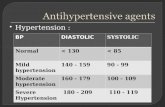
Management of Severe Hypertension, Hypertension in Special Condition
Sponsored by a grant from The Medicines Company to the Center for Outcomes Research, University of...
-
Upload
louisa-blankenship -
Category
Documents
-
view
216 -
download
3
Transcript of Sponsored by a grant from The Medicines Company to the Center for Outcomes Research, University of...

Sponsored by a grant from The Medicines Company to the Center for Outcomes Research, University of Massachusetts Medical School
Acute Severe Hypertension has Acute Severe Hypertension has High Morbidity, Mortality, High Morbidity, Mortality, and Readmission Rates: and Readmission Rates:
Results from the STAT Registry Results from the STAT Registry
CB Granger, J Gore, J Katz, K Kleinschmidt, A Wyman, F Peacock, F Anderson, on behalf of the STAT Investigators. Presented at the European Society of Cardiology Annual Meeting, September 2, 2008.

2www.outcomes.org/stat
STAT Steering CommitteeSTAT Steering Committee
Albert Einstein College of MedicineAlbert Einstein College of MedicineAlan S MultzAlan S Multz
Cleveland ClinicCleveland ClinicW Frank PeacockW Frank Peacock
Columbia UniversityColumbia UniversityStephan A MayerStephan A Mayer
Duke University Duke University Christopher Granger*Christopher Granger*Eric PetersonEric Peterson
Ohio State UniversityOhio State UniversityJoseph F DastaJoseph F Dasta
Uniformed Services UniversityUniformed Services UniversityAndrew F ShorrAndrew F Shorr
University of California University of California Alpesh Amin Alpesh Amin
University of MassachusettsUniversity of MassachusettsFred AndersonFred AndersonJoel GoreJoel Gore
University of PennsylvaniaUniversity of PennsylvaniaCharles PollackCharles Pollack
University of Southern CaliforniaUniversity of Southern CaliforniaGene Yong SungGene Yong Sung
University of Texas at HoustonUniversity of Texas at HoustonJames FergusonJames FergusonJoseph VaronJoseph Varon
University of Texas SouthwesternUniversity of Texas SouthwesternKurt KleinschmidtKurt Kleinschmidt
Project and data coordinated by and Project and data coordinated by and analyzed by Center for Outcomes analyzed by Center for Outcomes Research, University of Massachusetts, Research, University of Massachusetts, Worcester, MAWorcester, MA
*Chair

3www.outcomes.org/stat
Participating Investigators in STAT Participating Investigators in STAT (25 US Centers)(25 US Centers)
Cleveland, OH
Worcester MA
Dallas, TX
New York, NYPhiladelphia, PA
Houston, TX
Durham, NC
Sacramento, CA
Columbus, OH
Miami, FL
Boston, MA
Detroit, MI (3)Detroit, MI (3)
New Orleans, LA
Chandler, AZ
Royal Oak, MIRoyal Oak, MI Stony Brook, NY
Charleston, SC
Winston-Salem, NC
Ann Arbor, MIAnn Arbor, MI
Joseph VaronPhil LevyRichard NowakJohn DevlinJim FroehlichBrian O'NeilMarc LapointKurt KleinschmidtW Frank PeacockJoel M GoreCharles PollackJon Schrock
Lala DunbarJohn CienkiStephan S MayerAnthony GerlachDeborah DiercksAdam SingerGene Yong SungJason KatzNamarata PatilDavid ClineIcima FergusBrian Tiffany

4www.outcomes.org/stat
BackgroundBackground
Acute severe hypertension is common – Acute severe hypertension is common – occurring in 1-2% of the 72 million US patients occurring in 1-2% of the 72 million US patients with hypertension, and comprising up to 25% of with hypertension, and comprising up to 25% of visits in busy urban emergency departmentsvisits in busy urban emergency departments
Contemporary patient features, management Contemporary patient features, management and outcomes are not well knownand outcomes are not well known
Traditional definitions of who needs intensive Traditional definitions of who needs intensive therapy have been based on target organ therapy have been based on target organ dysfunctiondysfunction

5www.outcomes.org/stat
GoalGoal
Improve understanding of clinical Improve understanding of clinical
conditions of acute severe hypertension conditions of acute severe hypertension
managed in a critical care setting and managed in a critical care setting and
treated with IV antihypertensive drugstreated with IV antihypertensive drugs

6www.outcomes.org/stat
Inclusion CriteriaInclusion Criteria
>18 years of age>18 years of age Presenting to the hospital with acute Presenting to the hospital with acute
severe HTNsevere HTN Treated in a critical care settingTreated in a critical care setting Acute severe HTN treated with an IV agentAcute severe HTN treated with an IV agent Severe hypertensionSevere hypertension
– At least one SBP >180 mmHg and/orAt least one SBP >180 mmHg and/or– At least one DBP >110 mmHgAt least one DBP >110 mmHg
SAH patients with SBP >140 and/or DBP >90SAH patients with SBP >140 and/or DBP >90

7www.outcomes.org/stat
Study PopulationStudy Population
PatientsPatients 1,588 1,588
Age - median (IQR) Age - median (IQR) 58 (49, 70)58 (49, 70)
Female sexFemale sex 49% 49%
Black raceBlack race 56% 56%
White raceWhite race 34% 34%
Qualifying BP (mmHg)Qualifying BP (mmHg)
SystolicSystolic 200 (186, 220)200 (186, 220)
DiastolicDiastolic 110 (93, 123)110 (93, 123)
LOS - median (IQR) LOS - median (IQR) 5 days (2, 9)5 days (2, 9)

8www.outcomes.org/stat
Patient DemographicsPatient Demographics
Medical historyMedical history %%
HypertensionHypertension 8989
Tobacco or alcohol useTobacco or alcohol use 3838
DiabetesDiabetes 3535
Chronic kidney diseaseChronic kidney disease 3131
End stage renal diseaseEnd stage renal disease 1111
Previous hospitalization for HTNPrevious hospitalization for HTN 2727
Neurological eventNeurological event 2323
Drug abuseDrug abuse 1515

10www.outcomes.org/stat
Predisposing Factors Contributing to Predisposing Factors Contributing to Hypertensive Event (35%)Hypertensive Event (35%)
FactorsFactors %%
Medication non-adherenceMedication non-adherence 2525
ChronicChronic 1616
CurrentCurrent 1010
Missed or incomplete dialysisMissed or incomplete dialysis 33
Anxiety/psychosocial reactionAnxiety/psychosocial reaction 22
Drug abuseDrug abuse 1111

11www.outcomes.org/stat
Primary Admitting DiagnosisPrimary Admitting Diagnosis
2%
2%
3%
4%
7%
8%
10%
11%
27%
0% 10% 20% 30%
Aortic dissection
Subdural hematoma
Renal failure
Ischemic stroke
Hemorrhagic stroke
Acute heart failure/PE
MI/ACS
Subarachnoid hemorrhage
Hypertension

12www.outcomes.org/stat
Resource UseResource Use
Procedure/care Procedure/care % %
Brain imaging (CT, MRI) Brain imaging (CT, MRI) 48 48
Admitted to ICUAdmitted to ICU 4848
Echocardiography Echocardiography 4545
Arterial lineArterial line 2525
Mechanical ventilation Mechanical ventilation 1818
Funduscopic examFunduscopic exam 1313

13www.outcomes.org/stat
Time from Qualifying BP to Initiation Time from Qualifying BP to Initiation of IV Therapyof IV Therapy
46.6%
74.2%
88.5%
97.0%100.0%
0%
20%
40%
60%
80%
100%
Within 1 hr Within 3 hrs Within 6 hrs Within 12 hrs Within 24 hrs

14www.outcomes.org/stat
First IV AntihypertensiveFirst IV Antihypertensive
8%
5%
8%
15%
15%
17%
32%
0% 10% 20% 30% 40%
Other
Sodium nitroprusside
Nicardapine
Hydralazine
Nitroglycerin
Metoprolol
Labetolol

15www.outcomes.org/stat
Number of Different IV Antihypertensives Number of Different IV Antihypertensives During Hospitalization by First ReceivedDuring Hospitalization by First Received
22%
51%
41%
41%
40%
32%
32%
28%
45%
27%
37%
42%
46%
21%
14%
32%
23%
25%
Sodium nitroprusside (n=82)
Nicardapine (n=121)
Hydralazine (n=235)
Nitroglycerin (n=241)
Metoprolol (n=277)
Labetolol (n=501)
Fir
st
IV A
nti
hy
pe
rte
ns
ive
Percent of Patients
One Two Three or more

16www.outcomes.org/stat
Median Systolic BP Over 24 h by First Median Systolic BP Over 24 h by First IV AntihypertensiveIV Antihypertensive
-40
-30
-20
-10
0
0 2 4 6 8 10 12 14 16 18 20 22 24
Time since IV initiation (h)
Ch
ang
e fr
om
qu
alif
yin
g (
%)
Enalaprilat* Hydralazine*Labetolol* Metoprolol*Nicardipine* Sodium Nitroprusside*Nitroglycerin*

17www.outcomes.org/stat
STAT ResultsSTAT Results
Median time to SBP of <160 mmHg: 4 hrsMedian time to SBP of <160 mmHg: 4 hrs 60% increased to >180 after initial control60% increased to >180 after initial control 4% had iatrogenic hypotension4% had iatrogenic hypotension 29% had recurrent, severe HTN 29% had recurrent, severe HTN
necessitating reinstitution of parenteral necessitating reinstitution of parenteral therapytherapy
65% had no documentation of follow-up 65% had no documentation of follow-up appointment being either scheduled or appointment being either scheduled or attendedattended

18www.outcomes.org/stat
Patient OutcomesPatient Outcomes
6.9%
11.0%
37.0%
9.3%
0%
10%
20%
30%
40%
50%
In-hospitaldeath*
Admit to 90-daydeath*
90-dayreadmission†
90-dayreadmission due
to HTN†
*n=1588 (all patients); †n=1415 (all patients alive at discharge and with 90-day follow-up)

19www.outcomes.org/stat
Short-Term (2 to 6 month) OutcomesShort-Term (2 to 6 month) Outcomes
Acute ConditionAcute Condition DeathDeath RehospitalizationRehospitalization
ACSACS1,2,31,2,3 5-7%5-7% 30%30%
CHFCHF44 8.5%8.5% 26%26%
Severe HypertensionSevere Hypertension55 11%11% 37%37%
1. OASIS-5 NEJM 20062. GUSTO IIb NEJM 19963. GRACE JAMA 20074. IMPACT-HF J Cardiac Failure 20045. STAT Registry results

20www.outcomes.org/stat
SummarySummary
Acute severe hypertensionAcute severe hypertension– Recurrent conditionRecurrent condition
– Associated with poor medical adherenceAssociated with poor medical adherence
– Heterogeneous management: ICU admission, Heterogeneous management: ICU admission, drugs used, BP targetsdrugs used, BP targets
– Alarmingly low rates of follow-upAlarmingly low rates of follow-up
– High mortality and morbidity, especially with High mortality and morbidity, especially with new or worsening end-organ damagenew or worsening end-organ damage
Major need to improve prevention and Major need to improve prevention and treatment of this understudied conditiontreatment of this understudied condition