Splicing RNA: Mechanisms. Splicing of Group I and II introns Introns in fungal mitochondria,...
-
Upload
darcy-fletcher -
Category
Documents
-
view
225 -
download
0
Transcript of Splicing RNA: Mechanisms. Splicing of Group I and II introns Introns in fungal mitochondria,...

Splicing RNA: Mechanisms

Splicing of Group I and II introns
• Introns in fungal mitochondria, plastids, Tetrahymena pre-rRNA
• Group I– Self-splicing– Initiate splicing with a G nucleotide– Uses a phosphoester transfer mechanism – Does not require ATP hydrolysis.
• Group II– self-splicing– Initiate splicing with an internal A– Uses a phosphoester transfer mechanism– Does not require ATP hydrolysis

Self-splicing in pre-rRNA in Tetrahymena : T. Cech et al. 1981
Exon 1 Exon 2Intron 1 Exon 1 Exon 2 Intron 1
+
pre-rRNASpliced exon
Intron circleIntron linear
pre-rRNANuclear extract
GTP
+ + + +- + - +- + + -
•Products of splicing were resolved by gel electrophoresis:
Additional proteinsare NOT needed forsplicing of this pre-rRNA!
Do need a G nucleotide (GMP, GDP, GTP or Guanosine).

Self-splicing by a phosphoester transfer mechanism
Exon 1
Exon 2
Intron 1
Exon 1 Exon 2
Intron 1+
PP
P P
G
OHU
UG
A P
U
P
U
P
P
G
OH
G
AP
N15 N16
G AN15
P OH
Circular intron+

A catalytic activity in Group I intron
• Self-splicing uses the intron in a stoichiometric fashion.
• But the excised intron can catalyze cleavage and addition of C’s to CCCCC

Group I intron catalyzes cleavage and nucleotide addition
5'pCCCCCC-OH+
2 pCCCCC-OH pCCCC-OH + pCCCCCC-OH
GGGAGG 5'
3'GOH
5'pCCCCC-OHGGGAGG 5'
3'GOHC-
5'pCCCCC-OH
GGGAGG 5'
3'GOH
GGGAGG 5'
GOHC-3'
5'pCCCC-OH

The intron folds into a particular 3-D structure
• Has active site for phosphoester transfer
• Has G-nucleotide binding site

Active sites in Group I intron self-splicing
GGGAGG
GGGAGG 5'
G
GGGAGG5'
3'GOH
GGGAGG
G
+
G414
G-OH
3'
CUCUCU5'
G-binding site
Substrate binding site
IGS
ex1
ex2
414
3'1st transfer
CUCUCU5'
ex1
G
OH
ex2
ex1
ex2+
414
2nd transfer
UUUACCUG
3rd transfer
414
5' G UUUACCU

Domains of the Group I intron ribozyme

RNAs that function as enzymes
• RNase P
• Group I introns
• Group II introns
• rRNA: peptide bond formation
• Hammerhead ribozymes: cleavage
• snRNAs involved in splicing

Hammerhead ribozymes
• A 58 nt structure is used in self-cleavage
• The sequence CUGA adjacent to stem-loops is sufficient for cleavage
CUGAG
ACCGGGGCC
AAA
ACUC G
AGU C
ACCACUGGUG
U
Bond that is cleaved.
5'3'
CUGA is required for catalysis

Design hammerhead ribozymes to cleave target RNAs
CUGAG
ACCGGGGCC
AAA
ACUC
GU A
A
GAGU C
ACCACUGGUG
U
Bond that is cleaved.
5'
3'
substrate strand
enzyme strand
Potential therapy for genetic disease.

Mechanism of hammerhead ribozyme
• The folded RNA forms an active site for binding a metal hydroxide
• Abstracts a proton from the 2’ OH of the nucleotide at the cleavage site.
• This is now a nucleophile for attack on the 3’ phosphate and cleavage of the phosphodiester bond.

Phosphotransfers for Group I vs. Group II & pre-mRNA
2’
GHO
3’Exon 1 Exon 2
OH
G
OH
Exon 1+2G
AHO
2’
Exon 1 Exon 2
OH
A
Exon 1+22’ A
++
Group IGroup II and pre-mRNA

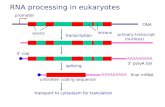
Splicing of pre-mRNA
• The introns begin and end with almost invariant sequences: 5’ GU…AG 3’
• Use ATP to assemble a large spliceosome
• Mechanism is similar to that of the Group II fungal introns:
– Initiate splicing with an internal A
– Uses a phosphoester transfer mechanism for splicing

Initiation of phosphoester transfers in pre-mRNA
• Uses 2’ OH of an A internal to the intron
• Forms a branch point by attacking the 5’ phosphate on the first nucleotide of the intron
• Forms a lariat structure in the intron
• Exons are joined and intron is excised as a lariat
• A debranching enzyme cleaves the lariat at the branch to generate a linear intron
• Linear intron is degraded

Splicing of pre-mRNA, step 1

Splicing of pre-mRNA, step 2

Investigation of splicing intermediates
5' 3'
RNARNase H
5'
oligodeoxyribonucleotide
+
|||||
In vitro splicing reaction: nuclear extracts + ATP+ labeled pre-mRNAResolve reaction intermediates and products on gels.Some intermediates move slower than pre-mRNA.Suggest they are not linear.Use RNase H to investigate structure of intermediate.RNase H cuts RNA in duplex with RNA or DNA.

RNase H + oligonucleotides complementary to different regions give very different products
5'3'
precursor RNA
exon 1 intron exon 2
1 2 3 4
splicing reaction
exons joined in a linear molecule excised intron, non-linear molecule
Map of positions of
oligodeoxyribonucleotides
that annealed to different
regions of the excised
intron. This is not the
structure of the excised
intron.
+
5'
5'
5'
3'
3'
3'
3'
3'
+
5'
RNase H

Analysis reveals a
lariate structure in inter-mediate
5'3'
precursor RNA
exon 1 intron exon 2
1 2 3 4
splicing reaction
excised intron, non-linear molecule
Map of positions of
oligodeoxyribonucleotides
that annealed to different
regions of the excised
intron.
+
5'
5'
3'
3'
3'
3'
3'
+
5'
After annealing with
the oligo, the
heteroduplexes were
treated with RNase H
Answer:
exons joined in a linear molecule
1
2
34
GU
AG
A
oligo 1
oligo 2
oligo 3
oligo 4

Involvement of snRNAs and snRNPs
• snRNAs = small nuclear RNAs• snRNPs = small nuclear ribonucleoprotein
particles• Antibodies from patients with the autoimmune
disease systemic lupus erythematosus (SLE) can react with proteins in snRNPs– Sm proteins
• Addition of these antibodies to an in vitro pre-mRNA splicing reaction blocked splicing.
• Thus the snRNPs were implicated in splicing

snRNPs
• U1, U2, U4/U6, and U5 snRNPs– Have snRNA in each: U1, U2, U4/U6, U5– Conserved from yeast to human– Assemble into spliceosome– Catalyze splicing
• Sm proteins bind “Sm RNA motif” in snRNAs– 7 Sm proteins: B/B’, D1, D2, D3, E, F, G– Each has similar 3-D structure: alpha helix
followed by 5 beta strands– Sm proteins interact via beta strands, may form
circle around RNA

Sm proteins may form ring around snRNAs
ANGUS I. LAMOND Nature 397, 655 - 656 (1999)RNA splicing: Running rings around RNA

Predicted structure of assembled Sm proteins
ANGUS I. LAMOND Nature 397, 655 - 656 (1999)RNA splicing: Running rings around RNA
Channel for singlestrand of RNA
4th beta strand of one Sm proteininteracts with 5th beta strand of next.

Assembly of spliceosome
• The spliceosome is a large protein-RNA complex in which splicing of pre-mRNAs occurs.
• snRNPs are assembled progressively into the spliceosome.– U1 snRNP binds (and base pairs) to the 5’ splice site– U2 snRNP binds (and base pairs) to the branch point– U4-U6 snRNP binds, and U4 snRNP dissociates– U5 snRNP binds
• Assembly requires ATP hydrolysis• Assembly is aided by various auxiliary factors and
splicing factors.

Spliceosome assembly and catalysis
Exon 1
AHO
2’
Exon 2
AHO
2’
U1 snRNP A
HO2’
U2 snRNP
U5 snRNP U4/U6
snRNP
U6 U4
AG
U
OH
Exons 1+2
2’ A
Sm proteins
snRNAs
Other proteins
U4?
AO2’G
U
U6
U6
U2
HU1
U5Spliceosome

Catalysis by U6/U2 on branch oligonucleotide in vitro
Figure 1 Base-pairing interactions in the in vitro-assembled complex ofU2–U6 and the branch oligonucleotide (Br). Shaded boxes mark the invariantregions in U6 and previously established base-paired regions are indicated.Dashed lines connect psoralen-crosslinkable nucleotides (S.V. and J.L.M.,unpublished data). The circled residues connected by a zigzag can becrosslinked by ultraviolet light. The underlined residues in Br constitute theyeast branch consensus sequence. Asterisks denote the residues involved inthe covalent link between Br and U6 in RNA X (see text). Arrowheads pointto residues involved in a genetically proven interaction in yeast22. Numbersindicate nucleotide positions from the 5' ends of full-length human U2 and U6.
Nature 413, 701 - 707 (2001) Splicing-related catalysis by protein-freesnRNAs SABA VALADKHAN & JAMES L. MANLEY

RNA editing
• RNA editing is the process of changing the sequence of RNA after transcription.
• In some RNAs, as much as 55% of the nucleotide sequence is not encoded in the (primary) gene, but is added after transcription.
• Examples: mitochondrial genes in trypanosomes and Leishmania.
• Can add, delete or change nucleotides by editing

Addition of nucleotides by editing
• Uses a guide RNA that is encoded elsewhere in the genome
• Part of the guide RNA is complementary to the mRNA in vicinity of editing
• U nt at the the 3’ end of the guide RNA initiates a series of phosphoester transfers that result in insertion of that U at the correct place.
• More U’s are added sequentially at positions directed by the guide RNA
• Similar mechanism to that used in splicing

What is a gene?• Making a correctly edited mRNA requires one
segment of DNA to encode the initial transcript and a different segment of DNA to encode each guide RNA.
• Thus making one mRNA that uses 2 guide RNAs requires 3 segments of DNA - is this 3 genes or 1 gene?
• Loss-of-function mutations in any of those 3 DNA segments result in an nonfunctional product (enzyme), but they will complement in trans in a diploid analysis!
• This is an exception to the powerful cis-trans complementation analysis to define genes.

Mammalian example of editing
• Apolipoprotein B in the intestine is much shorter than apolipoprotein B in the liver.
• They are encoded by the same gene.
• The difference results from a single nt change in codon 2153:
• CAA for Gln in liver, but UAA for termination of translation in intestine
• The C is converted to U in intestine by a specific deaminating enzyme, not by a guide RNA.