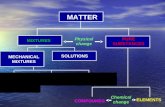
Section 3.3 Mixtures of Matter. Composition of Matter Matter SubstanceMixture.
Unit 1: Matter. Objective 1.01 Classify matter as elements, compounds, or mixtures Classify matter...
-
Upload
barnaby-baker -
Category
Documents
-
view
221 -
download
0
Transcript of Unit 1: Matter. Objective 1.01 Classify matter as elements, compounds, or mixtures Classify matter...

Unit 1: MatterUnit 1: Matter

Objective 1.01Objective 1.01
Classify matter as Classify matter as elements, elements, compounds, or compounds, or mixturesmixtures
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

What is matter?What is matter?
Two properties:Two properties:Has massHas massTakes up spaceTakes up space
Each substance is uniqueEach substance is unique
Chemistry – study of the Chemistry – study of the properties of matterproperties of matter

ElementsElements
Cannot be broken down into any Cannot be broken down into any other substanceother substance
All matter is composed of elementsAll matter is composed of elements
Symbol

CompoundsCompounds
Substance made of two or more Substance made of two or more elements chemically combinedelements chemically combined
Ex: NaClEx: NaCl
We use We use formulasformulas --combination --combination
of symbols to show whatof symbols to show what
elements make up aelements make up a
compoundcompound

MixturesMixtures
Made from:Made from:two or more substances (elements, two or more substances (elements,
compounds, mixtures)compounds, mixtures)They are together, but not They are together, but not
chemically combinedchemically combined

Diagrams of Elements, Diagrams of Elements, Compounds, and MixturesCompounds, and Mixtures

Objective 1.02Objective 1.02
Identify phases of Identify phases of matter by their matter by their propertiesproperties
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Definite Volume
Definite Shape
Definite Volume
Changes Shape
Changes Volume
Changes Shape

Changes in MatterChanges in Matter
Solid Solid Liquid Liquid
Liquid Liquid SolidSolid
Liquid Liquid Gas Gas
Gas Gas Liquid Liquid
Solid Solid Gas Gas

Changes in MatterChanges in Matter
Solid Solid Liquid……… Liquid………
Liquid Liquid Solid……….Solid……….
Liquid Liquid Gas………. Gas……….
Gas Gas Liquid………. Liquid……….
Solid Solid Gas………... Gas………...

Objective 1.03Objective 1.03
Use a phase diagram Use a phase diagram to determine phases to determine phases and phase changesand phase changes
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Phase DiagramPhase Diagram

Practice Problem #1Practice Problem #1
Draw a phase diagram where:Draw a phase diagram where: Solid from -15Solid from -15C to 10C to 10CC Liquid from 10Liquid from 10C to 130C to 130CC Gas from 130Gas from 130C and aboveC and above

Practice Problem #1Practice Problem #1

Objective 1.04Objective 1.04
Distinguish between Distinguish between physical and chemical physical and chemical properties and properties and changeschanges
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Physical vs. Chemical Physical vs. Chemical ChangesChanges
PhysicalPhysical Changes Changes
appearanceappearance Doesn’t convert Doesn’t convert
material into material into new substancesnew substances
Ex: chop woodEx: chop wood
bend wirebend wire
ChemicalChemical Produces new Produces new
substancesubstance ““Chemical Chemical
Reaction”Reaction” Ex: rustingEx: rusting
gas burninggas burning

Objective 1.05Objective 1.05
Identify the location, Identify the location, charge, and relative charge, and relative mass of the atomic mass of the atomic particlesparticles
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

AtomsAtoms
Smallest part of an elementSmallest part of an element
Characteristics:Characteristics:1.1. Can’t be broken into smaller Can’t be broken into smaller
piecespieces2.2. In an element, all atoms are In an element, all atoms are
alikealike3.3. Atoms of different elements are Atoms of different elements are
differentdifferent4.4. Atoms of two or more elements Atoms of two or more elements
can form compoundscan form compounds5.5. Atoms of each element have a Atoms of each element have a
unique massunique mass

Parts of an AtomParts of an Atom

Objective 1.06Objective 1.06
Identify elements on Identify elements on the periodic tablethe periodic table
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

The Periodic TableThe Periodic Table

Objective 1.07Objective 1.07
Identify and describe Identify and describe metals, nonmetals, metals, nonmetals, and metalloids on and metalloids on the periodic tablethe periodic table
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

The Periodic Table of the The Periodic Table of the ElementsElements
Arranges elements in terms of Arranges elements in terms of similar propertiessimilar propertiesElements in columns have same Elements in columns have same
characteristicscharacteristics
Parts:Parts:Groups / Families – Vertical Groups / Families – Vertical
columns(#1-18)columns(#1-18)Periods – Horizontal rows (#1-7)Periods – Horizontal rows (#1-7)

MetalsMetals
Location: Left of “Staircase”Location: Left of “Staircase”
Physical Properties:Physical Properties: HardnessHardness ShininessShininess Malleable – pounded into shapeMalleable – pounded into shape Ductile – pulled into long wireDuctile – pulled into long wire
Other Info:Other Info: Most metals are good conductorsMost metals are good conductors
Transmit heat and energy wellTransmit heat and energy well

NonmetalsNonmetals
Location: Right of “Staircase”Location: Right of “Staircase” Lack properties of metalsLack properties of metals
Most properties are opposites of metalsMost properties are opposites of metals NOT malleable, ductile, hard, or shinyNOT malleable, ductile, hard, or shiny
Families:Families: Carbon (Group 14)Carbon (Group 14) Nitrogen (Group 15)Nitrogen (Group 15) Oxygen (Group 16)Oxygen (Group 16) Halogen (Group 17)Halogen (Group 17) Noble Gases (Group 18)Noble Gases (Group 18) HydrogenHydrogen

MetalloidsMetalloids
Location: Along “Staircase”Location: Along “Staircase” Properties: Some Metal / Some Properties: Some Metal / Some
NonmetalNonmetal Seven elements:Seven elements:
Boron – BBoron – B Silicon – SiSilicon – Si Germanium – GeGermanium – Ge Arsenic – AsArsenic – As
Most are semiconductorsMost are semiconductors This means they carry electricity This means they carry electricity under some conditions onlyunder some conditions only
Antimony –SbAntimony –Sb Tellurium – TeTellurium – Te Astatine – AtAstatine – At

Objective 1.08Objective 1.08
Determine the Determine the number of atomic number of atomic particles of elements particles of elements on the periodic tableon the periodic table
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Structure of an AtomStructure of an Atom
Number of…Number of… Protons = atomic numberProtons = atomic number Electrons = number of protonsElectrons = number of protons Neutrons = atomic mass – atomic numberNeutrons = atomic mass – atomic number

But what about those But what about those numbers…numbers…
HHydrogen
1
1.0079

Number of pNumber of p++, n, e, n, e--
Element Protons Neutrons Electrons
Hydrogen
Potassium
Iodine
Gold
Fluorine

Objective 1.09Objective 1.09
Determine the Determine the valence electrons for valence electrons for any element on the any element on the periodic tableperiodic table
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Valence ElectronsValence Electrons Electrons farthest from the nucleusElectrons farthest from the nucleus

Objective 1.10Objective 1.10
Determine whether a Determine whether a pair of elements will pair of elements will bond ionically or bond ionically or covalentlycovalently
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Two types of bondsTwo types of bonds
IonicIonicBetween metal and nonmetalBetween metal and nonmetal
Ex: Sodium (Na) and Bromine (Br)Ex: Sodium (Na) and Bromine (Br)
CovalentCovalentBetween nonmetal and nonmetalBetween nonmetal and nonmetal
Ex: Nitrogen (N) and Sulfur (S)Ex: Nitrogen (N) and Sulfur (S)Between a metalloid and a nonmetalBetween a metalloid and a nonmetal
Ex: Silicon (Si) and Oxygen (O)Ex: Silicon (Si) and Oxygen (O)

Objective 1.11Objective 1.11
Identify the parts of Identify the parts of a chemical equation, a chemical equation, including reactants including reactants and productsand products
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Parts of an EquationParts of an Equation
REACTANTS PRODUCTS
The elements or compounds
we start with
“yields”
The elements or compounds
we end up with

Objective 1.12Objective 1.12
Identify the four Identify the four types of chemical types of chemical reactionsreactions
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Four types of reactionsFour types of reactions
1.1. SynthesisSynthesis Putting two elements togetherPutting two elements together
Ex: 2HEx: 2H22 + O + O22 2H 2H22OO
2.2. DecompositionDecomposition Breaking compound apart to get Breaking compound apart to get
two elementstwo elements Ex: 2HEx: 2H22O O 2H2H22 + O + O22

Four types of Four types of reactions, cont…reactions, cont…
3.3. Single ReplacementSingle Replacement Compound + Element Compound + Element different Compound + different Compound +
different Elementdifferent Element
Ex: 2NaCl + HEx: 2NaCl + H22 2HCl + 2Na 2HCl + 2Na
4. Double Replacement4. Double Replacement Two compounds Two compounds Two different compounds Two different compounds
Ex: CaFEx: CaF22 + Li + Li22SOSO44 CaSOCaSO44 + 2LiF + 2LiF

Objective 1.13Objective 1.13
Distinguish between Distinguish between solutions, solutions, suspensions, and suspensions, and colloidscolloids
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

SolutionsSolutions
A well-mixed mixtureA well-mixed mixture
Same properties throughoutSame properties throughout
Ex: Salt water, KoolaidEx: Salt water, Koolaid

SuspensionSuspension
Mixture in which the particles can be seen Mixture in which the particles can be seen and easily separatedand easily separated Usually by filtering or settlingUsually by filtering or settling Ex: Italian Salad DressingEx: Italian Salad Dressing
You can easily see the particles floatingin the salad dressing

ColloidColloid
A mixture with small undissolved particles A mixture with small undissolved particles that don’t settlethat don’t settle Larger particles than solutionsLarger particles than solutions Ex: mayonnaise, shaving cream, whipped Ex: mayonnaise, shaving cream, whipped
creamcream

Objective 1.14Objective 1.14
Identify the five Identify the five different types of different types of solutionssolutions
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Types of solutions…Types of solutions…
1.1. Dilute SolutionDilute SolutionLittle solute, lots of solventLittle solute, lots of solvent
2.2. Concentrated SolutionConcentrated SolutionMore solute, lots of solventMore solute, lots of solvent
3.3. Unsaturated SolutionUnsaturated SolutionNot as much solute as solvent can holdNot as much solute as solvent can hold

Types of solutions, Types of solutions, cont…cont…
4.4. Saturated SolutionSaturated SolutionAs much solute as solvent can holdAs much solute as solvent can hold
5. 5. Supersaturated SolutionSupersaturated Solution More solute than solvent should be able More solute than solvent should be able
to holdto hold

Objective 1.15Objective 1.15
Describe the solution Describe the solution process and factors process and factors that affect its ratethat affect its rate
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Factors affecting Factors affecting solubilitysolubility
TemperatureTemperature For solids: as temperature increases, For solids: as temperature increases,
solubility increasessolubility increases
PressurePressure For gases: as pressure increases, solubility For gases: as pressure increases, solubility
increasesincreases
Type of SolventType of Solvent Solvent affects how much solute can Solvent affects how much solute can
dissolvedissolve

Objective 1.16Objective 1.16
Identify acids and Identify acids and bases by their bases by their propertiesproperties
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Properties of AcidsProperties of Acids
1.1. Tastes sourTastes sour
2.2. Reacts with metalsReacts with metals
3.3. Turns blue litmus paper redTurns blue litmus paper red
“Reacts with metals”

Examples of acids…Examples of acids…
Fertilizer (Nitric Acid)Fertilizer (Nitric Acid)
Batteries (Sulfuric Acid)Batteries (Sulfuric Acid)
FoodFood Oranges (Ascorbic Acid)Oranges (Ascorbic Acid) Green, leafy vegetables (Folic Acid)Green, leafy vegetables (Folic Acid)
Body (Lactic Acid)Body (Lactic Acid)

Acid RainAcid Rain

Properties of BasesProperties of Bases
1.1. Tastes bitterTastes bitter
2.2. Feels slipperyFeels slippery
3.3. Turns red litmus paper blueTurns red litmus paper blue

Examples of bases…Examples of bases…
CleanersCleaners AmmoniaAmmonia WindexWindex LyeLye
Health productsHealth products Milk of MagnesiaMilk of Magnesia TumsTums
FoodFood Baking sodaBaking soda

Measuring Acids/BasesMeasuring Acids/Bases
We want to know the strength of acids and We want to know the strength of acids and basesbases
pH ScalepH Scale 0 –140 –14 Acids: 0-7…….0 is most acidicAcids: 0-7…….0 is most acidic Neutral: 7Neutral: 7 Bases 7-14……14 is most basicBases 7-14……14 is most basic

A closer look…A closer look…

Objective 1.18Objective 1.18
Determine the Determine the products of acid / products of acid / base reactionsbase reactions
PA Anchor: S8.C.1.1PA Anchor: S8.C.1.1

Acid / Base ReactionsAcid / Base Reactions
When you combine an acid with a base, you When you combine an acid with a base, you alwaysalways get: get:
Water + A SaltWater + A Salt
Ex: Ex: HH22O + KClO + KCl
HH22O + NaClO + NaCl