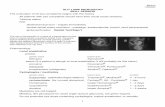
Slit lamp biomicroscopy
-
Upload
sssihms-pg -
Category
Health & Medicine
-
view
701 -
download
2
Transcript of Slit lamp biomicroscopy

SLIT LAMP BIOMICROSCOPY

We are forever indebted to Allavar Gullstrand, who won the Nobel prize in physiology or medicine in 1911 for his works on the dioptrics of the eye. It was he who first gave the concept of a slit lamp.
The word slit lamp is a misnomer as a slit is only one of the many diaphragms of the instrument. Hence the term “biomicroscopy” was introduced.

The most important advantage of slit lamp is the 3 dimensional view of the ocular structures.The 3 prerequisites for this 3d view are
-Stereopsis (which is provided by the binocular microscope)
-Direction of the light beam (which can be changed)
-The shape of the light beam (which can be changed)

PARTS OF THE SLIT LAMP
1.The observation system
2. The illumination system
3. The mechanical system

ILLUMINATION SYSTEM
Haag streit type- with vertical decoupling.
Zeiss type- with no vertical decoupling.

HALOGEN BULB
CONDENSING LENS
SLIT APERTURE
LENS
TILTED MIRROR
EYE
The light is controlled by a transformer and can be changed in intensity, height, width, direction, angle and colour.

OBSERVATION SYSTEM
Magnifications available are 5x 16x 25x 40x and in some 100x.Resolution is more important than magnification. This is provided by the short wave length light of the halogen bulb.
The resolution of a slit-lamp is dependent on the wavelength of light used, the refractive index between the eye and objective, the working distance, and the diameter of the objective lens.

MECHANICAL SYSTEMThe slit lamp is mounted on a stage designed for movement of microscope and patient positioning. The joystick controls the microscope position.

PARTS OF A SLIT LAMP

PRE EXAMINATION: 1.PATIENT POSITION:
The slit lamp should be locked in the farthest away position when the patient sits to ensure safetyExplain the procedure to the patient. Ask him to keep both eyes open and that he can blink his eyes. Adjust the height and distance of the table or chair.Patients back should be straight and neck aligned with back. He should lean forwards at the hips.The head should be such that the lateral canthus lies at the mark on the headbar.Ask the patient to grasp the handle.Children may stand or sit on their parents lap.


Adjust the ocular eyepieces according to the examiner’s refractive error and inter pupillary distance.
Fixation: ask the patient to fixate on a distant target or light or the observer’s ear with the other eye.
Magnification: Start from the least and increase as needed.
Focussing: Start with the stage all the way forward, you know that the only possible motion in order to focus is to pull back. Move the stage back slowly with the joystick until the eye is focused. you can look at the beam on the patient’s eye from the side of the instrument.

ILLUMINATION TECHNIQUES:1. DIFFUSE ILLUMINATION-
IS USED FOR GROSS SURVEY OF THE EYE. AFTER LOOKING FOR GROSS PATHOLOGY THE INTERESTED AREA CAN BE FOCUSSED.
• LIGHT AT 45 DEGREES• MICROSCOPE STRAIGHT• LEAST ILLUMINATION/
DIFFUSER/ NEUTRAL DENSITY FILTER/ BLUE/GREEN FILTER.
• LEAST MAGNIFICATION.• FULL WIDTH BEAM.

2. DIRECT IILLUMINATIONA) BEAM/NARROW BEAM:
USES A 0.5-1MM SLIT HIGH ILLUMINATIONLOW OR MEDIUM MAGNIFICATION.
• TO SEE CORNEAL THICKNESS, SHAPE, BULLAE, DELLEN
• SITE OF FOREIGN BODY• OPACITIES, SCARSS• DEPTH OF AC• LOCATION OF
CATARACT



B) CONICAL/ PIN POINTTO SEE AC FOR CELLS AND FLARE• HIGH MAGNIFICATION
(16X)• HIGH ILLUMINNATION• MICROSCOPE STRAIGHT
AHEAD• LIGHT 45 TO 60 DEGREES.
FLARE IS DUE TO PROTEINS HENCE APPEARS GREY / MILKYCELLS REFLECT LIGHT AND HENCE APPEAR AS WHITE DOTS.SEEN DUE TO TYNDALL EFFECT.

TYNDALL EFFECT
Light scattering by particles in a colloid or suspension.

C) BROAD BEAM/ TANGENTIAL/ PARALLELIPIPED
GIVES A 3 DIMENTIONAL VIEW OF CORNEA DUE TO SHADOWS.• 2-4MM SLIT• LIGHT AT 45 DEGREESAs the beam sweeps the cornea, it enhances surface irregularities by creating shadows.
TO SEE TEAR DEBRISCORNEAL NERVES AND VESSELSCORNEAL OPACITIESSTRIAE OR DM FOLDSKRUKENBERG SPINDLESPSEUDOEXFOLIATION ON LENS

D) SPECULAR REFLECTION
This technique is used to view the endothelium.Specular reflection is achieved by positioning the beam of light and microscope in such a position so that the angle of incidence is equal to the angle of reflection.This method is monocular.In this technique, position the illuminator about 30 degrees to one side and the microscope 30 degrees to the other side. The angle of the illuminator to the microscope must be equal and opposite.

A parallelepiped is used for specular reflection.The focus is moved back toward the endothelial cells. There will be a point where two images are seen, one bright, and the other ghost-like or copper-yellow in color. Then move the light a little to the side, and look adjacent to it, at the reflection from the endothelial surface. Now switch to the highest magnification available.When the biomicroscope is focused on the ghost-like filament a mosaic of hexagonal cells are seen.Can be used to see anterior and posterior lens capsule too.

SPECULAR REFLECTION

3) INDIRECT ILLUMINATION:A) PROXIMAL ILLUMINATION:
Use a parallelepiped beam sharply focused on a given structure like the cornea. The light passes through the cornea and falls out of focus on the iris. The dark area just lateral or proximal to the parallelepiped is the indirect or proximal zone of illumination.

B) RETROILLUMINATION: The light is reflected off the deeper structures, such as the iris or retina, while the microscope is focused to study the more anterior structures in the reflected light. IRIS RETROILLUMINATION:DIRECT- to view corneaINDIRECT- to view cornea (and angles)
RETROILLUMINATION FROM RETINA: to see the lens and cornea.

Direct iris retroilllumination
Indirect iris retroillumination
Light beam hits iris behind the pathology.
Illuminates cornea from behind.
Accentuates the refractive properties of the cornea.
Against a light background.
Decentered beam hits the iris adjacent to the corneal pathology.Illuminates cornea from behind
Against a dark background.

Direct iris retroilllumination
Indirect iris retroillumination

Directretroilluminatio
n
Indirect retroilluminati
on

Retroillumination from the fundus:
Slit beam is placed nearly coaxial with microscope and rotated slightly off axis. This allows the red light reflected from the fundus to pass through thee lens and cornea.

Direct iris retroillumination
Indirect iris retroilluminat
ion
Retroilluminationfrom fundus

TRANSILLUMINATION FROM IRIS
In transillumination, the iris is evaluated by how light passes through it.This technique also takes advantage of the red reflex.It helps to see defects in the iris.

SCLEROTIC SCATTER:
The optical principle is same as that of fibre optics- total internal reflection of light.The slit beam is directed at the limbus from where the sclera scatters the light. Some of this enters the corneal stroma and travels the entire cornea getting internally reflected. An opacity causes light to scatter and be visualised by the examiner.

•This is such as to make the system “parfocal” •i.e the focus of the slit and the focus of the microscope are at the same point. •This parfocality may occasionally need to be dissociated as for example in the technique of sclerotic scatter.The coupling between the slit lamp and the biomicroscope •This allows both the slit and the microscope to rotate about the point of focus (i.ethe eye)Dissociation of parfocality can be done in “Haag Streit” type slit lamps by loosening the sclerotic scatter knob
The coupling between the slit lamp and the biomicroscope

SCLEROTIC SCATTER

FILTERS IN A SLIT LAMP:1) COBALT BLUE FILTER:
The cobalt blue filter is used in conjunction with fluorescein dye. The dye pools in areas where the corneal epithelium is broken or absent. The blue light excites the fluorescein, which then takes on a yellowish glow.

2) GREEN FILTER (RED FREE FILTER):
• The green filter obscures anything that is red.Thus, blood vessels or hemorrhages appear black. This increases contrast, revealing the path and pattern of inflamed blood vessels.
• Areas of the episclera where lymphocytes have gathered in response to an inflammatory or immune response will appear as yellow spots under the red-free light.
• Fleischer ring can also be viewed satisfactorily with the red-free filter.

3) DIFFUSER:Some instruments have a diffuser, which is a piece of frosted glass or plastic that flips in front of the illuminator. The diffuser scatters the light, causing an even spread of light over the entire ocular surface.


ATTACHMENTS:
1. GAT2. PACHYMETRY3. GONIOSCOPY4. HRUBY LENS5. DIGITAL CAMERA.

Thank you