The porphyry copper system: hydrothermal alteration€¦ · Porphyry Cu (Au, Mo), epithermal, VMS,...
Transcript of The porphyry copper system: hydrothermal alteration€¦ · Porphyry Cu (Au, Mo), epithermal, VMS,...

9/29/20
Arribas & Hedenquist (2020) 1
Antonio ArribasThe University of Texas at El Paso
Ambientes genéticos de alteración argílica avanzada: relevancia en la exploración en sistemas pórfido-epitermal
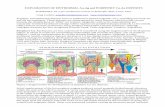
The Grand Canyon of the Yellowstone (~1895), by T. Moran
Epithermal deposits - Alteration is ever present
Hedenquist et al. (2000)
Arribas (1995)

9/29/20
Arribas & Hedenquist (2020) 2
The porphyry copper system: hydrothermal alteration
Sillitoe (2010)
… includes aluminosilicates (kaolinite, dickite, halloysite, pyrophyllite), plus alunite, and diaspore, andalusite, zunyite, and topaz”
Advanced argillic = advanced hydrolytic alterationHemley & Jones, 1964; Meyer & Hemley, 1967
Hedenquist et al. 1996
Al+3 Si+4SO4
-2K+
OH- F-Cl-PO4-3
Ca+2
H+HCO3-
Na+
Forms in acidic conditions: pH~2-<5 Residual quartz (vuggy silica): pH ~1
Shallow: typically <1 km
Epithermal deposits -Alteration is ever present

9/29/20
Arribas & Hedenquist (2020) 3
Five environments: simplified chemical reactions
2 KAl3Si3O10(OH)2 + 2 H+ + 6 SiO2 à 3 Al2Si4O10(OH)2 + 2 K+
muscovite quartz pyrophyllite
4 SO2 + 4 H2O à 3 H2SO4 + H2S
H2SO4 à H+ + HSO4- HCl à H+ + Cl-
alunite, kaolinite, dickite,
pyrophyllite, diaspore, zunyite,
residual vuggy silica, etc.
CO2 + H2O à H2CO3 kaolinite (illite-smectite, Fe carbonates, pyrite)
H2S + O2 à H2SO4 kaolinite, alunite, native S, opal
Fe sulfide + H2O + 02 à Fe oxy-hydroxides + H2SO4kaolinite, halloysite,
alunite, jarosite
Minerals formed in acid aqueous (alteration) environments
Same 5 environments of acid alteration minerals
Cooling of a magmatic-hydrothermal fluid
Hypogene acid sulfate-chloride (SO2 + H2O, HCl, HF)
Steam-heated CO2-rich (CO2 + H2O)
Steam-heated acid-sulfate (H2S + O2)
Supergene oxidation of pyrite (FeS2 + O2)
320-260°
300-200°
170-120°
120-70°
<40°C(approx.)

9/29/20
Arribas & Hedenquist (2020) 4
Five environments with ‘advanced argillic’ minerals
Modified from Sillitoe (2010)
Subepithermal
Porphyry
IS Epithermal
Sed-hosted disseminated
SkarnCarbonate replacement
Au-Cu HS Epithermal
HS Lode
1. Cooling of a magmatic-hydrothermal fluid
(white mica to pyrophyllite)
2. Hypogene acid sulfate-chloride
3. Steam-heated acid sulfate 4. Steam-heated
CO2-rich
5. Supergene oxidation of pyrite
<40°C(approx.)
70-120°120-170°200-300°260-300°
Forms: Above a porphyry system, cooling of white mica-stable fluid
Simplified reaction:2 KAl3Si3O10(OH)2 + 2 H+ + 6 SiO2
à 3 Al2Si4O10(OH)2 + 2 K+
2 muscovite + 6 quartz à 3 pyrophyllite
1. Cooling of white mica to pyrophyllite
Typical minerals:White mica (muscovite), pyrophyllite, diaspore (dickite) (nodular – patchy – replacement texture if in lithocap)
white mica
Porphyry intrusion
~1 k
m
à
pyrophyllite
white mica

9/29/20
Arribas & Hedenquist (2020) 5
Damiana exotica
El Salvador porphyry Cu, Chile Looking east, late 1950s
Muscovite
Pyrophyllite
Watanabe & Hedenquist, 2001
Cu ore at 2600 m

9/29/20
Arribas & Hedenquist (2020) 6
1
2
2) Hypogene vapor condensation Lithocap environment,
residual qtz, alunite halo, hotter pyrophyllite (roots)
Hemley & Jones, 1964; Watanabe & Hedenquist, 2001
pyrophyllite
muscovite± andalusite
Two environments of hypogene pyrophyllite:
2. Vapor condensation
El Salvador porphyry Cu deposit
K-Al-Si-H-O
ßlower pH
1) Fluid cooling (retrograde)Muscovite (A) to pyrophyllite (B) to dickite (C)
1. Cooling
Forms: Adjacent to volcanic vents, discharge of high-temperature magmatic vapors
Simplified reactions:4 SO2 + 4 H2Oà 3 H2SO4 + H2S,H2SO4 à H+ + HSO4
-
HCl à H+ + Cl-Cooling and dissociation leads to increased reactivity (<250–300oC)
2. Hypogene magmatic vapor condensation
Typical minerals: Core of residual (vuggy) quartz
(potential ore host)Alunite-kaolinite-dickite halo
(pyrophyllite in feeder zone)
~1 k
m
AluniteDickite- Kaolinite
Pyrophyllite
H2SO4 , HCl
White mica
K-silicate
H2O, CO2, SO2, H2S, HCl, metals
⤳ ⤳Hedenquist & Taran (2013)

9/29/20
Arribas & Hedenquist (2020) 7
Hemley et al., 1969, Stoffregen, 1987
KA
l 3(S
O4)
2(O
H),
Alu
nitepH
~1, AlO
H soluble;
residual quartz
Residual quartz (vuggy)
Res.Qtz Alun Kao Mica Clay Chl
Steven & Ratté, 1960
~1 2 to 4 4 to 6 >6 pH
ß lower pH
Tabular, platy alunite
Martabe, Indonesia
Arribas et al., 2000

9/29/20
Arribas & Hedenquist (2020) 8
Arribas et al., 2000
Forms: Within the vadose zone, above water table, by oxidation of H2S (pH = 2-3, ~70-120 °C, up to 150 °C)
Simplified reaction:H2S+ O2 à H2SO4(bacterial involvement)
3. Steam-heated, H2S oxidation
Typical minerals: Kaolinite, alunite, opal, cristobalite, native S, montmorillonite, jarosite
Sinter Terrace
Hot Springs
Hedenquist et al. (2000)

9/29/20
Arribas & Hedenquist (2020) 9
Typical mud-pools (Goshogake Geothermal Area, Hachimantai, Akita)
4. Steam-heated, H2S oxidation
4. Steam-heated, H2S oxidationLa Coipa, Chile, mining district, to west from 4700 m elevation
1 km

9/29/20
Arribas & Hedenquist (2020) 10
4. Steam-heated, H2S oxidationLa Coipa, Chile, mining district, to west from 4700 m elevation
Coipa Norte: Steam-heated alunite-kaolin blanket, chalcedony, over residual quartz, Ag-Au ore
vuggy silica Ag-Au ore
chalcedony blanket
4. Steam-heated, H2S oxidation
La Coipa, Chile, mining district: Puren open pit

9/29/20
Arribas & Hedenquist (2020) 11
The old Sulphur district (Nevada) looking north: S, Hg, clay mined since the late 1800s
4. Steam-heated, H2S oxidation
Native sulfur and cinnabar
Photo: mindat.org (R.Luetcke, 1978) Photo: JWH (~1982)
Silica sinter
Steam-heated acid-sulfate alteration blanket (overprint due to falling water table)
H2S + 2 O2 = H2SO4 (oxidation in vadose zone)
Qtz vein, ad, cc, Au
massive opal horizon - deeper FeS2
SiO2, Fe+3
4. Steam-heated, H2S oxidation
Crofoot-Lewis. South pit

9/29/20
Arribas & Hedenquist (2020) 12
Forms: Below water table where vapor + CO2 & H2S condense (on margins)
Simplified reaction:CO2 + H2O à H2CO3
4. Steam-heated: vapor + CO2 condensation
Typical minerals: Smectite, illite-smectite, illite, kaolinite, pyrite, Fe carbonates
carbonic acid (beer pH ~4-4.5)
CO2-rich
SO4-rich
Taupo Volcanic Zone, New Zealand: rift behind an arc Looking northeast: volcanic rift to west, volcanic arc to east
Waiotapu geothermal system
rhyolite domeandesite cone
basalt dike
Broadlands geothermal system
Ohaaki pool, neutral pH,
sinter
rift margin

9/29/20
Arribas & Hedenquist (2020) 13
Extent of kaolinite
Extent of siderite
Acid-sulfate kaolin pool,
pH~2-3
Hedenquist, 1990; Simmons and Browne, 2000 Browne, 1971
CO2-rich froth,BR-6
Ohaaki Pool, neutral pH Broadlands,
NZ
Deep upflow
Boiling,
vapor loss
smectite, I/S, kaolinite, Fe
carbonate: CO2-rich vapor condensate, pH 4-5
CO2-rich condensate
Forms: Above water table by supergene oxidation of pyritic rocks
Simplified reaction:Fe sulfide + H2O + 02 =Fe oxy-hydroxides + H2SO4
5. Supergene weathering, sulfide oxidation
Sillitoe (1993)
Assemblage: Kaolinite, halloysite, alunite, jarosite, hydrous Fe oxides
tubular halloysite
rhombohedral alunite
Riaza kaolinite mine (Spain)
rhombohedral alunitewith nearly cubic angles
Rodalquilar Au deposit (Spain)

9/29/20
Arribas & Hedenquist (2020) 14
5. Supergene weathering, sulfide oxidation
Associated deposits:Porphyry Cu (Au, Mo), epithermal, VMS, etc.
Typical occurrence:Irregular veinlets, nodular masses, local pervasive replacement
Texture and Color:Chalky, porcelaneousmasses and fine powdery aggregates; white, cream,yellow-brown (jarosite)
Rodalquilar Miocene epithermal Au (Spain)
Supergene alunite (~4Ma)
5. Supergene weathering, sulfide oxidationRiaza kaolinite mine (Spain)Silurian (~430 Ma) pyrite-bearing black shale
Kaolinite-alunite formed during Miocene (13.5 Ma) weathering
Typical occurrence:Irregular veinlets, nodular masses, local pervasive replacement
Texture and Color:Chalky, porcelaneousmasses and fine powdery aggregates; white, cream,yellow-brown (jarosite)
Assemblage: Kaolinite, halloysite, alunite, jarosite, hydrous Fe oxides

9/29/20
Arribas & Hedenquist (2020) 15
5. Supergene weathering, sulfide oxidation
Features of interest
Refractory sulfides are oxidized:
- Au can be recovered by heap leaching
- Cu by SX-EW (secondary Cu minerals)
Copper Alliance 2018
320-260°
300-200°
170-120°
120-70°
5 environments of acid alteration minerals
Cooling of a magmatic-hydrothermal fluid White mica to pyrophyllite ± diaspore transition
Hypogene acid-sulfate (SO2 + H2O) Residual quartz (±Au); alunite-kaolinite halo, pyrophyllite±dickite proximal to feeder zone
Steam-heated CO2-rich (CO2 + H2O)Marginal (deep) illite/smectite-kaolinite-Fe carbonate
Steam-heated acid sulfate (H2S + O2)Blanket of kaolinite-alunite, no metals (except Hg)
Supergene oxidation of pyrite (FeS2 + O2)Horizon of kaolinite-alunite-Fe hydroxides, no pyrite<40°C
(approx.)

9/29/20
Arribas & Hedenquist (2020) 16
• Distinguish environment of formation, based on mineral assemblage, textures, morphology
• Map: integrate with lithology, structure, etc. • Interpret location relative to mineralization potential
e.g., lithocap vs quartz veins; erosion level; assessment of metal anomalies, etc.
• Determine mineralogy relevant to exploration and each stage of assessment
• Focus on mineralogy, and avoid names“Horizon of kaolinite – alunite – Fe hydroxides, no pyrite”
• Do not use “advanced argillic”, ”intermediate argillic”, etc.
Practical recommendations
Steam-heated alteration over Pascua-Lama epithermal HS Au-Ag deposit, Chile
BarrickMod. Chouinard et al. (2005)

9/29/20
Arribas & Hedenquist (2020) 17
Yellowstone River valley alteration/mineralization?