While multiple
active ingredients are allowable for marketing, we only observed hypersensitivity adverse
events with benzoyl peroxide and salicylic acid. The reason for this observation is not
known; however high use associated with products containing these two active
ingredients may contribute to this observation (see Figure 2). In addition, based on the
information reported to FDA, we cannot determine if the hypersensitivity reactions were
triggered by the acne products’ active ingredients, benzoyl peroxide and salicylic acid, the
inactive ingredients, or by a combination of both.
Based on these findings, the FDA issued a drug safety communication in June 2014 to
alert the public regarding rare but serious hypersensitivity reactions with certain OTC
topical acne products. This warning alerts consumers to the following:
● To test the product upon first use, by applying a small amount topically to affected
area for three days; if no discomfort is experienced, then continued product use is
recommended per directions.
● To stop use with topical acne products and immediately seek medical attention if
they experience hypersensitivity reactions such as throat tightness, difficulty
breathing, feeling faint, swelling of the eyes, face, lips or tongue; develop hives or
itching.
● To avoid using an OTC topical acne product again if they have previously
experienced a hypersensitivity reaction with its use.
RESULTS
We identified 131 cases associated with OTC topical acne products (benzoyl peroxide,
n=110; salicylic acid, n=21). Reported adverse events ranged from application site reactions
to anaphylaxis. Of the 131 cases, 50 met our case definition for anaphylaxis. No fatalities
were identified; however, hospitalization was required in 44% of these cases. Four patients
reported a reappearance of their hypersensitivity reaction once the suspected product was
reintroduced (positive rechallenge). The OTC topical acne products cited in these cases
were marketed under various brand names such as Proactiv, Neutrogena, MaxClarity, Oxy,
Ambi, Aveeno, Clean & Clear, and store brands. These products are available as gels,
lotions, face washes, solutions, cleansing pads, toners, and face scrubs.
In 2012, approximately 15.7 million bottles/packages of OTC topical acne products were
sold from retail store outlets. Of these, the majority of OTC topical acne products contained
salicylic acid at approximately 61% (9.6 million bottles/packages). OTC topical acne
products containing benzoyl peroxide followed at 35% (5.4 million bottles/packages).
0.0
2.0
4.0
6.0
8.0
10.0
12.0
2007 2008 2009 2010 2011 2012
Num
ber o
f bot
tles/
pack
ages
(mill
ions
)
Year
Nationally estimated number of bottles/packages of over-the-counter topical acne products sold from U.S. retail store outlets, years 2007-2012
SALICYLIC ACID BENZOYL PEROXIDE SALICYLIC ACID/BENZOYL PEROXIDE (Acne Pack)
SULFUR RESORCINOL/SULFUR (Combo) RESORCINOL
SALICYLIC ACID/SULFUR (Combo)
Source: OTC International Market Tracking (OTCIMS). Years 2007-2012. Data extracted April 2013.
FIGURE 2. Nationally estimated number of bottles/packages of OTC topical acne products sold from U.S. retail store outlets, years
2007-2012
Selected Characteristics
Benzoyl
Peroxide
n=110
Salicylic Acid
n=21
Age (years)
Range
Mean
Median
n=80
11-78
32
27
n=16
14-55
30
28
Sex
Female
Male
93
17
20
1
Time to Onset (Days)
Immediate
15 minutes to < 24 hours
1 Day – 3 Days
4 Days - 30 Days
> 30 Days
n=76
15
31
8
18
4
n=14
0
9
4
1
0
Hypersensitivity
Classification1
Anaphylaxis
Non-Anaphylactic
Hypersensitivity
43
67
7
14