The ATOM Discovery of the Electron In 1897, J.J. Thomson used a cathode ray tube to deduce the...
-
Upload
isabella-byrd -
Category
Documents
-
view
232 -
download
1
Transcript of The ATOM Discovery of the Electron In 1897, J.J. Thomson used a cathode ray tube to deduce the...

The ATOMThe ATOM


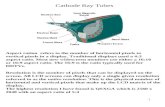
Discovery of the ElectronDiscovery of the ElectronIn 1897, J.J. Thomson used a cathode ray tube to deduce the presence of a negatively charged particle.
Cathode ray tubes pass electricity through a gas that is contained at a very low pressure.

Thomson’s Atomic Thomson’s Atomic ModelModel
Thomson believed that the electrons were like plums embedded in a positively charged “pudding,” thus it was called the “plum pudding” model.

Rutherford’s Gold Foil Rutherford’s Gold Foil ExperimentExperiment
Alpha particles are helium nuclei Particles were fired at a thin sheet of gold foil Particle hits on the detecting screen (film) are recorded



The Great The Great Niels Bohr (1885 - 1962)

…produces all of the colors in a continuous spectrum
Spectroscopic analysis of the visible Spectroscopic analysis of the visible spectrum…spectrum…

…produces a “bright line” spectrum
Spectroscopic analysis of the Spectroscopic analysis of the hydrogen spectrum…hydrogen spectrum…

This produces bands of light with definite wavelengths.
Electron Electron transitionstransitionsinvolve jumps of involve jumps of definite amounts definite amounts ofofenergy.energy.

Bohr Model Energy Levels

Types of electromagnetic radiation:Types of electromagnetic radiation:

c = C = speed of light, a constant (3.00 x 108 m/s)
= frequency, in units of hertz (hz, sec-1)
= wavelength, in meters
Electromagnetic radiation propagates Electromagnetic radiation propagates through space as a wave moving at the through space as a wave moving at the speed of light.speed of light.

TRY PROBLEM 39

Prepare yourself toPrepare yourself to
C

E = h EE = Energy, in units of Joules= Energy, in units of Joules
hh = Planck’s constant (6.626 x 10-34 J·s)= Planck’s constant (6.626 x 10-34 J·s)
= frequency, in units of hertz (hz, sec= frequency, in units of hertz (hz, sec-1-1))
The energy (The energy (E E ) of electromagnetic ) of electromagnetic radiation is directly proportional to radiation is directly proportional to the frequency (the frequency () of the radiation.) of the radiation.

Long Wavelength
= Low Frequency
= Low ENERGY
Short Wavelength
= High Frequency
= High ENERGY
Wavelength TableWavelength Table

Relating Frequency, Relating Frequency, Wavelength and EnergyWavelength and Energy
c hE
hc
E
Common re-arrangements:
E
hc

TRY PROBLEM 51

Types of electromagnetic radiation:Types of electromagnetic radiation:

PLANCK’S PRACTICE PROBLEMS 1.When we see light from a neon sign, we are observing radiation from excited neon atoms. If this radiation has a wavelength of 0.640 µm (micrometer or micron), what is the energy of the photons being emitted?
2. A photon of light produced by a surgical laser has an energy of 3.027 x 10 -22 kJ. Calculate the frequency and the wavelength of the photon.


Schrodinger Wave EquationSchrodinger Wave Equation
22
2 2
8dh EV
m dx
Equation for probabilityprobability of a single electron being found along a single axis (x-axis)Erwin Schrodinger

Heisenberg Uncertainty Heisenberg Uncertainty PrinciplePrinciple
You can find out where the electron is, but not where it is going.
OR…
You can find out where the electron is going, but not where it is!
“One cannot simultaneously determine both the position and momentum of an electron.”
Werner Heisenberg

Quantum NumbersQuantum Numbers
Each electron in an atom has a unique set of 4 quantum numbers which describe it.
Principal quantum number Angular momentum quantum number Magnetic quantum number Spin quantum number
(n)(l)
(m)(s)

Principal Quantum NumberPrincipal Quantum NumberGenerally symbolized by n, it denotes the shell (energy level) in which the electron is located.
The principal quantum number (n) cannot be zero. n must be 1, 2, 3, etc. Number of electrons that can fit in a shell:
2n2

Angular Momentum Quantum Angular Momentum Quantum NumberNumber
The angular momentum quantum number, generally symbolized by l, denotes the orbital (subshell) in which the electron is located. The angular momentum quantum number (l ) can be any integer between 0 and n - 1.
l =3f

Orbital shapes are defined as the surface that contains 90% of the total electron probability.
An orbital is a region within an atom where thereAn orbital is a region within an atom where thereis a probability of finding an electron. This is a is a probability of finding an electron. This is a probability diagram for the s orbital in the probability diagram for the s orbital in the first first energy level…energy level…

Magnetic Quantum NumberMagnetic Quantum NumberThe magnetic quantum number, generally symbolized by m, denotes the orientation of the electron’s orbital with respect to the three axes in space. The magnetic quantum number (ml) can be any integer between -l and +l.

Pauli Exclusion PrinciplePauli Exclusion Principle
No two electrons in an atom can have the same four quantum numbers.
Wolfgang Pauli

Spin Quantum NumberSpin Quantum NumberSpin quantum number denotes the behavior (direction of spin) of an electron within a magnetic field.
Possibilities for electron spin:
1
2
1
2

Assigning the NumbersAssigning the Numbers The three quantum numbers (n, l, and m) are integers. The principal quantum number (n) cannot be zero. n must be 1, 2, 3, etc. The angular momentum quantum number (l ) can be any integer between 0 and n - 1. For n = 3, l can be either 0, 1, or 2. The magnetic quantum number (ml) can be any integer between -l and +l. For l = 2, m can be either -2, -1, 0, +1, +2.

Principle, angular momentum, and Principle, angular momentum, and magnetic quantum numbers: magnetic quantum numbers: nn, , ll, and , and mmll

Aufbau

IN BOHR’S ATOMIC THEORY, WHEN AN ELECTRON MOVES FROM ONE ENERGY LEVEL TO ANOTHER ENERGY LEVEL MORE DISTANT FROM THE NUCLEUS.
1. energy is emitted
2. energy is absorbed
3. no change in energy occurs
4. light is emitted
5. none of these
36%
45%
0%
14%
5%
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24

WHICH FORM OF ELECTROMAGNETIC RADIATION HAS THE LONGEST WAVELENGTHS?
1. gamma rays
2. microwaves
3. radio waves
4. infrared radiation
5. x-rays
gamma ra
ys
micr
owaves
radio w
aves
infra
red ra
dia...
x-ray
s
17%
0%
9%4%
70%
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24

HOW MANY ELECTRONS IN AN ATOM CAN HAVE THE QUANTUM NUMBERS N = 3, L = 2?
1. 2
2. 5
3. 10
4. 18
5. 62 5 10 18 6
17%13%
4%4%
61%
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24

WHICH OF THE FOLLOWING COMBINATIONS OF QUANTUM NUMBERS IS NOT ALLOWED?
n l m s
1. 1 1 0 ½
2. 3 0 0 –½
3. 2 1 –1 ½
4. 4 3 –2 –½
5. 4 2 0 ½
41%
36%
0%
5%
14%
5%
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24

WHICH OF THE FOLLOWING FREQUENCIES CORRESPONDS TO LIGHT WITH THE LONGEST WAVELENGTH?
1. 3.00 x 1013 s–1
2. 4.12 x 105 s–1
3. 8.50 x 1020 s–1
4. 9.12 x 1012 s–1
5. 3.20 x 109 s–1
14%
45%
0%
5%5%
32%
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24

THE ELECTRON CONFIGURATION OF INDIUM IS
1. 1s22s22p63s23p64s23d104p65s24d105p15d10
2. 1s22s22p63s23p64s23d104d104p1
3. 1s23s22p63s23p64s24d104p65s25d105p1
4. 1s22s22p63s23p64s23d104p65s24d105p1
5. none of these
1s22s2
2p63s23p...
1s22s2
2p63s23p...
1s23s2
2p63s23p...
1s22s2
2p63s23p...
none of these
0%9%
4%
87%
0%
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
21 22 23 24

Orbital filling tableOrbital filling table

Element Configuration notation
Orbital notation Noble gas notation
Lithium 1s22s1 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s1
Beryllium 1s22s2 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s2
Boron 1s22s2p1 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s2p1
Carbon 1s22s2p2 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s2p2
Nitrogen 1s22s2p3 ____ ____ ____ ____ ____
1s 2s 2p
[He]2s2p3
Oxygen 1s22s2p4 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s2p4
Fluorine 1s22s2p5 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s2p5
Neon 1s22s2p6 ____ ____ ____ ____ ____ 1s 2s 2p
[He]2s2p6

The s orbital has a spherical shape centered around the origin of the three axes in space.
s orbital shape

There are three peanut-shaped p orbitals in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.
PP orbital shape orbital shape

Things get a bit more complicated with the five d orbitals that are found in the d sublevels beginning with n = 3. To remember the shapes, think of:
…and a “peanut with a donut”!
d orbital shapes
“double peanut”

Shape of f orbitalsShape of f orbitals
Things get even more complicated with the seven f orbitals that are found in the f sublevels beginning with n = 4. To remember the shapes, think of:
Flower

Electron configuration of the Electron configuration of the elements of the first three elements of the first three
seriesseries

PES

PES• method that provides information on all the occupied
energy levels of an atom (that is, the ionization energies of all electrons in the atom) is known as photoelectron spectroscopy; this method uses a photon (a packet of light energy) to knock an electron out of an atom.

PHOTOELECTRON SPECTRUMThe photoelectron spectrum is a plot of the number of electrons emitted versus their kinetic energy. In the diagram below, the “X” axis is labeled high to low energies so that you think about the XY intersect as being the nucleus.
http://www.chem.arizona.edu/chemt/Flash/photoelectron.html


Interpretations from the data:1. There are no values on the y axis in the tables above. Using the Periodic Table and Table 1, put numbers on the y axis.2. Label each peak on the graphs above with s, p, d, or f to indicate the suborbital they represent..3. What is the total number of electrons in a neutral potassium atom?
Orbital names s, p, d, and f stand for names given to groups of lines in the spectra of the alkali metals. Early chemists called the line groups sharp, principal, diffuse, and fundamental.
1-2-
6-
1s 2s 3s4s
2p 3p

PES QUESTION
• If a certain element being studied by an X-ray PES displays an emission spectrum with 5 distinct kinetic energies. What are all the possible elements that could produce this spectrum?
• Determine the orbitals that the spectral lines are originating from and then determine the elements that have electrons in only these orbitals.

Periodicity

ATOMIC SIZE
Atomic Radius = half the distance between two nuclei of a diatomic molecule.}
Radius

TRENDS IN ATOMIC SIZE
• Influenced by three factors.• Energy Level
• Higher energy level is further away.• Charge on nucleus
• More charge pulls electrons in closer.• Shielding
• Layers of electrons shield from nuclear pull.

SHIELDING
• The electron on the outside energy level has to look through all the other energy levels to see the nucleus

SHIELDING
• The electron on the outside energy level has to look through all the other energy levels to see the nucleus.
• A second electron has the same shielding.

GROUP TRENDS
• As we go down a group
• Each atom has another energy level,
• So the atoms get bigger.
HLi
Na
K
Rb

PERIODIC TRENDS
• As you go across a period the radius gets smaller.
• Same energy level.
• More nuclear charge.
• Outermost electrons are closer.
Na Mg Al Si P S Cl Ar

Table of Table of
Atomic Atomic RadiiRadii

IONIC SIZE
• Cations form by losing electrons.
• Cations are smaller that the atom they come from.
• Metals form cations.
• Cations of representative elements have noble gas configuration.

IONIC SIZE
• Anions form by gaining electrons.
• Anions are bigger that the atom they come from.
• Nonmetals form anions.
• Anions of representative elements have noble gas configuration.


OVERALL
Atomic Number
Ato
mic
Radiu
s (n
m)
H
Li
Ne
Ar
10
Na
K
Kr
Rb

DO PROBLEM 88

IONIZATION ENERGY
• The amount of energy required to completely remove an electron from a gaseous atom.
• Removing one electron makes a +1 ion.
• The energy required is called the first ionization energy.

IONIZATION ENERGY
• The second ionization energy is the energy required to remove the second electron.
• Always greater than first IE.
• The third IE is the energy required to remove a third electron.

Symbol First Second ThirdH
HeLiBeBCNOF Ne
1312 2731 520 900 800 1086 1402 1314 1681 2080
5247 7297 1757 2430 2352 2857 3391 3375 3963
11810 14840 3569 4619 4577 5301 6045 6276

WHAT DETERMINES IE
• The greater the nuclear charge the greater IE.
• Distance from nucleus increases IE
• Filled and half filled orbitals have lower energy, so achieving them is easier, lower IE.
• Shielding

GROUP TRENDS
• As you go down a group first IE decreases because
• The electron is further away.• More shielding.

PERIODIC TRENDS
• All the atoms in the same period have the same energy level.
• Same shielding.
• Increasing nuclear charge
• So IE generally increases from left to right.
• Exceptions at full and 1/2 fill orbitals.

Firs
t Io
niz
ati
on e
nerg
y
Atomic number
H
He
Li
Be
B
C
N
O
F
Ne Na has a lower
IE than Li Both are s1
Na has more shielding
Greater distance
Na

Affinity tends to increase across a period
Affinity tends to decrease as you go down in a group
Electrons farther from the nucleus experience less nuclear attraction
Some irregularities due to repulsive forces in the relatively small p orbitals
Electron AffinityElectron Affinity - the energy change - the energy change associated with the addition of an electronassociated with the addition of an electron

DO PROBLEM 89

ELECTRONEGATIVITY
• The tendency for an atom to attract electrons to itself when it is chemically combined with another element.
• How fair it shares.
• Big electronegativity means it pulls the electron toward it.
• Atoms with large negative electron affinity have larger electronegativity.

GROUP TREND
• The further down a group the farther the electron is away and the more electrons an atom has.
• More willing to share.
• Low electronegativity.

PERIODIC TREND
• Metals are at the left end.
• They let their electrons go easily
• Low electronegativity
• At the right end are the nonmetals.
• They want more electrons.
• Try to take them away.
• High electronegativity.

Ionization energy, electronegativityElectron affinity INCREASE

Atomic size increases, shielding constant
Ionic size increases

Another Way to Look at Ionization Energy


Summary of Periodic Summary of Periodic TrendsTrends

Put the following in order of Put the following in order of Decreasing atomic radius:Decreasing atomic radius:
a)a)Cl,Ar,KCl,Ar,Kb)b)b) O, Ob) O, O--, O, O2-2-
c) Co, Rh, Nic) Co, Rh, Ni
Now put them in order of Now put them in order of Decreasing ionization energy:Decreasing ionization energy: