ROLE OF THE CONTRACTILE VACUOLE COMPLEX AS A ......relies on protein secretion of...
Transcript of ROLE OF THE CONTRACTILE VACUOLE COMPLEX AS A ......relies on protein secretion of...

ROLE OF THE CONTRACTILE VACUOLE COMPLEX AS A
TRAFFICKING HUB IN TRYPANOSOMA CRUZI
by
SAYANTANEE NIYOGI
(Under the Direction of Roberto Docampo)
ABSTRACT
Trypanosoma cruzi is the etiologic agent of Chagas disease. It contains a Contractile
Vacuole Complex (CVC) that plays a vital role in the regulation of its cell volume and in
its responses to osmotic stresses in all its life cycle stages. It is peculiar that though,
T.cruzi is not a free-living organism it has a CVC; thus suggesting that the CVC could
have functions beyond just osmoregulation as occurs in some other protists; where the
CVC is involved in regulating calcium homeostasis and in the transfer of proteins to the
surface. Besides, the approach of combined proteomic and bioinformatics analyses
identified proteins localized to the CVC, several of them having trafficking roles, and
implying to a potential novel role of the CVC.
Here we used a combination of genetic and biochemical approaches to establish the
contribution of the CVC as a trafficking hub. T. cruzi relies on protein secretion of
glycosylphosphatidylinositol (GPI)-anchored surface proteins for invasion of host cells
and establishment of infection. In this study we show that the CVC acts as a trafficking
intermediate before GPI-anchored proteins reach the cell surface. Additionally we also
identify CVC-located TcRab11 as a regulator of protein transport of GPI-anchored trans-

sialidase to the plasma membrane, a process essential for the establishment of infection.
Demonstration of the role of TcTS in infection has been previously difficult given the
large number of genes encoding for this protein distributed through the genome of the
parasite. We also studied the role of another CVC-located Rab. Rab32 is located in
lysosome-related organelles (LRO) and since acidocalcisomes are LROs we investigated
whether TcRab32 is needed for the structure and function of acidocalcisomes. By
constructing GDP-bound dominant negative mutants of TcRab32 we were able to show a
defect in trafficking, which ultimately affects parasite infectivity. This study with
TcRab32 provides the link between the acidocalcisome and the contractile vacuole
complex as observed in T. cruzi and in some other protists like Chlamydomonas
reinhardtii and Dictyostelium discoideum.
Our results are consistent with a role of the CVC in regulating membrane traffic to
maintain the function of the acidocalcisome as well as traffic to the plasma membrane of
T. cruzi.
INDEX WORDS: T.cruzi, Contractile Vacuole Complex (CVC), acidocalcisomes,
TcRab32, TcRab11, trans-sialidase, trafficking, membrane

ROLE OF THE CONTRACTILE VACUOLE COMPLEX AS A
TRAFFICKING HUB IN TRYPANOSOMA CRUZI
by
SAYANTANEE NIYOGI
BSc., Asutosh College, Kolkata, India, 2006
MSc., University of Calcutta, Kolkata, India, 2008
A Dissertation Submitted to the Graduate Faculty of The University of Georgia in Partial
Fulfillment of the Requirements for the Degree
DOCTOR OF PHILOSOPHY
ATHENS, GEORGIA
2014

© 2014
SAYANTANEE NIYOGI
All Rights Reserved

ROLE OF THE CONTRACTILE VACUOLE COMPLEX AS A
TRAFFICKING HUB IN TRYPANOSOMA CRUZI
by
SAYANTANEE NIYOGI
Major Professor: Roberto Docampo
Committee: Boris Striepen
Rick Tarleton
Steve Hajduk
Electronic Version Approved:
Julie Coffield
Interim Dean of the Graduate School
The University of Georgia
August 2014

iv
DEDICATION
Dedicated to Maa, Baba, Titli and Deep for their constant encouragement, support and
unconditional love.

v
ACKNOWLEDGEMENTS
I would like to thank my mentor Dr Docampo for the opportunity, the support, the time
and patience he provided for me and the great projects he had lined up for me. He had to
start right from scratch with me!! When I joined the lab, I did not have a lot of experience
on the bench, in designing experiments. But thanks to him, I think I have become slightly
better at it. I thank him for all that he has taught me, all the knowledge he imparted on me
and guiding me all along. His love for science, his dedication to work has been an
inspiration to me; that helped me to work hard on my projects with full sincerity. He has
always been keen on answering my doubts, correcting my mistakes and also making sure
that I do not repeat those mistakes. He always encouraged me to present my work both in
external meetings as well as in internal seminars; something that has helped boost my
level of confidence. I would also like to thank Dr Moreno for all her invaluable
suggestions during lab meeting; which definitely made my dissertation a lot more solid.
Also her review whenever I presented during a lab meeting or practiced for an upcoming
seminar with her; is invaluable. I think I have learnt a lot about how important it is to be
able to present your work and make sure that people can follow the talk from these
discussions. Also I would like to thank my committee members Dr Striepen, Dr Tarleton
and Dr Hajduk whose advice and suggestion added value to my thesis and towards the
flow of the project.
Thanks to Veronica who trained me in my first year and for her positive criticizm. I am
and will be ever grateful to Melina for being the best lab manager and being a great

vi
friend; for always answering my questions and always encouraging me in the toughest of
time. Thanks to Noelia who is a great friend and a wonderful labmate; she helped answer
some of my doubts regarding the writing of the dissertation. And everyone else in the
Docampo-Moreno lab; an environment that always inspired and taught me to work hard,
help each other, to work together as a unit, discuss and share problems; and also taught
me how important it is to recognize everyone’s contribution and also to be able to
critically review each other’s as well as your own work. I can undoubtedly say these were
the best 5 years of my life!!
A big thank you to my parents; for giving us the best education and the best childhood.
And most importantly making sure that we become nice human beings; something that I
will carry with me wherever I go. They have made countless sacrifices to support me and
my sister and give us a life which I know was very difficult for them to provide at the
moment. My sister, for being my biggest cheerleader and for all the love she gives me.
Though I am the elder one, but her wisdom and mature suggestions have definitely taught
me a lot in life. Thank you to my brother-in-law who is more like my own brother; for all
the encouragement, the sense of humor you keep pouring in at difficult times which
always manages to bring a smile to my face on those days, when going gets tough. My
gratitude to my parents-in-law for being understanding and very supportive throughout.
And a big thank you to my husband for being my strength. Words will never do justice to
what you mean to me. Thank you for being by my side, for backing me up when I fall
down, for respecting my space and also being my biggest critique. You have indeed been
my guide, my teacher and my best friend. Surprisingly all my worries disappeared after I

vii
came back home to him. The support and love from my family has been my biggest
strength and instrumental in whatever I have been able to achieve.

viii
TABLE OF CONTENTS
Page
ACKNOWLEDGEMENTS v
LIST OF FIGURES xi
CHAPTER
1 INTRODUCTION………………………………………………………………….1
Introduction…………………………………………………………………………….…...1
Structure of the Dissertation……………………………………………..…………………3
References ……………………………………………………………………….…………4
2 LITERATURE REVIEW…………………………………………………………..5
Trypanosoma cruzi and Chagas disease ……………………………………………………5
Life cycle of Trypanosoma cruzi …………………………………………………………..6
Contractile Vacuole Complex………………………………………………………………7
Acidocalcisomes…………………………………………………………………………..10
Traffic in trypanosomes…………………………………………………………………...13
Rab proteins………………………………………………………….……………………15
Tools to investigate the function of Rab proteins in vesicle fusion and
transport mechanism……………………………………………………………………....16
GDP bound “OFF” stage of Rab proteins: examples……………………………………..18
Role of Rab32 protein in trafficking……………………………………………………...18
Role of Rab11 protein in trafficking………………………………………………………19

ix
Rab protein prenylation and potential treatment of Chagas disease………………………20
Overview of Trypanosoma cruzi infection……………………………………………......21
GPI-anchored surface proteins…………………………………………………………....23
Trans-sialidase…………………………………………………………………………....24
References………………………………………………………………………………...27
3 RAB11 REGULATES TRAFFICKING OF TRANS-SIALIDASE TO
THE PLASMA MEMBRANE THROUGH THE CONTRACTILE VACUOLE
COMPLEX OF TRYPANOSOMA CRUZI ………………………………………………..45
Abstract……………………………………………………………………………………46
Author Summary…………………………………………………………………………..47
Introduction……………………………………………………………………………..…47
Results …………………………………………………………………………………….50
Discussion…………………………………………………………………………………59
Materials and Methods….………………………………………………………………...64
References………………………………………………………………………………...73
4 RAB32 IS ESSENTIAL FOR MAINTAINING
FUNCTIONAL ACIDOCALCISOMES AND FOR GROWTH AND
VIRULENCE OF TRYPANOSOMA CRUZI……………………………………………101
Abstract………………………………………………………………………………..…102
Author Summary ………………………………………………………………………...102
Introduction………………………………………………………………………………103
Results……………………………………………………………………………………105
Discussion……………………………………………………………………………......111

x
Materials and methods…………………………………………………………………...113
References ……………………………………………………………………………….121
5 CONCLUSION…………………………………………………………………..139
Summary of key finding…………………………………………………………………139
Future work………………………………………………………………………………141
References ……………………………………………………………………………….147

xi
LIST OF FIGURES
Page
Figure 2.1: Life cycle of T. cruzi…………………………………………………………………39
Figure 2.2: The CVC in T. cruzi epimastigotes…………………………………………...40
Figure 2.3: Diagramatic representation of the enzymes and transporters
tentatively identified in the acidocalcisome of T. cruzi……………………………...……41
Figure 2.4: The GTP-GDP cycle of Rab-GTPases………………………………………..42
Figure 2.5: Schematic model summarizing the molecules involved on parasite-host
cell interaction process exposed on the surface of a host cell and in trypomastigotes
of T. cruzi……………………………………………………………………………..........43
Figure 2.6: Model of T. cruzi invasion………………………………………………..…..44
Figure 3.1: Fluorescence microscopy analysis of TcRab11 in different stages
of T. cruzi……………………………………………………………………………....................82
Figure 3.2: GFP-TcRab11DN localizes to the cytoplasm of different life cycle stages.....84
Figure 3.3: Regulatory volume changes of epimastigotes………………………………...85
Figure 3.4: Co-localization of GFP-TcRab11 and TcTS during amastigote differentiation
in human foreskin fibroblasts……………………………………………………………...87
Figure 3.5: Localization of TcTS during differentiation to cell-derived and metacyclic
trypomastigotes………………………………………………………………….............88
Figure 3.6: Cryo-immunoelectron microscopy localization of GFP-TcRab11 and TcTS
in amastigotes………………………………………………………………………….......89

xii
Figure 3.7: Overexpression of GFP-TcRab11DN reduces the surface
expression of TcTS……………………………………………………………………….91
Figure 3.8: Localization of surface proteins in GFP-TcRab11OE and
GFP-TcRab11DN-expressing parasites…………………………………………………..92
Figure 3.9: Localization of anti-Gal antibodies…………………………………………...93
Figure 3.10: Association of CVC proteins with lipid rafts and reduced infectivity
Of TcRab11DN trypomastigotes………………………………………………………….94
Figure 3.11: Cryo-immunoelectron microscopy localization of GFP-TcRab11
in epimastigotes…………………………………………………………………………..96
Figure 3.12: Growth rate, and western blot analyses of overexpressed TcRab11…..........97
Figure 3.13: TcAQP1 localization is not affected in GFP-TcRab11DN mutants
and western blot analysis of wild type and GFP-TcRab11DN shows specificity
of anti-SAPA antibodies………………………….............................................................98
Figure 3.14: Localization of GFP-TcRab11 and gp35/50 mucins
during metacyclogenesis………………………………………………………………….99
Figure 3.15: Infections of host cells by trypomastigotes overexpressing TcRab11……..100
Figure 4.1: TcRab32 localization in different life stages of T. cruzi……………………127
Figure 4.2: TcRab32 is digeranylated in vitro…………………………………………..129
Figure 4.3: Localization of GFP-TcRab32 mutants……………………………………..130
Figure 4.4: Lack of colocalization between GFP-TcRab32 and mitochondrial
marker and localization of mitochondrial marker is not affected
in TcRab32 mutants……………………………………………………………………..131
Figure 4.5: Colocalization of GFP-TcRab32 and VP1 under osmotic stress…………...132

xiii
Figure 4.6: Reduced short chain poly P and PPi levels in TcRab32DN
epimastigotes in comparison to wild type epimastigotes………………………………...133
Figure 4.7: Reduction in electron dense acidocalcisomes and considerable
increase in empty vacuole in TcRab32DN epimastigotes in comparison
to wild type……………………………………………………………………………...134
Figure 4.8: Traffic of trans-sialidase is not affected in TcRab32DN
mutant trypomastigotes…………………………………………………………...……..135
Figure 4.9: Effect of TcRab32 mutations on the cell growth of epimastigotes
and their response to hyposmotic and hyperosmotic
stress conditions………………………………………………………….......................136
Figure 4.10: Reduced infectivity of TcRab32 mutant trypomastigotes…………………137

1
CHAPTER 1
INTRODUCTION
Introduction
Kinetoplastids are a group of flagellated protozoans that include the species
Trypanosoma and Leishmania, which are human pathogens with devastating health and
economic effects. Because of their early divergence from other eukaryotes, they exhibit
unusual characteristics. They are distinguished by the presence of a DNA-containing
region known as “kinetoplast” in their single large mitochondrion. This was the first
extranuclear DNA ever discovered, long before mammalian mitochondria were shown to
contain DNA. Besides, trypanosomatids have unique peculiarities like the presence of
organelles like glycosomes, which are specialized peroxisomes containing most
glycolytic enzymes [1] [2]; acidocalcisomes, acidic organelles rich in calcium and
polyphosphate required for pH homeostasis and osmoregulation [3] [4]; contractile
vacuole complex (CVC); needed to maintain osmoregulation [5], and as a trafficking
intermediate; and biological processes first described in these organisms like RNA
editing, glycosylphosphatidylinositol(GPI)-anchor synthesis [6] and trans-splicing [7].
Details of the CVC and acidocalcisomes will be discussed in the following chapters.
Kinetoplastids are evolutionarily more early branched compared to the majority of other
groups of parasitic protists, widespread and adaptable, which is an apparent reflection of
their extremely successful life style. Although the different kinetoplastid pathogens have
a similar genomic organization and similar cellular structures and all undergo

2
morphogenesis during their life cycle, these pathogens are transmitted by different
vectors and cause specific diseases [8]. They are the causative agents of important
diseases such as African sleeping sickness (caused by the Trypanosoma brucei group),
Chagas' disease (caused by Trypanosoma cruzi) and Leishmaniases (caused by
Leishmania spp). People primarily in tropical and subtropical areas of the world are at
risk of contracting these diseases. Some of these parasites (T. brucei group) have an
efficient capability to adapt to their hosts, evading the host immune system by antigenic
variation.
A deep knowledge of what is occurring in the structures, organelles and in the cell
biology of these parasites may open new perspectives for the control of disease through
the development of (a) new chemotherapeutic agents, (b) vaccines or (c) more specific
diagnostic procedures. The completion of genome sequences of trypanosomatids, T.
brucei [9], T. cruzi [10] and Leishmania major [11] and also transcriptome and proteomic
analyses have generated information that provide helpful tools for investigation. Besides,
the TriTryp genome has advanced our understanding of the biology of these parasites and
their host-parasite interaction.
Though significant advance has been made in understanding the mechanism used by
these organisms in invading the host cell, very little is known about the molecular
machinery involved in trafficking in T.cruzi; specifically traffic of surface proteins and
endosomal targeting in T. cruzi. In this work we provide experimental evidence for the
role of the contractile vacuole complex (CVC) as a trafficking hub, involved in the traffic
of GPI-anchored proteins to the plasma membrane of the parasite and also its role as

3
endosomal system that transfers membrane proteins to the acidocalcisomes. We use a
combination of genetic and biochemical approach to address the goal.
Structure of the Dissertation
The dissertation is subdivided into five chapters. Chapter 2 reviews the current
knowledge regarding topics which are pertinent to the specific aims of my dissertation. In
this chapter I try to portray a detailed analysis of key pathways that were necessary for
our study, with a focus on mechanism of trafficking of GPI-anchored protein in other
organisms, and also a detailed analysis of the function of Rab-GTPases and tools to
investigate Rab function, as studied in other systems. This chapter also provides
structural and functional overview of organelles studied in this research. Chapter 3
describes the role of T. cruzi Rab11 in the traffic of trans-sialidase to the plasma
membrane via the contractile vacuole complex. This work was published in PLoS
Pathogens [12]. Chapter 4 describes the role of the other Rab-GTPase, T. cruzi Rab32, in
maintaining the function of acidocalcisomes and its involvement in growth and virulence
of the parasite. Chapter 5 provides an overall conclusion for this research in elucidating
the role of the CVC as a trafficking intermediate in T. cruzi. It also highlights open
questions pertinent for future study, not only limited to the cell biology of T. cruzi, but
also to the other trypanosomatids.

4
REFERENCES
1. Parsons M (2004) Glycosomes: parasites and the divergence of peroxisomal purpose.
Mol Microbiol 53: 717-724.
2. Opperdoes FR, Borst P (1977) Localization of nine glycolytic enzymes in a
microbody-like organelle in Trypanosoma brucei: the glycosome. FEBS Lett 80:
360-364.
3. Docampo R, de Souza W, Miranda K, Rohloff P, Moreno SN (2005) Acidocalcisomes
- conserved from bacteria to man. Nat Rev Microbiol 3: 251-261.
4. Docampo R, Scott DA, Vercesi AE, Moreno SN (1995) Intracellular Ca2+ storage in
acidocalcisomes of Trypanosoma cruzi. Biochem J 310 ( Pt 3): 1005-1012.
5. Rohloff P, Docampo R (2008) A contractile vacuole complex is involved in
osmoregulation in Trypanosoma cruzi. Exp Parasitol 118: 17-24.
6. Ferguson MA (1999) The structure, biosynthesis and functions of
glycosylphosphatidylinositol anchors, and the contributions of trypanosome
research. J Cell Sci 112 ( Pt 17): 2799-2809.
7. Liang XH, Haritan A, Uliel S, Michaeli S (2003) trans- and cis-splicing in
trypanosomatids: mechanism, factors, and regulation. Eukaryot Cell 2: 830-840.
8. Mableson HE, Okello A, Picozzi K, Welburn SC (2014) Neglected zoonotic diseases-
the long and winding road to advocacy. PLoS Negl Trop Dis 8: e2800.
9. Berriman M, Ghedin E, Hertz-Fowler C, Blandin G, Renauld H, et al. (2005) The
genome of the African trypanosome Trypanosoma brucei. Science 309: 416-422.
10. El-Sayed NM, Myler PJ, Bartholomeu DC, Nilsson D, Aggarwal G, et al. (2005) The
genome sequence of Trypanosoma cruzi, etiologic agent of Chagas disease.
Science 309: 409-415.
11. Ivens AC, Peacock CS, Worthey EA, Murphy L, Aggarwal G, et al. (2005) The
genome of the kinetoplastid parasite, Leishmania major. Science 309: 436-442.
12. Niyogi S, Mucci J, Campetella O, Docampo R (2014) Rab11 regulates trafficking of
trans-sialidase to the plasma membrane through the contractile vacuole complex
of Trypanosoma cruzi. PLoS Pathog 10: e1004224.

5
CHAPTER 2
LITERATURE OVERVIEW
Trypanosoma cruzi and Chagas disease
The obligate intracellular parasite Trypanosoma cruzi is the causative agent of Chagas
disease, which is the leading cause of cardiac death in endemic areas throughout Latin
America, where it is mostly vector-borne transmitted to humans by contact with faeces of
triatomine bugs, known as ‘kissing bugs’. The invertebrate hosts are Hemiptera and
Reduvidae such as Rhodinus prolixus, Triatoma infestans, and Panstrongylus megistus.
More than 11 million people are infected with the parasite and some 40 million more are
at risk. Among other Neglected Tropical Diseases (NTD), Chagas disease ranks near the
top in terms of annual death and DALYs (Disability Adjusted Life Years) lost [1,2]. In
the past decades it has been increasingly detected in the United States of America,
Canada, many European and some Western Pacific countries. This is due mainly to
population mobility between Latin America and the rest of the world [3] [4]. Although
currently available nitroheterocyclic drugs (benznidazole and nifurtimox) are moderately
efficacious when administered during the acute phase, they have been minimally
successful in treating chronic infection. Chronic Chagas' cardiomyopathy is the most
serious and frequent manifestation of Chagas’ disease characterized by cardiac
arrhythmias, heart failure, and risk of sudden death from ventricular fibrillation or
tachycardia [5]. It is the main cause of mortality among these patients and is associated to

6
a poorer survival compared with other forms of cardiomyopathies. Early detection of
heart involvement in seropositive individuals remains challenging.
Life cycle of Trypanosoma cruzi
Persistent infection with T. cruzi causes Chagas disease. The parasite is transmitted to
humans by infected blood-sucking Triatominae insects, which defecate after obtaining a
blood meal and thus release the trypomastigotes in faeces. Scratching the area of bite
causes the trypomastigotes to enter the wound and invade nearby cells. While
intracellular, they differentiate into amastigotes that multiply by binary fission. The
amastigotes differentiate into trypomastigotes, which are released into the bloodstream
and infect cells of multiple organs and tissues, including the heart, gut, CNS, smooth
muscle, and adipose tissue and once again become amastigotes. The Triatominae insects
become infected when they take a parasite-containing blood meal from an infected
human or animal. The trypomastigotes undergo morphological and physiological
transformations in the midgut of the vector and differentiate into infective
trypomastigotes in the hindgut. The morphological characteristics of these developmental
forms (intracellular, blood and insect stages) have been extensively investigated by
different microscopy techniques. The structural details of the different forms are as
following:
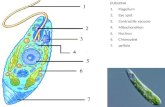
1) Amastigotes: They are spherical in shape, able to divide and are infective.
2) Trypomastigotes: these forms have a length of about 25 μm and a diameter of
about 2 μm. The kinetoplast is located posterior to the nucleus. These forms are
not able to divide. The nucleus is elongated and organized in the central portion of
the cell.

7
3) Epimastigotes: They are spindle-shaped, 20–40 μm long with kinetoplast located
anterior to the nucleus. These forms are able to divide. The nucleus has a rounded
shape.
Contractile vacuole complex
The Contractile Vacuole Complex (CVC) was first described in Paramecium more than
200 years ago (Spallanzani, 1799) and was later found in a wide range of amoeba,
photosynthetic and nonphotosynthetic flagellates and ciliates. Clark (1959) (J. Protozool.,
1959) was the first to describe the presence of a CVC in T. cruzi and reported a pulsation
period (time between contractions) in epimastigotes between 1 min and 1 min and 15 s.
Besides T. cruzi the CVC is also present in Leishmania sp [6] and in monogenetic
trypanosomes like Leptomonas collosoma [7] and Crithidia luciliae [8] and apparently
absent in Trypanosoma brucei.
Architecture: Structure and composition
The CVC is an intracellular compartment with an osmoregulatory role in different
protists (discussed below). This compartment has a bipartite structure, consisting of a
central vacuole or bladder and a surrounding loose network of tubules and vesicles
named the spongiome [9]; [10]. Functional distinctions between these 2 components of
the CVC were evidenced by the localization of different proteins to each compartment.
Recent proteomic analysis and microscopy studies of green fluorescent protein (GFP)-
tagged proteins have revealed the presence of the vacuolar H+-ATPase, Rab11, Rab32,
AP180, VAMP1 and a putative phosphate transporter (PT) in the bladder while
calmodulin and two SNAREs are localized to the spongiome [11]. The CVC is present in
all the different life cycle stages of T.cruzi. Fig 2.2 shows a turgid central vacuole and

8
interconnected tubules forming a network in a well preserved contractile vacuole. In fact
the contractile vacuole is believed to be docked to a domain of the flagellar pocket (Fig
2C, 2D) with the presence of an electron-dense region between the two. This domain of
the contractile vacuole seems to get deformed because of its physical connection with the
flagellar pocket (Fig 2.2D). This feature has been shown before in Leptomonas spp where
the contractile vacuole membrane is permanently attached to the plasma membrane of the
flagellar pocket by a dense adhesion plaque [7].
The search for other functions of the CVC
The function of the CVC with regard to osmoregulation in T.cruzi has been a subject of
study in our lab for many years with the result of several publications stating the
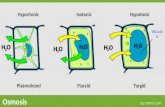
mechanistic role of this organelle. The CVC accumulates water through an aquaporin or
water channel [12] [13] [14] and expels it out of the cell through pores in the plasma
membrane [9,10]. It is important for regulatory volume decrease (RVD) after hyposmotic
stress [13], as well as for shrinking of the cells when submitted to hyperosmotic stress
[4]. The CVC bladder does not burst during volume regulation phenomenon. It has been
proposed [15] that the connected tubular spongiome acts as a reservoir for water which
increases in surface area by virtue of the phospholipids present in the membrane to
accommodate the increase in volume during hyposmotic stress. This result is supported
by our data as shown in Figure 3.3C-D and 3.11B-C and discussed in chapter 3.
Other roles of the CVC in T. cruzi had not been investigated before this dissertation.
CVC has been studied in several protists and we will discuss its role below. The presence
of several proteins related to calcium signaling [10] underscore the role of the CVC in
Ca2+
homeostasis. It also has a role in transfer of some proteins to the plasma membrane

9
[16-18]. In Dictyostelium discoideum, the vacuolar proton ATPase (V-H+-ATPase) and
calmodulin (CaM) move to the plasma membrane when cells are starved during
stationary phase [16], and the Ca2+
-ATPase PAT1 moves to the plasma membrane when
cells are incubated at high Ca2+
concentrations [17]. Some luminal proteins, such as the
adhesins DdCAD-1 and discoidin-1 can also be targeted to the cell surface via the CVC
in D. discoideum [18,19]. We recently reported (Chapter 3) the role of the CVC in traffic
of GPI-anchored surface proteins in T. cruzi [20]. In T. cruzi epimastigotes, the
polyamine transporter TcPOT1.1, which localizes to CVC-like structures, has also been
reported to appear in the plasma membrane when the culture medium is deficient in
polyamines [21]. Also a phosphate transporter (TcPHO1) has been localized to the CVC
[11]. It is interesting to note that dajumin-GFP (the CVC marker) is trafficked to the
CVC of D. discoideum via the plasma membrane and is internalized by a clathrin-
dependent mechanism, suggesting that clathrin-mediated endocytosis may have a role in
the biogenesis and/or, maintenance of the contractile vacuole by functioning in retrieval
of proteins from the cell membrane [22]
The proteomic and bioinformatics study [11] of the CVC of T. cruzi identified a cohort of
proteins having trafficking roles. This study detected the presence of SNAREs 2.1 and
2.2, VAMP1 (VAMP7 homolog), AP180, and the small GTPases Rab11 and Rab32. The
accumulation of all of these proteins which have role in vesicle fusion/fission and
tethering events in the CVC, suggests that the CVC of T. cruzi was acts as a trafficking
hub.

10
Acidocalcisome
Acidocalcisomes were first described in trypanosomes and later found in Apicomplexan
parasites, algae, slime molds, fungi, eggs of different origins, and human cells [23].
These organelles are acidic compartments storing high concentrations of calcium and
polyphosphate (polyP) [24]. Figure 2.3 shows the pumps and antiporters that are present
in the membrane of the acidocalcisome and are necessary for their cation and water
accumulation and release, as well as enzymes involved in the synthesis and degradation
of pyrophosphate and polyP. A number of these pumps, channels, and exchangers in the
membranes were biochemically characterized and their genes cloned and expressed.
Acidocalcisome: Structure
Acidocalcisome of protists in general are spherical in shape. Trypanosomatids are rich in
very short chain polyP such as polyP3, polyP4, and polyP5. PolyP is arbitrarily divided
into two forms: short-chain (from 3 to ~300 Pi) and long-chain (from 300 to ~1000 Pi)
polyP, based on the method used for its extraction. Besides polyP, trypanosomatids also
contain orthophosphate (Pi) and PPi. These phosphorus compounds are in close
association to cations (sodium, potassium, magnesium, calcium, zinc, and iron) and basic
amino acids [24] [25]. In eukaryotic cells, polyP is present in different compartments,
including the cytosol, nucleus, lysosomes, and mitochondria, but is preferentially
accumulated in acidic vacuoles such as the yeast vacuole and acidocalcisomes [23,26].
Taking into account its total concentration and the relative volume of acidocalcisomes in
some of these cells (about 1–2% of the total cell volume), the intraorganellar
concentration is in the molar range (~3 M) [24]. These vesicles are acidic and thus
accumulate dyes like acridine orange [27]. DAPI can be used to detect polyP in these

11
organelles [28]. By standard electron microscopy, they appear as empty vacuoles or
vacuoles containing a thin layer of dense material or an inclusion that sticks to the inner
face of the membrane. The electron-dense material inside acidocalcisomes is better
preserved with the use of cryomethods [29] where the organelles seem completely filled
by an electron-dense material. Two proton pumps were found in acidocalcisomes of
protists. One is the vacuolar-type H+-ATPase, a macromolecular complex of 14 subunits
[30,31], and the other is the V-H+-PPase, a single subunit protein that uses PPi instead of
ATP to transport protons.
Acidocalcisome: biogenesis
Acidocalcisome of eukaryotes is considered lysosome related organelles (LROs) like
platelets dense granules and mast cell granules. Human platelet dense granules contain
polyP and are similar to acidocalcisomes of bacteria and unicellular eukaryotes.
Polyphosphate released from platelets modulates blood coagulation and fibrinolysis. Mast
cell granules also have polyP, that is released and acts as a novel pro-inflammatory
regulator. Adaptor protein (AP) complexes are important mediators for vesicular
transport of membrane proteins between cellular compartments, such as Golgi complex,
endosomes, lysosomes, and plasma membrane [32]. AP-3 is involved in sorting of
proteins to lysosomes and LROs from the Golgi or from endosomes. Knockdown of the
β3 or δ subunits of the AP-3 complex led to a decrease in the number of acidocalcisomes
in both procyclic (PCF) and bloodstream forms of T. brucei [33].
Functional roles
Storage of phosphorus compounds (Pi, PPi, and polyP) and cations (calcium, magnesium,
sodium, potassium, zinc, and iron) is one of the main roles of acidocalcisomes from

12
different protists. This storage in an intracellular compartment reduces the osmotic effect
of large pools of these compounds in the cytosol. The recent discovery that polyP has
critical roles in blood clotting [34], and inflammation [35] suggests that polyP present in
microorganisms could be involved in their pathogenicity. Decrease in the levels of polyP
in parasites such as T. brucei, T. gondii, or L. major (reviewed in [36]) reduces their
pathogenicity. It is not known whether this is due to osmotic fragility of the parasites as a
result of changes in polyP levels that impact their ability to grow in vivo, making the
immune response against them more successful, or to a role of polyP in modulating the
immune response directly.
The discovery of an inositol 1,4,5-trisphosphate receptor (IP3R) in acidocalcisomes of T.
brucei [37] indicates that these organelles have a significant role in Ca2+
signaling. Ca2+
release via IP3Rs stimulates activities critical for life.
Acidocalcisomes also appear to have a role in regulation of intracellular pH.
Acidocalcisomes have also an important role in osmoregulation. There is rapid hydrolysis
or synthesis of acidocalcisome polyP during hypo- or hyperosmotic stress, respectively,
in T. cruzi [38], as well as changes in sodium and chloride content in acidocalcisomes of
L. major in response to acute hyposmotic stress [39]. It has been proposed that the
stimulus of cell swelling causes a spike in intracellular cAMP through an as yet
unidentified adenylyl cyclase, which causes aquaporin (TcAQP1) containing
acidocalcisome to fuse with the contractile vacuole and translocation of aquaporin [13].
This process helps the elimination of water by the contractile vacuole.

13
Traffic in trypanosomes
Trypanosomes appear to have a less complicated trafficking pathway in comparison to
eukaryotes, partly due to their unicellular structure and also due to a reduction in the copy
number of organelles in comparison to multicellular organisms. Trypanosomes have an
elongated shape, with the presence of tightly spaced subpellicular microtubules
subtending the plasma membrane. Endocytic and exocytic trafficking is restricted to the
posterior flagellar pocket (FP). It sometimes also occur at areas of the plasma membrane
where the cell cytoskeleton, formed by sub-pellicular microtubules, is absent.
Endocytosis in T. cruzi also occurs through the cytostome, present in both epimastigotes
and amastigotes.
One of the surface proteins whose traffic has been studied in T. brucei is the GPI-
anchored Variant Surface Glycoprotein (VSG), which is responsible for antigenic
variation in them. VSG is a major secretory cargo of T. brucei bloodstream forms, which
is trafficked to the surface; from where it is endocytosed and recycled via the flagellar
pocket [40], [41]. Secretory cargos leave the ER from defined ER exit sites (ERES)
where they are loaded into COPII secretory vesicles [42]. Though the post-Golgi
trafficking pathway is not very clear, it is known that cargo is destined either for the
lysosome or the cell surface. Some players which belong to the Rab family of proteins
responsible for vesicular fusion have been identified. These include TbRab5A/B (early
endosome), TbRab11 (recycling endosome), and TbRab7 (late endosome) (reviewed in
[43]). The pathway from the post-Golgi to the lysosome, or the flagellar pocket or to the
cell surface needs to be delineated. There are some basic similarities between the
secretory pathways of trypanosomes with model organisms like yeast or vertebrate cells,

14
but there are some defined differences as well. Nevertheless, because of their streamlined
architecture they offer unique opportunities to study general eukaryotic cell biology.
Endocytosis is rapid in T. brucei, probably because of the phenomenon of immune
evasion of this parasite. Clathrin-mediated mechanisms are the major route for
endocytosis in T. brucei and GPI-anchored proteins are endocytosed by clathrin-
dependent pathways in trypanosomes [44].
The mechanisms involved in exocytosis, endocytosis and recycling in T. cruzi are poorly
understood compared to mammalian cells or to the related organism T. brucei. Most of
what is known comes from structural and biochemical studies with regard to enzymes
and endocytic markers, as will be discussed below. T. cruzi ingests nutrients from the
environment by endocytosis, but the endocytic pathway and molecules/organelles
involved in this important metabolic pathway are still poorly known. Data on fluid-phase
pinocytosis of peroxidase and on receptor-mediated endocytosis of gold-labeled albumin,
peroxidase, transferrin and LDL [45] by T. cruzi showed that the ingested material
entered the cells through the cytostome and/or the flagellar pocket region [46,47]. Both
sites open at the anterior cell end, where the single flagellum emerges. Endocytosis of
transferrin-gold nanoparticles has been studied by confocal microscopy [48]. But unlike
T. brucei, endocytosis is mostly clathrin-independent in T. cruzi. In an attempt to identify
the compartments involved in endocytosis in T. cruzi, it has been found that ingested
material concentrates in the reservosome, an acidic pre-lysosomal compartment in the
posterior end of the cell, rich in cysteine proteinase, but which does not contain acid
phosphatase or other lysosomal membrane proteins [49].

15
Rab proteins
Proteomic and bioinformatics analyses of proteins localized to the CVC identified several
proteins with trafficking roles [11]. Among them, two Rab (Ras-related proteins in the
brain) GTPases (Rab32 and Rab11) were identified, which are the subject of my research.
Rab proteins are members of the highly evolutionarily conserved Rab superfamily of
GTPases that are structurally related to the Ras proteins. They regulate different
intracellular transport processes. Other members of the Ras superfamily such as Rho, Rab
and Ran proteins, are regulated by similar interactions with nucleotides. However, they
interact with distinct regulators and downstream target proteins, allowing them to
contribute to unique cellular functions (Fig. 2.3). The related regions include at least four
protein domains found in all GTPases that are involved in the binding of GTP or GDP
[50]. When Rabs, are in the GTP-bound state, they are thought to be functionally active
and are inactive when they bind GDP [50]. Conversion of the GDP-bound Rab into the
GTP-bound form occurs through the exchange of GDP for GTP, which is catalyzed by a
guanine nucleotide exchange factor (GEF) and causes a conformational change (Fig. 2.4).
The GTP-bound ‘active’ conformation is recognized by multiple effector proteins and is
converted back to the GDP-bound ‘inactive’ form through hydrolysis of GTP, which is
stimulated by a GTPase-activating protein (GAP) and releases an inorganic phosphate
(Pi). The newly synthesized Rab, in the GDP-bound form, is recognized by a Rab escort
protein (REP). The REP presents the Rab to a geranylgeranyl transferase (GGT), which
geranylgeranylates the Rab on one or two carboxy-terminal Cys residues. The
geranylgeranylated, GDP-bound Rab is recognized by Rab GDP dissociation inhibitor
(GDI), which regulates the membrane cycle of the Rab. Targeting of the Rab–GDI

16
complex to specific membranes is mediated by interaction with a membrane-bound GDI
displacement factor (GDF) that catalyzes the dissociation of Rab-GDI complex at
particular membrane surfaces. Coordinated regulation of Rab proteins is instrumental in
ensuring precision and fidelity of membrane trafficking. Accumulated evidence suggests
that Rab GTPases recruit tethering and docking factors to establish firm contact between
the membranes to fuse, after which SNAREs (Soluble NSF Attachment Protein Receptor)
become involved and complete the fusion process [51,52]. Crystallographic structure of
Rab proteins have been identified which include structural motifs and modes of effector
interaction that are distinct from those of other GTPase families. The active conformation
(GTP-bound) is stabilized by additional hydrogen bonding i
phosphate of GTP, mediated by serine residues in the P-loop and switch I region, as well
as an extensive hydrophobic interface between the switch I and II regions [53,54].
Besides the presence of a hydrophobic triad (residues Phe-58, Trp-75, and Tyr-90) leads
to a structural flexibility, thus contributing to the mechanism by which different Rabs
interact with their specific subset of effector proteins.
A total of 17 Rab proteins have been identified in T. cruzi. In addition to Rab32 and
Rab11, only three other Rab proteins: Rab4, Rab5 and Rab7 were studied in T. cruzi [55-
57]. The lack of genetic tools in T.cruzi prevented investigation regarding the mechanism
of function of these Rabs.
Tools to investigate the function of Rab proteins in vesicle fusion and transport
mechanism
There are several tools available to study the localization and function of Rab proteins in
mammalian cells and to study the involvement of Rab isoforms in specialized membrane

17
trafficking events [58]. The tools include study of enhanced green fluorescent protein
(EGFP)-tagged mouse and human Rabs, FLAG-tagged Rabs, glutathione S-transferase
(GST)-tagged Rabs, Gal4-binding domain (GBD)-tagged Rabs, Tre-2/Bub2/Cdc16
(TBC) domain-containing Rab-GTPase activating proteins (GAPs), and small interfering
RNAs. EGFP-Rabs are used to screen for Rabs that are localized on specific organelles
and regulate their transport, and GST-Rabs and GBD-Rabs are used to screen for novel
Rab effectors by GST pull-down assays and yeast two-hybrid assays, respectively.
Several methods have often been used to investigate the function of specific Rab
isoforms in membrane traffic. The first, and most commonly used method, has been
overexpression in cells of a constitutive active (CA) mutant that mimics the GTP-bound
form or of a constitutive negative (CN) mutant that mimics the GDP-bound form (Fig.
2(a)). The second method, which has come into use recently, is knockdown of a specific
Rab by RNA interference technology. The third method is based on a genetic approach in
which a specific Rab effector domain is overexpressed in cells. As the effectors that bind
to Rab proteins and their binding domains have not been studied in detail, the third
method has severe limitations. Since Rab-GAP is able to inactivate its substrate Rab by
promoting GTPase activity, overexpression of Rab-GAP in cells should result in specific
inactivation of its substrate Rab, which, in turn, would inhibit specific organelle transport.
Although the specific Rab-GAP of most mammalian Rabs has yet to be identified, a TBC
domain is generally thought to function as a Rab-GAP. Although TBC/Rab-GAP proteins
are useful for inactivating the function of endogenous Rab proteins, the results need to be
interpreted carefully based on the specificity of some TBC/Rab-GAPs (e.g.,[59]).
Unfortunately the RNAi machinery is absent in T.cruzi [23]. Hence, expression of the CN

18
or CA form that mimics loss-of-function or gain-of function effects was used for our
research. .
GDP bound “OFF” stage of Rab proteins: examples
Dominant-negative Rab mutants work in cells by competing with endogenous Rabs for
binding to Rab-GEFs. The mutants cannot interact with downstream target proteins
within cells, so when they are expressed in cells in excess they bind to GEFs and form
‘dead-end’ complexes. Thus sequestration of Rab-GEFs prevents the activation of
endogenous Rabs [60]. Biological experiments supporting this view [61] have shown that
the growth-inhibitory effect of Ras17N expression in mammalian cells, or of Ras15A
expression in yeast, can be overcome by increased expression of either a Ras-specific
GEF or wild-type Ras. In addition, mutations within the region of Ras that interacts with
GEFs suppress the inhibitory phenotype of Ras17N.
Role of Rab32 protein in trafficking
Different Rab-GTPases localize to different organelles which gives every organelle a
unique identity. Rab32 has been shown to regulate post-Golgi trafficking of melanogenic
enzymes in mammalian cells [62] and melanosome transport and melanocyte biogenesis
in Xenopus laevis [63]. It is known to regulate pigmentation, but it is not directly required
for the formation of melanosomes [62]. Rab32 has also been shown to regulate
phagosome maturation along with a network of other Rab GTPases [64]. It is required for
the formation of autophagic vacuoles and is involved in regulation of the clearance of
aggregated proteins by autophagy in a nucleotide binding state dependent manner [65].
Human Rab32 expressed in COS cells localizes to mitochondria as an A-kinase

19
anchoring protein (AKAP), and the expression of its GDP-bound form causes the
fragmentation of mitochondria [66].
No function of Rab32 has yet been reported in trypanosomes, although acidocalcisomes,
as melanosomes, are lysosome-related organelles [67]. Interestingly, Rab32 was found in
both granule and membrane fractions from human platelets [68]. Platelet dense granules
are the most similar to acidocalcisomes in that they contain PPi and polyP and are rich in
calcium (reviewed in [23]).
In this dissertation (Chapter 4) I study if the function of TcRab32 is conserved in T. cruzi,
by regulating function of acidocalcisomes. TcRab32 has the “DIAGQ” domain that is
present in Rab32 across all species. A similar replacement is found in Rab38, Rab29, and
Rab7L1/29 of mammalian cells, and in RabE from Dictyostelium discoideum [65], but
there are no orthologs to any of these other Rabs in T. cruzi.
Role of Rab11 protein in trafficking
Rab11 is one of the best studied Rab-GTPases, other than Rab5. Rab11 regulates
exocytic and recycling processes, thereby directing proteins and membranes towards the
cell surface. Rab11 generally localizes to the trans-Golgi as well as post-Golgi
endosomes of secretory pathway [69]. Rab11 has been shown to regulate traffic of
several receptors and adhesion proteins which have roles in cell-cell adhesion, migration
and invasion; with diverse cellular functions including ciliogenesis, cytokinesis,
neuritogenesis, and oogenesis [70-73]. This high degree of functional complexity is
achieved by mutually exclusive recruitment of a range of Rab11 effector proteins
In T. brucei, Rab11 localizes to the recycling endosomes [74]. It mediates the transfer of
the glycosylphosphatidylinositol (GPI)-anchored proteins transferrin [75] and variant

20
surface glycoprotein (VSG) [76] to the plasma membrane. Rab11 depletion inhibited
export, but not uptake, of internalized transferrin, thus implying its involvement in
secretion pathway [77]. Besides, Rab11 localizes to the CVC of D. discoideum [78]. Our
observation [11] that Rab11 localizes to the CVC in T. cruzi suggested that an
uncharacterized membrane transport exists connecting the CVC to the plasma membrane.
That is the subject of Chapter 3 of my dissertation.
Rab protein prenylation and potential treatment of Chagas disease:
Protein prenylation is a post–translational modification that occurs in many eukaryotic
cells which functions to bind proteins to cell membrane and they may direct protein-
protein interactions and thus are needed for many biological activities. Among the many
prenylated proteins Rabs form a distinct class. The C-terminus of Ras superfamily
GTPases terminates in a so-called CAAX box (where C is cysteine, A is usually but not
necessarily an aliphatic amino acid, and X is a variety of different amino acids). The
CAAX box serves as a signal for a series of post-translational modifications: 1)
farnesylation or geranylgeranylation of the cysteine sulfhydryl group, 2) endoproteolytic
removal of AAX, and 3) methylation of the -carboxyl group of the prenylated cysteine
residue. The hydrophobic C termini of Ras superfamily GTPases are thought to be
important for anchoring these proteins to cellular membranes [79] [80]. The three
structural classes of prenylation that have been identified are C-terminal farnesylation, C-
terminal geranylation and C-terminal digeranylgeranylation. It involves transfer of a 15-
carbon farnesyl or a 20-carbon geranylgeranyl from the corresponding prenyl-
pyrophosphate to the sulfhydryl group of the carboxyl-terminal cysteine, respectively
[81] [28]. Since prenylation is required for the function of important regulators of cell

21
growth, inhibitors of these enzymes are likely to have therapeutic potential for the
treatment of parasitic diseases. The fact that growth of T. brucei, T. cruzi, and L.
mexicana is blocked by protein farnesyl transferase (PFT) inhibitors suggests that
trypanosomatid PFT is a good target for treating sleeping sickness, Chagas disease, and
leishmaniasis In addition the mechanism of action of bisphosphonates involves the
inhibition of the enzyme farnesyl pyrophosphate synthase, thereby preventing the
prenylation of small GTPase signaling proteins, suggesting that they can be used to treat
parasitic diseases [82].
Overview of Trypanosoma cruzi infection:
Adhesion of T. cruzi to Vertebrate Cells
The first steps of the T. cruzi-host cell interaction process can be divided into three
stages: adhesion and recognition, signaling, and invasion. Invasion depends on the T.
cruzi strain and which developmental stage is used, the morphology of the
trypomastigote, whether slender or stout, and which host cell it is invading, as reviewed
in [83]. The mechanisms by which T. cruzi infective forms gain access to the intracellular
milieu are still being studied. The adhesion step involves the recognition of molecules
present on the surface of both parasite and host cells (Figure 2.4). T. cruzi, need to escape
their vacuole and instead replicate in the host cell cytosol. This vacuolar escape is the
first step of egress, which needs to be perfectly controlled in order to lyse the vacuole but
preserve host cell integrity. After replication, a second egress event then leads to the
release of the progeny from the host cell. Importantly, both steps need to be individually
regulated. This illustrates that the completion of replication must play a central role in
triggering egress for vacuolar as well as cytosolic pathogens. The timing is likely

22
controlled by intrinsic cues to optimize the number of progeny to be released and to
ensure that the replication and maturation of the transmission forms have been completed
Parasite Molecules
Different strains of T. cruzi as well as different forms of the parasite (tissue culture
derived trypomastigotes, metacyclic trypomastigotes and amastigotes), express different
molecules on their surface. These surface molecules interact with host components to
invade mammalian cells. Some of these surface antigens central to our study have been
discussed below.
Host cell molecules
One class of receptors present in mammalian cells is represented by lectin-like molecules.
Lectins are sugar-binding proteins which are highly specific for their sugar moieties and
are involved in attachment between pathogens and host cells [84]. Carbohydrate residues
present in the plasma membrane of mammalian cells can function as receptors. Studies
show galactosyl, mannosyl and sialyl residues play a role in parasite internalization [85].
Integrins, receptors that mediate attachment between two cells or cell and extracellular
matrix, are involved in the invasion processes [86]. Another molecule present on the host
cell surface and involved in trypomastigotes’ entry is the TGF receptor [87].
Model of T. cruzi invasion
As reviewed in [83] the model indicates three distinct mechanisms of T. cruzi entry into
host cell (Fig. 2.5). (a) The lysosome dependent pathway is initiated by targeted Ca2+
-
regulated exocytosis of lysosomes in the plasma membrane; (b) in the actin dependent
pathway trypomastigotes penetrate into a host cell through a plasma membrane expansion
that culminates in assembly of a parasitophorous vacuole. Either early endosomes or

23
lysosomes can fuse with the parasitophorous vacuole; (c) in the lysosome-independent
pathway, parasites enter cells through plasma membrane invaginations that accumulate
PIP3 (product of class I PI3K activation). Subsequently, internalized parasites are
contained in a vacuole formed from the plasma membrane that maturates with the
acquisition of early endosome markers (Rab5 and EEA1) and subsequently with the
acquisition of lysosome markers; the trypomastigote forms gradually transform into an
amastigote form with simultaneous lysis of the parasitophorous vacuole membrane. Then,
amastigotes in direct contact with the cytoplasm start to divide.
GPI-anchored surface proteins
Glycosylphosphatidylinositol (GPI)-anchoring is a common, relevant posttranslational
modification of eukaryotic surface proteins [88]. GPI-anchored proteins have been
postulated to serve diverse functions such as cell surface protection in protozoan
parasites, cell wall synthesis in yeast or cell adhesion and transmembrane signaling in
mammalian cells. GPI-anchored proteins are also the major cell surface molecules
expressed by the kinetoplastids; T. brucei, T. cruzi and Leishmania spp. Considering their
role in host cell invasion, protection from the host cell milieu, they are attractive targets
for drugs against parasitic diseases and for design of diagnostic probes [89,90].
GPI-anchored proteins are usually transported from the endoplasmic reticulum (ER) to
the plasma membrane through the Golgi apparatus, where lipid raft-like structures form
[91]. Sorting is achieved by the formation of domains rich in sphingolipids, cholesterol
and GPI-anchored proteins, specifically incorporated into vesicular carriers destined for
fusion with the plasma membrane. Though sorting is achieved mainly at the ER or the
Golgi, it can be achieved at several steps in the secretory pathway [92].

24
Trypanosomatids have an abundance of GPI-anchored surface molecules. T. brucei is
covered by a dense coat of GPI-anchored VSG protein. This primary secretory cargo is a
stage-specific protein expressed by T. brucei [93]. Only correctly folded GPI-anchored
VSG is able to reach the cell surface; GPI-deficient VSG is retained in the ER and later
degraded. In these parasites GPI-anchored homodimers are formed in the ER and reache
the flagellar pocket via the Golgi apparatus [94].
GPI-anchored surface proteins are expressed in all developmental stages of T. cruzi and
encoded by thousands of members of multigene families: mucins, mucin associated
surface proteins (MASP) [95] and members of the trans-sialidase family/gp85
glycoprotein [96,97] and metalloproteinase gp63. But, the traffic route taken by GPI-
anchored proteins and the carrier proteins are yet to be characterized in T. cruzi. This
topic is the aim of our study in Chapter 3.
Trans-sialidase
T. cruzi is unable to synthesize sialic acid and it depends on the host cell for it [98]. It is
achieved by the expression of trans-sialidase on its surface. This enzymatic activity is
different from the eukaryotic sialyltransferases present in the Golgi complex that
exclusively use CMP-sialic acid as the donor substrate. Trans-sialidase is
developmentally regulated in T. cruzi. The enzyme, located on the trypanosome surface,
is responsible for transferring sialyl residues from host glycoconjugates to parasite
molecules. Trans-sialidase is capable of directly transferring sialic acid residues between
a variety of molecules ([99] [100] [101]). TcTS is crucial in the life cycle of the parasite
because it allows the acquisition of sialyl residues from the host glycoconjugates
preventing their lysis by the alternative complement pathway [102,103], and opsonization

25
followed by killing by natural antibodies [104]. Trans-sialidases are important for neural,
glial and epithelial cell invasion through binding to the nerve growth factor receptors
[105,106], to prevent apoptosis during infection [107], and to trigger the appearance of
protective CD4+ and CD8
+ T cells [108]. It also enables the parasite to infect/attach cells
[101,109], and exit the parasitophorous vacuole [110]. Pereira and colleagues [104] using
trypomastigotes expressing trans-sialidases (TS+) and trypomastigotes that do not express
trans-sialidases (TS−) demonstrated that the TS
+ population was highly invasive, whereas
TS− was extremely inefficient to infect nonphagocytic cells.
TcTS is shed to the extracellular medium, including within the host cells [111], through
the action of an endogenous phospholipase C, and also with vesicles of the plasma
membrane [112]. The shed TcTS induces several hematological abnormalities and alters
the immune system [113], [114,115]. SAPA (Shed-Acute-Phase-Antigen) is a family of
three to six proteins of 160-200 kDa encoded by related genes which are mainly
expressed in the infective (trypomastigote) stage of the parasite [116]. The amino acid
sequence of SAPA as deduced from the DNA sequence showed that its C-terminal
portion contained a variable number of repeated units of 12 amino acids in length [117].
The SAPA N-terminal region contained two Ser-X-Asp-X-Gly-X-Thr-Trp motifs that are
conserved in bacterial and viral neuraminidases [118]. In addition, SAPA contained two
other of such motifs having three out of the five amino acids similar. These repetitive
motifs are readily detected by antibodies present in the sera from infected patients, thus
suggesting that they are major targets of the immune system.
The trans-sialidase displayed by the epimastigote (the parasite form present in the
reduviid vector) has a potential trans-membrane domain and is not released, even after

26
addition of exogenous phospholipase. But, the enzyme present in the trypomastigote (the
infective form of the parasite that circulates in the blood of the vertebrate host) is
anchored by a glycosylphosphatidylinositol (GPI) linkage to the T. cruzi surface and is
released into the environment [119].
TcTS genes are distributed in several families of which only one is composed by genes
encoding the active enzyme (TS) and its inactive isoform (iTS), which differs in only one
mutation (Tyr342His) [120]that completely abolishes its TS activity, but retains its
property to recognize terminal galactoses. The crystal structure of iTS has been
determined [121]. The 680 amino acids-amino terminal contains the catalytic activity.
The recombinant protein binds sialic acid and galactose in vitro and competes with a
neutralizing antibody to a discontinuous epitope of TS indicating that it is properly folded
[109].
Although TcTS has been known for several years, its structure has been solved and its
catalytic role been studied, our understanding of its trafficking is still limited. Many
biological roles have been attributed to TcTS in connection with Chagas disease; but due
to the lack of efficient inhibition, its direct effect on invasion had been difficult to study.
This dissertation delineates its traffic pathway and demonstrates the effect of TS on host
cell invasion (as addressed in Chapter 3).

27
REFERENCES
1. Hotez PJ, Dumonteil E, Heffernan MJ, Bottazzi ME (2013) Innovation for the 'bottom
100 million': eliminating neglected tropical diseases in the Americas. Adv Exp
Med Biol 764: 1-12.
2. Hotez PJ, Bottazzi ME, Franco-Paredes C, Ault SK, Periago MR (2008) The neglected
tropical diseases of Latin America and the Caribbean: a review of disease burden
and distribution and a roadmap for control and elimination. PLoS Negl Trop Dis
2: e300.
3. Gascon J, Bern C, Pinazo MJ (2010) Chagas disease in Spain, the United States and
other non-endemic countries. Acta Trop 115: 22-27.
4. Bern C, Kjos S, Yabsley MJ, Montgomery SP (2011) Trypanosoma cruzi and Chagas'
Disease in the United States. Clin Microbiol Rev 24: 655-681.
5. Rassi A, Jr., Rassi A, Marin-Neto JA (2010) Chagas disease. Lancet 375: 1388-1402.
6. Figarella K, Uzcategui NL, Zhou Y, LeFurgey A, Ouellette M, et al. (2007)
Biochemical characterization of Leishmania major aquaglyceroporin LmAQP1:
possible role in volume regulation and osmotaxis. Mol Microbiol 65: 1006-1017.
7. Linder JC, Staehelin LA (1979) A novel model for fluid secretion by the
trypanosomatid contractile vacuole apparatus. J Cell Biol 83: 371-382.
8. Baqui MM, De Moraes N, Milder RV, Pudles J (2000) A giant phosphoprotein
localized at the spongiome region of Crithidia luciliae thermophila. J Eukaryot
Microbiol 47: 532-537.
9. Allen RD, Naitoh Y (2002) Osmoregulation and contractile vacuoles of protozoa. Int
Rev Cytol 215: 351-394.
10. Docampo R, Jimenez V, Lander N, Li ZH, Niyogi S (2013) New insights into roles of
acidocalcisomes and contractile vacuole complex in osmoregulation in protists.
Int Rev Cell Mol Biol 305: 69-113.

28
11. Ulrich PN, Jimenez V, Park M, Martins VP, Atwood J, 3rd, et al. (2011)
Identification of contractile vacuole proteins in Trypanosoma cruzi. PLoS One 6:
e18013.
12. Montalvetti A, Rohloff P, Docampo R (2004) A functional aquaporin co-localizes
with the vacuolar proton pyrophosphatase to acidocalcisomes and the contractile
vacuole complex of Trypanosoma cruzi. The Journal of biological chemistry 279:
38673-38682.
13. Rohloff P, Montalvetti A, Docampo R (2004) Acidocalcisomes and the contractile
vacuole complex are involved in osmoregulation in Trypanosoma cruzi. The
Journal of biological chemistry 279: 52270-52281.
14. Figarella K, Uzcategui NL, Zhou Y, LeFurgey A, Ouellette M, et al. (2007)
Biochemical characterization of Leishmania major aquaglyceroporin LmAQP1:
possible role in volume regulation and osmotaxis. Molecular microbiology 65:
1006-1017.
15. Clarke M, Kohler J, Arana Q, Liu T, Heuser J, et al. (2002) Dynamics of the vacuolar
H+-ATPase in the contractile vacuole complex and the endosomal pathway of
Dictyostelium cells. J Cell Sci 115: 2893-2905.
16. Heuser J, Zhu Q, Clarke M (1993) Proton pumps populate the contractile vacuoles of
Dictyostelium amoebae. J Cell Biol 121: 1311-1327.
17. Moniakis J, Coukell MB, Janiec A (1999) Involvement of the Ca2+
-ATPase PAT1
and the contractile vacuole in calcium regulation in Dictyostelium discoideum. J
Cell Sci 112 ( Pt 3): 405-414.
18. Sesaki H, Wong EF, Siu CH (1997) The cell adhesion molecule DdCAD-1 in
Dictyostelium is targeted to the cell surface by a nonclassical transport pathway
involving contractile vacuoles. The Journal of cell biology 138: 939-951.
19. Sriskanthadevan S, Lee T, Lin Z, Yang D, Siu CH (2009) Cell adhesion molecule
DdCAD-1 is imported into contractile vacuoles by membrane invagination in a
Ca2+
- and conformation-dependent manner. The Journal of biological chemistry
284: 36377-36386.

29
20. Niyogi S, Mucci J, Campetella O, Docampo R (2014) Rab11 regulates trafficking of
trans-sialidase to the plasma membrane through the contractile vacuole complex
of Trypanosoma cruzi. PLoS Pathog 10: e1004224.
21. Hasne MP, Coppens I, Soysa R, Ullman B (2010) A high-affinity putrescine-
cadaverine transporter from Trypanosoma cruzi. Molecular microbiology 76: 78-
91.
22. Macro L, Jaiswal JK, Simon SM (2012) Dynamics of clathrin-mediated endocytosis
and its requirement for organelle biogenesis in Dictyostelium. J Cell Sci 125:
5721-5732.
23. Docampo R (2011) Molecular parasitology in the 21st century. Essays Biochem 51:
1-13.
24. Docampo R, de Souza W, Miranda K, Rohloff P, Moreno SN (2005)
Acidocalcisomes - conserved from bacteria to man. Nat Rev Microbiol 3: 251-
261.
25. Rohloff P, Rodrigues CO, Docampo R (2003) Regulatory volume decrease in
Trypanosoma cruzi involves amino acid efflux and changes in intracellular
calcium. Mol Biochem Parasitol 126: 219-230.
26. Rao NN, Gomez-Garcia MR, Kornberg A (2009) Inorganic polyphosphate: essential
for growth and survival. Annu Rev Biochem 78: 605-647.
27. Docampo R, Scott DA, Vercesi AE, Moreno SN (1995) Intracellular Ca2+ storage in
acidocalcisomes of Trypanosoma cruzi. Biochem J 310 ( Pt 3): 1005-1012.
28. Cuevas IC, Rohloff P, Sanchez DO, Docampo R (2005) Characterization of
farnesylated protein tyrosine phosphatase TcPRL-1 from Trypanosoma cruzi.
Eukaryot Cell 4: 1550-1561.
29. Scott DA, Docampo R, Dvorak JA, Shi S, Leapman RD (1997) In situ compositional
analysis of acidocalcisomes in Trypanosoma cruzi. J Biol Chem 272: 28020-
28029.
30. Ruiz FA, Marchesini N, Seufferheld M, Govindjee, Docampo R (2001) The
polyphosphate bodies of Chlamydomonas reinhardtii possess a proton-pumping
pyrophosphatase and are similar to acidocalcisomes. J Biol Chem 276: 46196-
46203.

30
31. Yagisawa F, Nishida K, Yoshida M, Ohnuma M, Shimada T, et al. (2009)
Identification of novel proteins in isolated polyphosphate vacuoles in the
primitive red alga Cyanidioschyzon merolae. Plant J 60: 882-893.
32. Besteiro S, Tonn D, Tetley L, Coombs GH, Mottram JC (2008) The AP3 adaptor is
involved in the transport of membrane proteins to acidocalcisomes of Leishmania.
Journal of cell science 121: 561-570.
33. Huang G, Fang J, Sant'Anna C, Li ZH, Wellems DL, et al. (2011) Adaptor protein-3
(AP-3) complex mediates the biogenesis of acidocalcisomes and is essential for
growth and virulence of Trypanosoma brucei. The Journal of biological chemistry
286: 36619-36630.
34. Smith SA, Mutch NJ, Baskar D, Rohloff P, Docampo R, et al. (2006) Polyphosphate
modulates blood coagulation and fibrinolysis. Proceedings of the National
Academy of Sciences of the United States of America 103: 903-908.
35. Muller F, Mutch NJ, Schenk WA, Smith SA, Esterl L, et al. (2009) Platelet
polyphosphates are proinflammatory and procoagulant mediators in vivo. Cell
139: 1143-1156.
36. Docampo R, Moreno SN (2011) Acidocalcisomes. Cell calcium 50: 113-119.
37. Huang G, Bartlett, P.D., Thomas, A.P., Moreno, S.N.J., Docampo, R. (2013)
Acidocalcisomes of Trypanosoma brucei have an inositol 1,4,5-trisphosphate
receptor that is required for growth and infectivity. Proc Natl Acad Sci USA in
press.
38. Li ZH, Alvarez VE, De Gaudenzi JG, Sant'Anna C, Frasch AC, et al. (2011)
Hyperosmotic stress induces aquaporin-dependent cell shrinkage, polyphosphate
synthesis, amino acid accumulation, and global gene expression changes in
Trypanosoma cruzi. The Journal of biological chemistry 286: 43959-43971.
39. LeFurgey A, Ingram P, Blum JJ (2001) Compartmental responses to acute osmotic
stress in Leishmania major result in rapid loss of Na+ and Cl. Comparative
biochemistry and physiology Part A, Molecular & integrative physiology 128:
385-394.

31
40. Engstler M, Thilo L, Weise F, Grunfelder CG, Schwarz H, et al. (2004) Kinetics of
endocytosis and recycling of the GPI-anchored variant surface glycoprotein in
Trypanosoma brucei. J Cell Sci 117: 1105-1115.
41. Bangs JD, Andrews NW, Hart GW, Englund PT (1986) Posttranslational
modification and intracellular transport of a trypanosome variant surface
glycoprotein. J Cell Biol 103: 255-263.
42. Hughes H, Stephens DJ (2008) Assembly, organization, and function of the COPII
coat. Histochem Cell Biol 129: 129-151.
43. Ackers JP, Dhir V, Field MC (2005) A bioinformatic analysis of the RAB genes of
Trypanosoma brucei. Mol Biochem Parasitol 141: 89-97.
44. Allen CL, Goulding D, Field MC (2003) Clathrin-mediated endocytosis is essential in
Trypanosoma brucei. Embo j 22: 4991-5002.
45. Soares MJ, de Souza W (1991) Endocytosis of gold-labeled proteins and LDL by
Trypanosoma cruzi. Parasitol Res 77: 461-468.
46. Rocha GM, Seabra SH, de Miranda KR, Cunha-e-Silva N, de Carvalho TM, et al.
(2010) Attachment of flagellum to the cell body is important to the kinetics of
transferrin uptake by Trypanosoma cruzi. Parasitol Int 59: 629-633.
47. Souza W (2009) Structural organization of Trypanosoma cruzi. Mem Inst Oswaldo
Cruz 104 Suppl 1: 89-100.
48. Eger I, Soares MJ (2012) Endocytosis in Trypanosoma cruzi (Euglenozoa:
Kinetoplastea) epimastigotes: visualization of ingested transferrin-gold
nanoparticle complexes by confocal laser microscopy. J Microbiol Methods 91:
101-105.
49. Soares MJ, Souto-Padron T, De Souza W (1992) Identification of a large pre-
lysosomal compartment in the pathogenic protozoon Trypanosoma cruzi. J Cell
Sci 102 ( Pt 1): 157-167.
50. Peter F, Nuoffer C, Pind SN, Balch WE (1994) Guanine nucleotide dissociation
inhibitor is essential for Rab1 function in budding from the endoplasmic
reticulum and transport through the Golgi stack. J Cell Biol 126: 1393-1406.
51. Pfeffer S, Aivazian D (2004) Targeting Rab GTPases to distinct membrane
compartments. Nat Rev Mol Cell Biol 5: 886-896.

32
52. Stenmark H (2009) Rab GTPases as coordinators of vesicle traffic. Nat Rev Mol Cell
Biol 10: 513-525.
53. Merithew E, Hatherly S, Dumas JJ, Lawe DC, Heller-Harrison R, et al. (2001)
Structural plasticity of an invariant hydrophobic triad in the switch regions of Rab
GTPases is a determinant of effector recognition. J Biol Chem 276: 13982-13988.
54. Dumas JJ, Zhu Z, Connolly JL, Lambright DG (1999) Structural basis of activation
and GTP hydrolysis in Rab proteins. Structure 7: 413-423.
55. Ramos FP, Araripe JR, Urmenyi TP, Silva R, Cunha e Silva NL, et al. (2005)
Characterization of RAB-like4, the first identified RAB-like protein from
Trypanosoma cruzi with GTPase activity. Biochem Biophys Res Commun 333:
808-817.
56. Araripe JR, Ramos FP, Cunha e Silva NL, Urmenyi TP, Silva R, et al. (2005)
Characterization of a RAB5 homologue in Trypanosoma cruzi. Biochem Biophys
Res Commun 329: 638-645.
57. Araripe JR, Cunha e Silva NL, Leal ST, de Souza W, Rondinelli E (2004)
Trypanosoma cruzi: TcRAB7 protein is localized at the Golgi apparatus in
epimastigotes. Biochem Biophys Res Commun 321: 397-402.
58. Fukuda M (2010) How can mammalian Rab small GTPases be comprehensively
analyzed?: Development of new tools to comprehensively analyze mammalian
Rabs in membrane traffic. Histol Histopathol 25: 1473-1480.
59. Cuif MH, Possmayer F, Zander H, Bordes N, Jollivet F, et al. (1999) Characterization
of GAPCenA, a GTPase activating protein for Rab6, part of which associates with
the centrosome. Embo j 18: 1772-1782.
60. Feig LA (1999) Tools of the trade: use of dominant-inhibitory mutants of Ras-family
GTPases. Nat Cell Biol 1: E25-27.
61. Chen SY, Huff SY, Lai CC, Der CJ, Powers S (1994) Ras-15A protein shares highly
similar dominant-negative biological properties with Ras-17N and forms a stable,
guanine-nucleotide resistant complex with CDC25 exchange factor. Oncogene 9:
2691-2698.

33
62. Wasmeier C, Romao M, Plowright L, Bennett DC, Raposo G, et al. (2006) Rab38 and
Rab32 control post-Golgi trafficking of melanogenic enzymes. J Cell Biol 175:
271-281.
63. Park M, Serpinskaya AS, Papalopulu N, Gelfand VI (2007) Rab32 regulates
melanosome transport in Xenopus melanophores by protein kinase a recruitment.
Curr Biol 17: 2030-2034.
64. Seto S, Tsujimura K, Koide Y (2011) Rab GTPases regulating phagosome maturation
are differentially recruited to mycobacterial phagosomes. Traffic 12: 407-420.
65. Hirota Y, Tanaka Y (2009) A small GTPase, human Rab32, is required for the
formation of autophagic vacuoles under basal conditions. Cell Mol Life Sci 66:
2913-2932.
66. Alto NM, Soderling J, Scott JD (2002) Rab32 is an A-kinase anchoring protein and
participates in mitochondrial dynamics. J Cell Biol 158: 659-668.
67. Moreno SN, Docampo R (2009) The role of acidocalcisomes in parasitic protists. J
Eukaryot Microbiol 56: 208-213.
68. Bao X, Faris AE, Jang EK, Haslam RJ (2002) Molecular cloning, bacterial expression
and properties of Rab31 and Rab32. Eur J Biochem 269: 259-271.
69. Urbe S, Huber LA, Zerial M, Tooze SA, Parton RG (1993) Rab11, a small GTPase
associated with both constitutive and regulated secretory pathways in PC12 cells.
FEBS Lett 334: 175-182.
70. Hobdy-Henderson KC, Hales CM, Lapierre LA, Cheney RE, Goldenring JR (2003)
Dynamics of the apical plasma membrane recycling system during cell division.
Traffic 4: 681-693.
71. Knodler A, Feng S, Zhang J, Zhang X, Das A, et al. (2010) Coordination of Rab8 and
Rab11 in primary ciliogenesis. Proc Natl Acad Sci U S A 107: 6346-6351.
72. Shirane M, Nakayama KI (2006) Protrudin induces neurite formation by directional
membrane trafficking. Science 314: 818-821.
73. Schuh M (2011) An actin-dependent mechanism for long-range vesicle transport. Nat
Cell Biol 13: 1431-1436.

34
74. Jeffries TR, Morgan GW, Field MC (2001) A developmentally regulated rab11
homologue in Trypanosoma brucei is involved in recycling processes. J Cell Sci
114: 2617-2626.
75. Pal A, Hall BS, Jeffries TR, Field MC (2003) Rab5 and Rab11 mediate transferrin
and anti-variant surface glycoprotein antibody recycling in Trypanosoma brucei.
Biochem J 374: 443-451.
76. Grunfelder CG, Engstler M, Weise F, Schwarz H, Stierhof YD, et al. (2003)
Endocytosis of a glycosylphosphatidylinositol-anchored protein via clathrin-
coated vesicles, sorting by default in endosomes, and exocytosis via RAB11-
positive carriers. Mol Biol Cell 14: 2029-2040.
77. Hall BS, Smith E, Langer W, Jacobs LA, Goulding D, et al. (2005) Developmental
variation in Rab11-dependent trafficking in Trypanosoma brucei. Eukaryot Cell
4: 971-980.
78. Harris E, Yoshida K, Cardelli J, Bush J (2001) Rab11-like GTPase associates with
and regulates the structure and function of the contractile vacuole system in
Dictyostelium. J Cell Sci 114: 3035-3045.
79. Gelb MH (1997) Protein prenylation, et cetera: signal transduction in two dimensions.
Science 275: 1750-1751.
80. Glomset JA, Farnsworth CC (1994) Role of protein modification reactions in
programming interactions between ras-related GTPases and cell membranes.
Annu Rev Cell Biol 10: 181-205.
81. Nepomuceno-Silva JL, Yokoyama K, de Mello LD, Mendonca SM, Paixao JC, et al.
(2001) TcRho1, a farnesylated Rho family homologue from Trypanosoma cruzi:
cloning, trans-splicing, and prenylation studies. J Biol Chem 276: 29711-29718.
82. Raikkonen J, Taskinen M, Dunford JE, Monkkonen H, Auriola S, et al. (2011)
Correlation between time-dependent inhibition of human farnesyl pyrophosphate
synthase and blockade of mevalonate pathway by nitrogen-containing
bisphosphonates in cultured cells. Biochem Biophys Res Commun 407: 663-667.
83. de Souza W, de Carvalho TM, Barrias ES (2010) Review on Trypanosoma cruzi:
Host Cell Interaction. Int J Cell Biol 2010.

35
84. Ming M, Ewen ME, Pereira ME (1995) Trypanosome invasion of mammalian cells
requires activation of the TGF beta signaling pathway. Cell 82: 287-296.
85. Barbosa HS, de Meirelles Mde N (1992) Ultrastructural detection in vitro of WGA-,
RCA I-, and Con A-binding sites involved in the invasion of heart muscle cells by
Trypanosoma cruzi. Parasitol Res 78: 404-409.
86. Ciavaglia Mdo C, de Carvalho TU, de Souza W (1993) Interaction of Trypanosoma
cruzi with cells with altered glycosylation patterns. Biochem Biophys Res
Commun 193: 718-721.
87. Hall BS, Pereira MA (2000) Dual role for transforming growth factor beta-dependent
signaling in Trypanosoma cruzi infection of mammalian cells. Infect Immun 68:
2077-2081.
88. McConville MJ, Ferguson MA (1993) The structure, biosynthesis and function of
glycosylated phosphatidylinositols in the parasitic protozoa and higher
eukaryotes. Biochem J 294 ( Pt 2): 305-324.
89. Ferguson MA (1999) The structure, biosynthesis and functions of
glycosylphosphatidylinositol anchors, and the contributions of trypanosome
research. J Cell Sci 112 ( Pt 17): 2799-2809.
90. Guha-Niyogi A, Sullivan DR, Turco SJ (2001) Glycoconjugate structures of parasitic
protozoa. Glycobiology 11: 45r-59r.
91. Fujita M, Kinoshita T (2012) GPI-anchor remodeling: potential functions of GPI-
anchors in intracellular trafficking and membrane dynamics. Biochim Biophys
Acta 1821: 1050-1058.
92. Lemmon SK, Traub LM (2000) Sorting in the endosomal system in yeast and animal
cells. Curr Opin Cell Biol 12: 457-466.
93. Bangs JD (1998) Surface coats and secretory trafficking in African trypanosomes.
Curr Opin Microbiol 1: 448-454.
94. Grunfelder CG, Engstler M, Weise F, Schwarz H, Stierhof YD, et al. (2002)
Accumulation of a GPI-anchored protein at the cell surface requires sorting at
multiple intracellular levels. Traffic 3: 547-559.
95. Di Noia JM, Buscaglia CA, De Marchi CR, Almeida IC, Frasch AC (2002) A
Trypanosoma cruzi small surface molecule provides the first immunological

36
evidence that Chagas' disease is due to a single parasite lineage. J Exp Med 195:
401-413.
96. El-Sayed NM, Myler PJ, Bartholomeu DC, Nilsson D, Aggarwal G, et al. (2005) The
genome sequence of Trypanosoma cruzi, etiologic agent of Chagas disease.
Science 309: 409-415.
97. Freitas LM, dos Santos SL, Rodrigues-Luiz GF, Mendes TA, Rodrigues TS, et al.
(2011) Genomic analyses, gene expression and antigenic profile of the trans-
sialidase superfamily of Trypanosoma cruzi reveal an undetected level of
complexity. PLoS One 6: e25914.
98. Giorgi ME, de Lederkremer RM (2011) Trans-sialidase and mucins of Trypanosoma
cruzi: an important interplay for the parasite. Carbohydr Res 346: 1389-1393.
99. Previato JO, Andrade AF, Pessolani MC, Mendonca-Previato L (1985) Incorporation
of sialic acid into Trypanosoma cruzi macromolecules. A proposal for a new
metabolic route. Mol Biochem Parasitol 16: 85-96.
100. Zingales B, Carniol C, de Lederkremer RM, Colli W (1987) Direct sialic acid
transfer from a protein donor to glycolipids of trypomastigote forms of
Trypanosoma cruzi. Mol Biochem Parasitol 26: 135-144.
101. Schenkman S, Jiang MS, Hart GW, Nussenzweig V (1991) A novel cell surface
trans-sialidase of Trypanosoma cruzi generates a stage-specific epitope required
for invasion of mammalian cells. Cell 65: 1117-1125.
102. Tomlinson S, Pontes de Carvalho LC, Vandekerckhove F, Nussenzweig V (1994)
Role of sialic acid in the resistance of Trypanosoma cruzi trypomastigotes to
complement. J Immunol 153: 3141-3147.
103. Buscaglia CA, Campo VA, Frasch AC, Di Noia JM (2006) Trypanosoma cruzi
surface mucins: host-dependent coat diversity. Nat Rev Microbiol 4: 229-236.
104. Pereira-Chioccola VL, Acosta-Serrano A, Correia de Almeida I, Ferguson MA,
Souto-Padron T, et al. (2000) Mucin-like molecules form a negatively charged
coat that protects Trypanosoma cruzi trypomastigotes from killing by human anti-
alpha-galactosyl antibodies. J Cell Sci 113 ( Pt 7): 1299-1307.

37
105. de Melo-Jorge M, PereiraPerrin M (2007) The Chagas' disease parasite
Trypanosoma cruzi exploits nerve growth factor receptor TrkA to infect
mammalian hosts. Cell Host Microbe 1: 251-261.
106. Weinkauf C, Salvador R, Pereiraperrin M (2011) Neurotrophin receptor TrkC is an
entry receptor for Trypanosoma cruzi in neural, glial, and epithelial cells. Infect
Immun 79: 4081-4087.
107. Chuenkova MV, PereiraPerrin M (2009) Trypanosoma cruzi targets Akt in host cells
as an intracellular antiapoptotic strategy. Sci Signal 2: ra74.
108. Risso MG, Pitcovsky TA, Caccuri RL, Campetella O, Leguizamon MS (2007)
Immune system pathogenesis is prevented by the neutralization of the systemic
trans-sialidase from Trypanosoma cruzi during severe infections. Parasitology
134: 503-510.
109. Buschiazzo A, Muia R, Larrieux N, Pitcovsky T, Mucci J, et al. (2012)
Trypanosoma cruzi trans-sialidase in complex with a neutralizing antibody:
structure/function studies towards the rational design of inhibitors. PLoS Pathog
8: e1002474.
110. Rubin-de-Celis SS, Uemura H, Yoshida N, Schenkman S (2006) Expression of
trypomastigote trans-sialidase in metacyclic forms of Trypanosoma cruzi
increases parasite escape from its parasitophorous vacuole. Cell Microbiol 8:
1888-1898.
111. Frevert U, Schenkman S, Nussenzweig V (1992) Stage-specific expression and
intracellular shedding of the cell surface trans-sialidase of Trypanosoma cruzi.
Infect Immun 60: 2349-2360.
112. Bayer-Santos E, Aguilar-Bonavides C, Rodrigues SP, Cordero EM, Marques AF, et
al. (2013) Proteomic analysis of Trypanosoma cruzi secretome: characterization
of two populations of extracellular vesicles and soluble proteins. J Proteome Res
12: 883-897.
113. Mucci J, Hidalgo A, Mocetti E, Argibay PF, Leguizamon MS, et al. (2002)
Thymocyte depletion in Trypanosoma cruzi infection is mediated by trans-
sialidase-induced apoptosis on nurse cells complex. Proc Natl Acad Sci U S A 99:
3896-3901.

38
114. Tribulatti MV, Mucci J, Van Rooijen N, Leguizamon MS, Campetella O (2005) The
trans-sialidase from Trypanosoma cruzi induces thrombocytopenia during acute
Chagas' disease by reducing the platelet sialic acid contents. Infect Immun 73:
201-207.
115. Freire-de-Lima L, Alisson-Silva F, Carvalho ST, Takiya CM, Rodrigues MM, et al.
(2010) Trypanosoma cruzi subverts host cell sialylation and may compromise
antigen-specific CD8+ T cell responses. J Biol Chem 285: 13388-13396.
116. Macina RA, Affranchino JL, Pollevick GD, Jazin EE, Frasch AC (1989) Variable
number of repeat units in genes encoding Trypanosoma cruzi antigens. FEBS Lett
257: 365-368.
117. Pollevick GD, Affranchino JL, Frasch AC, Sanchez DO (1991) The complete
sequence of a shed acute-phase antigen of Trypanosoma cruzi. Mol Biochem
Parasitol 47: 247-250.
118. Roggentin P, Rothe B, Kaper JB, Galen J, Lawrisuk L, et al. (1989) Conserved
sequences in bacterial and viral sialidases. Glycoconj J 6: 349-353.
119. Agusti R, Couto AS, Campetella OE, Frasch AC, de Lederkremer RM (1997) The
trans-sialidase of Trypanosoma cruzi is anchored by two different lipids.
Glycobiology 7: 731-735.
120. Cremona ML, Campetella O, Sanchez DO, Frasch AC (1999) Enzymically inactive
members of the trans-sialidase family from Trypanosoma cruzi display beta-
galactose binding activity. Glycobiology 9: 581-587.
121. Oppezzo P, Obal G, Baraibar MA, Pritsch O, Alzari PM, et al. (2011) Crystal
structure of an enzymatically inactive trans-sialidase-like lectin from
Trypanosoma cruzi: the carbohydrate binding mechanism involves residual
sialidase activity. Biochim Biophys Acta 1814: 1154-1161.

39
FIGURES
Figure 2.1. Life cycle of T. cruzi. The insect vector becomes infected after taking a
parasite-containing blood meal from an infected human or animal. Two main forms are
present in the insect vector: epimastigotes undergo morphological and physiological
transformation in the midgut and differentiate into infective metacyclic trypomastigotes
in the hindgut of the insect; the parasite is transmitted to humans by infected blood
sucking insects which deposit trypomastigotes in their feces during feeding and two main
forms in the vertebrate host are: intracellular amastigotes and bloodstream
trypomastigotes. (Figure published in Docampo R. et al, 2013).

40
Figure 2.2 The CVC in T. cruzi epimastigotes. (A) Thin section of chemically fixed
epimastigote showing the CVC. Collapsed aspect of the spongiome is denoted by arrows.
(B) Thin section of a high pressure freeze substituted epimastigote showing the CV and
the interconnected tubules (arrows) that form the spongiome (Sp). (C) Virtual section
showing the CV docked to the flagellar pocket (FP) and the electron-dense region
between both structures (arrow and inset). (D) Virtual section and 3D model of the CVC
and flagellar pocket (FP) where a deformation in the FP was observed (black arrows) and
a tubule of the spongiome was connected to the central vacuole (white arrow). Scale
bars¼200 nm. (Figure published in Docampo R. et al, 2013)

41
Figure 2.3. Diagramatic representation of the enzymes and transporters tentatively
identified in the acidocalcisome of T. cruzi (Diagram from Docampo et al. 2011)

42
Figure 2.4. The GTP-GDP cycle of Rab-GTPases. The GDP-bound (Constitutive
Negative; CN), inactivated form of Rab is activated by specific GEF. The wild type form
(WT) which cycles between GDP and GTP-bound form of Rab is recruited to a specific
type of organelle/vesicle. WT promotes the transport of the organelle/vesicle by
interacting with specific effector molecules. The GTP bound (Constitutive Active; CA)
form is inactivated by GAP. (Diagram from Peter F. et al. 1994)

43
Figure 2.5. Schematic model summarizing the molecules involved on parasite-host
cell interaction process exposed on the surface of a host cell and in trypomastigotes
of T. cruzi. The recognition between parasite and mammalian host cell involves cross-
talk between numerous molecules present on the surface of both cell types; which is
instrumental for adhesion to precede before invasion. Out of the surface proteins present
in the cell surface of the trypomastigotes, we show results with trans-sialidase, Mucin,
GP35/50 in Chapter 3. (Diagram from de Souza et al. 2010).

44
Figure 2.6. Model of T. cruzi invasion. This diagram illustrates lysosome dependent, or
actin dependent or lysosome-independent invasion pathway (Diagram from de Souza et
al. 2010).

45
CHAPTER 3
RAB11 REGULATES TRAFFICKING OF TRANS-SIALIDASE TO THE
PLASMA MEMBRANE THROUGH THE CONTRACTILE VACUOLE
COMPLEX OF TRYPANOSOMA CRUZI
Sayantanee Niyogi, Juan Mucci, Oscar Campetella, Roberto Docampo. 2014.
PLOS Pathogens. June 26. 10(6): e1004224. doi:10.1371/journal.ppat.1004224.
Reprinted here with permission of publisher.

46
Abstract
Trypanosoma cruzi is the etiologic agent of Chagas disease. Although this is not a free-
living organism it has conserved a contractile vacuole complex (CVC) to regulate its
osmolarity. This obligate intracellular pathogen is, in addition, dependent on surface
proteins to invade its hosts. Here we used a combination of genetic and biochemical
approaches to delineate the contribution of the CVC to the traffic of
glycosylphosphatidylinositol (GPI)-anchored proteins to the plasma membrane of the
parasite and promote host invasion. While T. cruzi Rab11 (TcRab11) localized to the
CVC, a dominant negative (DN) mutant tagged with GFP (GFP-TcRab11DN) localized
to the cytosol, and epimastigotes expressing this mutant were less responsive to
hyposmotic and hyperosmotic stress. Mutant parasites were still able to differentiate into
metacyclic forms and infect host cells. GPI-anchored trans-sialidase (TcTS), mucins of
the 60-200 KDa family, and trypomastigote small surface antigen (TcTSSA II) co-
localized with GFP-TcRab11 to the CVC during transformation of intracellular
amastigotes into trypomastigotes. Mucins of the gp35/50 family also co-localized with
the CVC during metacyclogenesis. Parasites expressing GFP-TcRab11DN prevented
TcTS, but not other membrane proteins, from reaching the plasma membrane, and were
less infective as compared to wild type cells. Incubation of these mutants in the presence
of exogenous recombinant active, but not inactive, TcTS, and a sialic acid donor, before
infecting host cells, partially rescued infectivity of trypomastigotes. Taking together these
results reveal roles of TcRab11 in osmoregulation and trafficking of trans-sialidase to the
plasma membrane, the role of trans-sialidase in promoting infection, and a novel
unconventional mechanism of GPI-anchored protein secretion.

47
Author Summary
Several free-living protozoa possess a contractile vacuole complex (CVC) that protects
them from the hyposmotic environments where they live. Interestingly, the intracellular
parasite Trypanosoma cruzi, the etiologic agent of Chagas disease, has conserved a CVC
in all its developmental stages, where it has an osmoregulatory role under both
hyposmotic and hyperosmotic conditions. We found here that the CVC of T. cruzi has an
additional unconventional role in traffic of glycosylphosphatidylinositol (GPI)-anchored
proteins to the plasma membrane of the parasite. A combination of genetic and
biochemical approaches revealed the role of TcRab11, a protein localized to the CVC, in
traffic of trans-sialidase (TcTS), a GPI-anchored protein important for host cell invasion,
but not of other GPI-anchored proteins or integral membrane proteins, to the plasma
membrane. Demonstration of the role of TcTS in infection has been previously difficult
given the large number of genes encoding for this protein distributed through the genome
of the parasite. However, by constructing dominant negative TcRab11 we were able to
prevent traffic of TcTS to the plasma membrane and demonstrate its role in host invasion.
Introduction
The contractile vacuole complex (CVC) is an intracellular compartment with an
osmoregulatory role in different protists. This compartment has a bipartite structure,
consisting of a central vacuole or bladder and a surrounding loose network of tubules and
vesicles named the spongiome [1,2]. The CVC accumulates water through an aquaporin
[3-7] and expels it out of the cell through pores in the plasma membrane [1,2].
Trypanosoma cruzi, the etiologic agent of Chagas disease or American trypanosomiasis,
possesses a CVC [4,8,9] that is important for regulatory volume decrease (RVD) after

48
hyposmotic stress [4], as well as for shrinking of the cells when submitted to
hyperosmotic stress [10].
Besides its osmoregulatory role, the CVC of some protists is an acidic calcium store [11]
and has roles in calcium ion (Ca2+
) sequestration and excretion pathways [12-16], as well
as in transfer of some proteins to the plasma membrane [12,17,18]. Although it has been
indicated that there is no much mixing or “scrambling” of contractile vacuoles and
plasma membranes [19], transfer of membrane proteins from the CVC to the plasma
membrane has been observed. In Dictyostelium discoideum, the vacuolar proton ATPase
(V-H+-ATPase) and calmodulin (CaM) move to the plasma membrane when cells are
starved during stationary phase [17], and the Ca2+
-ATPase PAT1 moves to the plasma
membrane when cells are incubated at high Ca2+
concentrations [12]. Some luminal
proteins, such as the adhesins DdCAD-1 and discoidin-1 can also be targeted to the cell
surface via the CVC in D. discoideum [18,20]. In T. cruzi epimastigotes, the polyamine
transporter TcPOT1.1, which localizes to CVC-like structures, has also been reported to
appear in the plasma membrane when the culture medium is deficient in polyamines [21].
It is interesting to note that dajumin-GFP is trafficked to the CVC of D. discoideum via
the plasma membrane and is internalized by a clathrin-dependent mechanism, suggesting
that clathrin-mediated endocytosis may function as a back-up mechanism in case of
transfer of proteins from the CVC to the plasma membrane [22].
It is remarkable that Rab11, a GTPase that localizes in recycling endosomes in most cells
[23], including Trypanosoma brucei [24], localizes to the CVC of D. discoideum [25] and
T. cruzi [26], suggesting that it might have some function in trafficking of proteins from
the CVC to the plasma membrane, as recycling endosomes have. It was proposed [25]

49
that the CVC could be an evolutionary precursor to the recycling endosomal system in
other eukaryotes.
In T. brucei, Rab11 mediates the transfer of the glycosylphosphatidylinositol (GPI)-
anchored proteins transferrin [27] and variant surface glycoprotein (VSG) [28] to the
plasma membrane. T. cruzi is also rich in GPI-anchored proteins, among them the trans-
sialidase (TS)-like superfamily, which includes 1,430 gene members [29,30], and the
mucins, encoded by 500 to 700 genes distributed into three groups of which group III is
conformed by a single-copy gene named the trypomastigote small surface antigen (TSSA)
[31]. TcTS genes are actually distributed in several families of which only one is
composed by genes encoding the active enzyme (TS) and its inactive isoform (iTS),
which differs in only one mutation (Tyr342His) [32]. TcTS is crucial in the life cycle of
the parasite because it allows the acquisition of sialyl residues from the host
glycoconjugates preventing their lysis by the alternative complement pathway [33,34],
and opsonization followed by killing by natural antibodies [35]. It also enables the
parasite to infect/attach cells [36,37], and exit the parasitophorous vacuole [38]. The shed
TcTS induces several hematological abnormalities and alters the immune system [39-41].
Two major TcTSSA isoforms were originally recognized: TcTSSA I, present in TcI
parasite stocks, which are linked to the sylvatic cycle of the parasite, and TcTSSA II,
present in TcVI (previously TcIIe) isolates, which are linked to the more virulent strains
[31]. Since TcTSSA II is highly immunogenic it has been proposed as an immunological
marker for the most virulent T. cruzi types [31], and as an adhesin, engaging surface
receptor(s) and inducing signaling pathways in the host cell as a prerequisite for parasite
internalization [42]. Another group of GPI-anchored surface proteins is that formed by

50
the mucin family of 60-200 KDa proteins bearing several oligosaccharide chains and
present in tissue culture-derived trypomastigotes [43]. These T. cruzi O-linked
oligosaccharide-containing proteins are highly immunogenic under the conditions of
natural infection and are the targets for lytic anti-Gal antibodies [43-45]. Gp35/50 mucins
are also GPI-anchored glycoproteins rich in threonine and expressed in epimastigotes and
metacyclic forms of all T. cruzi isolates examined to date and are encoded by a large
multigene family [46]. Gp35/50 mucins are recognized by monoclonal antibodies 10D8
and 2B10 [47], which react with galactofuranose- and galactopyranose-containing
epitopes, respectively.
GPI-anchored proteins are usually transported from the endoplasmic reticulum (ER) to
the plasma membrane through the Golgi apparatus, where lipid raft-like structures form
[48]. In this work we demonstrate that TcTS, TcTSSA II, and other mucins are
transported to the plasma membrane of T. cruzi trypomastigotes through the CVC, which
also possesses lipid-raft like structures, and that expression of dominant-interfering
TcRab11 mutants altered their morphology, osmoregulation, traffic of TcTS to the
plasma membrane, and parasite infectivity. The results suggest the presence of a novel
unconventional mechanism of GPI-anchored protein transport to the cell surface of
eukaryotic cells.
Results
Localization of TcRab11 in different T. cruzi stages
In previous work we reported the N-terminal tagging of T. cruzi Rab11
(TcCLB.511407.60; TcRab11) with the green fluorescent protein (GFP) gene, and the
localization of GFP-TcRab11 to the bladder of the CVC of epimastigotes of T. cruzi [26].

51
Tagging with GFP was confirmed by western blot analysis [26]. Fig. 3.1A-C shows now
that GFP-TcRab11 localizes to the bladder of the CVC of epimastigotes, trypomastigotes,
and amastigotes. Fig. 3.1D shows the co-localization of GFP-TcRab11 with T. cruzi
aquaporin 1 (TcAQP1), a marker for the CVC [3,4]. These experiments were done after
submitting the cells to hyposmotic conditions, which increases the localization of
TcAQP1 to the CVC [4]. To confirm that the above results were not an artifact of protein
overexpression and/or mistargeting we also used affinity-purified antibodies against
TbRab11 [24] (Fig. 3.1E and 3.1F). This antibody was shown to predominantly react
with a protein of 24 kDa in all T. cruzi stages, as expected for TcRab11 (Fig. 3.1G).
TcRab11 is apparently less expressed in epimastigotes. Fig. 3.11 confirms the CVC
localization of GFP-TcRab11 in epimastigotes submitted to hyposmotic stress by cryo-
immunogold electron microscopy.
Localization of GFP-TcRab11DN mutant
Knockdown of Rabs by RNA interference (RNAi) is one of the preferred approaches to
investigate the function of specific Rab isoforms in membrane traffic [49]. Unfortunately,
T. cruzi lacks an RNAi system [50]. To perform a functional analysis of TcRab11, we
therefore developed an expression plasmid encoding a TcRab11 mutant that mimics the
GDP-bound form (dominant negative). An N-terminal GFP epitope tag was fused to the
T. cruzi point mutant TcRab11:S21N. TcRab11:S21N is predicted to bind GDP, based
upon homology to known Ras-related protein mutations [51]. In transfected T. cruzi
epimastigotes, GFP-TcRab11DN had a punctated cytosolic localization (Fig. 3.2A). This
localization was maintained when epimastigotes were differentiated into trypomastigotes
(Fig. 3.2B) and intracellular amastigotes (Fig. 3.2C). This localization is because the

52
dominant negative TcRab11 (GDP-bound) gets locked in an intermediate cytosolic
location. After membrane delivery by the GDP dissociation inhibitor (GDI), Rab proteins
interconvert between inactive, GDP-bound forms and active, GTP-bound forms [52]. The
growth rate of the mutant epimastigotes was not affected (Fig. 3.12A). We confirmed
tagging of the mutant by western blot analysis (Fig. S2B). Together these results suggest
that TcRab11 is localized to the membrane of the CVC in a GTP-dependent manner.
Densitometry analysis indicated that GFP-TcRab11 expression increased 5.2 fold
compared to that in wild type epimastigotes (Fig 3.12C). We also investigated whether
the dominant negative mutation of TcRab11 disrupted the structure and assembly of the
CVC. We did immunofluorescence studies on GFP-TcRab11DN mutant epimastigotes
using an antibody against T. cruzi aquaporin 1, a CVC marker [4]. The same aquaporin
distribution was observed in epimastigotes expressing the control GFP-TcRab11 (Fig.
3.13A) and the mutant GFP-TcRab11DN (Fig. 3.13B). The CVC can be identified in Fig.
3.13A and 3.13B because of its curvature and its location close to the kinetoplast. There
was a greater concentration of TcAQP1 in the CVC with some punctate labeling
corresponding to acidocalcisomes [4] (Fig. 3.13).
Cellular response to hyposmotic and hyperosmotic stresses
To examine the role of T. cruzi Rab11 in osmoregulation, wild-type, GFP-TcRab11-
overexpressing (GFP-TcRab11OE), and GFP-TcRab11DN-expressing epimastigotes
were submitted to hyposmotic stress and their regulatory volume decrease (RVD)
measured using the light-scattering technique, as described previously [53]. This
technique measures the changes in volume of the cells under hyposmotic (swelling and
recovery) and hyperosmotic conditions (shrinking and partial recovery). After recovery

53
the cells recuperate their normal morphology. DN mutants were less able to recover their
volume after hyposmotic stress than wild type cells, while recovery was faster in GFP-
TcRab11OE cells (OE, Fig. 3.3A). In addition, when submitted to hyperosmotic stress,
DN mutants shrank less while GFP-TcRab11OE cells shrank more than control cells
(Fig. 3.3B), and in all cases they did not recover their volume during the time of the
experiment. It has been shown previously that when epimastigotes are submitted to
hyperosmotic stress the parasites do not regain their normal volume at least during the
following two hours [10]. GFP-TcRab11OE epimastigotes were also studied under
hyposmotic and hyperosmotic stress conditions by video fluorescence microscopy.
Epimastigotes were immobilized on glass slides with poly-L-Lysine and bathed in
hyposmotic/hyperosmotic buffer. Video microscopy data were collected (Figs. 3.3C and
3.3D show selected frames), which revealed changes in the morphology of the CVC
when epimastigotes were treated under both hyposmotic (Fig. 3.3C) and hyperosmotic
(Fig. 3.3D) conditions. The single fluorescent spot corresponding to the CVC could be
seen enlarging and fusing with other vacuoles probably resulting from enlarged tubular
structures of the spongiome. Altogether, these results confirm the active participation of
the CVC on the cellular response to both hyposmotic and hyperosmotic stresses [10], and
indicate that alteration of TcRab11 function leads to disruption of osmoregulatory
processes.
Trans-sialidase co-localizes with GFP-TcRab11 during differentiation of amastigotes
into trypomastigotes
As Rab11 mediates the recycling of GPI-anchored proteins of T. brucei [27,28] we
investigated whether TcRab11 affected the traffic of GPI-anchored proteins in T. cruzi.

54
Trans-sialidase is an abundant GPI-anchored protein present in the cell surface of
trypomastigotes [54,55], where it catalyzes the transfer of sialic acid from host proteins to
parasite mucins [56].
To investigate the possibility that TcRab11 mediates the traffic of TcTS to the plasma
membrane, we infected L6E9 myoblasts with metacyclic trypomastigotes from stationary
cultures of GFP-TcRab11OE parasites and obtained cell culture-derived trypomastigotes.
GFP-TcRab11OE trypomastigotes were used to infect fibroblasts and labeling of TcTS
was detected by indirect immunofluorescence analysis using antibodies against the SAPA
repeats [57] at different time points during infection (Fig. 3.4A). We found reaction with
these antibodies starting 48 h after infection when the reaction co-localized with GFP-
TcRab11 in the contractile vacuole of intracellular amastigotes (Fig. 3.4A). Co-
localization progressed to almost 100% of the cells by 106 h, after which, labeling of the
CVC gradually disappeared and surface labeling was more evident (Figs. 3.4A, and
3.4B), suggesting that TcTS traffics through the contractile vacuole before reaching the
plasma membrane in differentiating trypomastigotes. Intermediate stages between
amastigotes and trypomastigotes (‘epimastigote-like’ forms) found in the supernatants of
tissue culture cells also showed co-localization of GFP-TcRab11 and TS (Fig. 3.5A) but
in fully differentiated trypomastigotes labeling of TcTS was predominantly in patches of
the plasma membrane while GFP-TcRab11 labeling remained in the CVC (Fig. 3.5B).
Cryo-immunogold electron microscopy confirmed the co-localization of GFP-TcRab11
and TcTS in the CVC (Fig. 3.6A). Co-localization was very intense in the spongiome of
collapsed vacuoles (Fig. 3.6B). TcTS was also observed in the flagellar pocket (Figs.
3.6C, and 3.6D) and in patches in the plasma membrane (Figs. 3.6A, D), at earlier time

55
points than by IFA analysis. At later time points stronger labeling of TcTS was detected
in patches of the plasma membrane and in vesicles close to the surface (Figs. 3.6E, F).
The surface localization of TcTS in trypomastigotes has been established before by
immunogold electron microscopy studies [55,58].
Trans-sialidase co-localizes with GFP-TcRab11 in intermediate stages of
differentiation from epimastigotes to metacylic trypomastigotes
As TcTS is also present in the surface of metacyclic trypomastigotes we investigated
whether there was co-localization of TcTS with GFP-TcRab11 during differentiation of
epimastigotes into metacyclic trypomastigotes as described under Materials and Methods.
Fig. 3.5C shows the co-localization of antibodies against TcTS with GFP-TcRab11 in
intermediate forms that appeared around day 5 of the metacyclogenesis process.
TcRab11DN mutant prevents plasma membrane localization of TcTS but not of
other plasma membrane proteins
To investigate whether mutation of TcRab11 affects general traffic of membrane proteins
to the cell surface of trypomastigotes, wild type and GFP-TcRab11DN trypomastigotes
were used to infect HF fibroblasts and labeling of TcTS and other membrane proteins
were detected by indirect immmunofluorescence analysis after a full cycle of
differentiation into trypomastigotes.
Wild type trypomastigotes showed labeling of TcTS in the plasma membrane (Fig. 3.7A)
while GFP-TcRab11DN intermediate forms (Fig. 3.7B) and trypomastigotes (Fig. 3.7C),
identified by the position of the kinetoplast anterior or posterior to the nucleus,
respectively, showed predominantly cytosolic labeling of TcTS (Fig. 3.7B-D). This weak
intracellular label with TcTS could be the result of ER retention and export to the cytosol

56
that ultimately results in its degradation by the ubiquitin/proteasome system [59].
Labeling of GFP-TcRab11DN was predominantly punctated cytosolic, as described
above for epimastigotes (Fig. 3.2A). These results suggest that DN mutation of TcRab11
inhibits traffic of TcTS to the plasma membrane. To further confirm this observation we
used SAPA antibodies to assess surface expression of TcTS by flow cytometry on GFP-
TcRab11DN and wild type trypomastigotes. As expected, flow cytometric analysis shows
reduction in surface expression of TcTS in the mutants as compared to control wild type
trypomastigotes (Fig. 3.7E). Western blot analyses showed that these trypomastigotes
maintained the overexpression of GFP-TcRab11 and GFP-TcRab11DN (Fig. 3.12D). To
address the specificity of the TcTS antibody, total parasite lysates of wild type and GFP-
TcRab11DN were subjected to western blot analyses. Signals were observed in both
lanes, matching the expected size of the TcTSs [37,60] (Fig. 3.13C).
We next investigated whether other GPI-anchored proteins or integral membrane proteins
required TcRab11 for trafficking to the surface. We selected for study TcTSSA II, which
is a mucin-type GPI-anchored protein [42], and GPI-anchored mucin-like glycoproteins
expressed on the cell surface of trypomastigotes that are recognized by anti-α-galactosyl
antibodies from patients with chronic Chagas disease [43-45]. Also selected was a P-
Type H+-ATPase, which is a proton pump important for maintenance of pH homeostasis
and plasma membrane potential of T. cruzi different stages [61,62] and that also localizes
to the endocytic pathway of the parasites [63]. Antibodies against TcTSSA II co-
localized with GFP-TcRab11 as assayed by indirect immunofluorescence analysis of
intermediate forms (Fig. 3.8A) and intracellular amastigotes (Fig. 3.8B) and trafficked to
the plasma membrane of trypomastigotes (Fig. 3.8C). Antibodies against α-Gal also co-

57
localized with GFP-TcRab11 in the intermediate forms (Fig. 3.9A) before reaching the
cell surface in the fully differentiated trypomastigotes (Fig. 3.9B). However, traffic of
both mucins to the plasma membrane was not prevented in GFP-TcRab11DN-expressing
parasites (Fig. 3.8D and 3.9C). Similarly, plasma membrane and intracellular localization
of the P-type H+-ATPase, which did not co-localize with GFP-TcRab11, was not affected
in GFP-Rab11DN parasites (Fig. 3.8E).
We also investigated the traffic of GPI-anchored surface antigens during
metacyclogenesis, as described under Materials and Methods. We followed traffic of
gp35/50 mucins, which are expressed in epimastigote and metacyclic forms.
Immunofluorescence assays on GFP-TcRab11OE parasites with monoclonal antibody
2B10 [64] demonstrates the co-localization of GFP-TcRab11 with gp35/50 in the CVC of
intermediate stages of differentiation (obtained at day 5 of metacyclogenesis) towards
metacyclics trypomastigotes (Fig. 3.14A) and the lack of co-localization in metacyclic
forms (obtained at day 10 of metacyclogenesis) (Fig. 3.14B). However, GFP-
TcRab11DN mutants did not show any defect on the surface localization of this protein
(Fig. 3.14C).
CVC is enriched in lipid rafts
It has been proposed that GPI-anchored proteins acquire detergent resistance by fatty acid
remodeling in the Golgi and their sorting is correlated with lipid raft formation at the
trans-Golgi (TG) network [48]. To investigate whether the CVC possesses rafts we
performed a detergent extraction of epimastigotes expressing different fusion constructs
previously demonstrated to associate with this organelle (TcSNARE2.1-GFP that
associates to the spongiome and GFP-TcRab11 that associates to the bladder [26],

58
followed by density gradient centrifugation in an Optiprep gradient to isolate detergent-
insoluble raft fractions. To determine whether rafts contained the fusion proteins,
detergent-insoluble fractions were separated using SDS-PAGE and analyzed by western
blotting with anti-GFP antibody. As a control for the isolation of lipid raft, a dually
acylated protein that is highly enriched in the flagellar membrane of T. cruzi, a 24-kDa
flagellar calcium-binding protein (FCaBP; [65]) was also used and detected with
monoclonal antibodies. Fractions from T. cruzi epimastigotes expressing cytoplasmic
GFP were used as negative control. Using this technique, we observed that GFP-
TcSNARE2.1, GFP-TcRab11, and FCaBP floated to the top of the Optiprep gradient
(Fig. 3.10A), suggesting the presence of lipid rafts in the CVC while GFP was associated
with the heavier fractions. The association of GFP-TcRab11 with lipid rafts was further
analyzed by another assay that is based on the temperature-dependence of lipid raft
sensitivity to detergent [66] (Fig. 3.10B). As expected GFP-TcRab11 remained insoluble
at 4°C and associated with the pellet fraction whereas it was soluble at 37oC after
centrifugation, and a cytoplasmic protein, GFP, remained soluble at either temperature
(Fig. 3.10B).
Trans-sialidase activity requirement for infection
As TcTS is important for infectivity [36] we investigated whether GFP-TcRab11DN
mutants were less effective than control cells or GFP-TcRab11OE parasites in the
establishment of T. cruzi infections. Invasion was significantly reduced in GFP-
TcRab11DN mutants as compared with controls transfected with GFP alone or GFP-
TcRab11OE parasites (Figs. 3.10C and 3.10D). There was no significant difference
between infections with wild-type trypomastigotes and trypomastigotes expressing GFP

59
alone (Fig. 3.15A and 3.15B). Pre-incubation of GFP-TcRab11DN-expressing
trypomastigotes for 30 min in the presence of recombinant TcTS and sialofetuin (as a
donor of sialic acid) [37] (Fig. 3.10D and 3.10E), but not asialofetuin (Fig. 3.15C and
3.15D) partially rescued the infectivity of the parasites demonstrating the importance of
TcTS activity for invasion of host cells.
The amino terminal 680 amino acids domain of TcTS contains the catalytic activity [67].
As a further control of the rescue experiments we did invasion experiments in the
presence of inactive recombinant TcTS (iTS), whose crystal structure has been
determined [68], and which differs in a single amino acid mutation Tyr342His that
completely abolishes its TS activity, but retains its property to recognize terminal
galactoses [32,69]. The recombinant protein binds sialic acid and galactose in vitro
[70,71] and competes with a neutralizing antibody to a discontinuous epitope of TS [37]
indicating that it is properly folded. Incubation in the presence of iTS did not rescue the
infectivity of GFP-TcRab11DN mutants (Fig. 3.10E and 3.10F). All invasion assays were
done in the absence of fetal bovine serum to prevent the presence of any other putative
exogenous sialic acid donors.
Discussion
The most significant finding of our studies is that GPI-anchored trans-sialidase (TcTS),
mucins from tissue culture-derived or metacyclic trypomastigotes, and trypomastigote
small surface antigen II (TcTSSA II) are trafficked to the plasma membrane of T. cruzi
by an unconventional pathway involving the CVC and that the CVC is enriched in lipid
rafts. We reported previously [26] that GFP-tagged TcRab11 localized to the CVC of

60
epimastigotes of T. cruzi. We now confirmed those results using antibodies against the
protein and found it in the CVC of different stages of the life cycle of the parasite. In
contrast, dominant negative TcRab11 has a punctated cytosolic localization indicating
that CVC localization is GTP-dependent. Expression of the dominant negative form of
TcRab11 makes epimastigotes less responsive to hyposmotic and hyperosmotic stresses.
These results, together with the detection by video microscopy of morphological changes
in the CVC under different osmotic conditions further demonstrate the role of the CVC in
both hyposmotic [4] and hyperosmotic [10] stresses. Expression of GFP-TcRab11DN
prevents traffic of TcTS, but not of other GPI-anchored (TcTSSA II, mucins) or integral
(H+-ATPase) membrane proteins to the plasma membrane of trypomastigotes, suggesting
a specific role of TcRab11 in trafficking of TcTS, and that this is not a default pathway
for all surface proteins. Dominant negative TcRab11 mutants might be acting by blocking
or reducing the function of endogenous TcRab11, by competing or sequestering Rab11
effector proteins [49]. GFP-TcRab11DN-expressing trypomastigotes were less virulent
but their pre-incubation with active, but not inactive, recombinant TcTS and a source of
sialic acid partially rescued their virulence, underscoring the relevance of TcTS activity
in infection. The identification of the specific role of TcTS in infection has been difficult
to demonstrate in the past because of the impossibility of doing knockouts of the
considerable number of gene copies encoding this protein scattered through the genome
of this parasite.
In mammalian cells the GPI anchor is synthesized and transferred to proteins in the ER.
GPI-anchored proteins (GPI-Aps) exit the ER from ER exit sites (ERES) and are
transported to the Golgi complex in COPII-coated vesicles [48]. Acquisition of detergent

61
resistance by fatty acid remodeling at the trans-Golgi facilitates their traffic to the plasma
membrane [48]. A similar pathway has been proposed in case of the GPI-AP variant
surface glycoprotein, or VSG, in Trypanosoma brucei, with the peculiarity that VSG
reaches first the flagellar pocket, which is the sole region for endo and exocytosis in this
organism [72]. GPI-APs are selectively endocytosed by a unique pathway involving
clathrin-independent vesicles in mammalian cells [48], while VSG is internalized via
clathrin-coated vesicles in T. brucei [72]. VSG can be retrieved from early and late
endosomes to the TbRab11-positive exocytic carriers and returned to the cell surface via
the flagellar pocket [72].
Very little is known about GPI-AP secretion or endocytosis in T. cruzi, although
uncoated vesicles containing transferrin have been observed budding off the flagellar
pocket membrane and cytostome of epimastigotes [73]. The trans-sialidase family of
proteins is predominantly expressed on the surface of trypomastigotes. Our results, using
anti-SAPA antibodies, are consistent with the synthesis of trans-sialidase in amastigotes
starting at least 48 h after infection [74] and its traffic through the CVC before reaching
the surface at the flagellar pocket. Anti-SAPA antibodies have been used before to
localize TcTS to the surface of trypomastigotes by immunoelectron microscopy [58]. The
presence of TcTS in the CVC by recycling from the surface is less probable because the
protein is only detected in the plasma membrane at later time points and no further
labeling of the CVC or endosomes is detected. It is possible that TcTS accumulates in the
CVC when rapidly synthesized during conversion of amastigotes into trypomastigotes
and then reaches a steady state and is below the limit of detection afterwards. In addition,
it is known that TcTS is shed to the extracellular medium, including within the host cells

62
[55], through the action of an endogenous phospholipase C, and also with vesicles of the
plasma membrane [75]. Other GPI-APs like TcTSSA II, and other mucins, also traffic
through the CVC before reaching the surface but its traffic to the surface is independent
of TcRab11. A possible explanation for the traffic of GPI-anchored proteins through the
CVC is that this organelle could be enriched in microdomains (or lipid rafts) in which
lipids with straight lipid chains, such as glycosphingolipids, phospholipids, and
palmitoylated proteins are packed together with cholesterol in a compact and stable
fashion [76]. Our results support the presence of lipid rafts in the CVC of T. cruzi. In this
regard, a proteomic analysis of GPI-anchored membrane protein fractions from
epimastigotes and metacyclic trypomastigotes, extracted using the neutral detergent
Triton X114 [77], detected several proteins that were later identified as present in the
CVC [26], such as TcRab11, and the membrane proteins V-H+-ATPase, and V-H
+-PPase.
Transfer of membrane [12,16,17,21] and luminal [18,20] proteins from the CVC to the
plasma membrane has been reported before in several cells, including T. cruzi
epimastigotes [21]. However, the mechanism involved was not known. In this work, we
provide evidence for a role of TcRab11 in the transfer of TcTS to the surface of the
infective stages of the parasite. The presence of vesicles labeled with antibodies against
TcTS in the proximity of the plasma membrane suggests that vesicle trafficking from the
CVC is involved in this process.
Rab proteins regulate a number of processes through their interactions with Rab effectors.
Rab11 effectors in mammalian cells comprise myosin Vb, Sec15, a component of the
exocyst complex, and a Family of Interacting Proteins or FIPs [52]. FIPS orthologues are
absent in trypanosomes, as well as class V myosins but T. brucei Rab11 has been shown

63
to interact with a Sec15 orthologue [78]. Interestingly both Rab11 [25] and Sec15 [79]
localize to the CVC of D. discoideum, and it was suggested that the CVC of D.
discoideum could be a precursor to the recycling endosomal system of other eukaryotes
[2,25].
Our results confirm the role of the CVC in both hyposmotic [4] and hyperosmotic [10]
stress and suggest that TcRab11 is important for the response of these cells to these
osmotic stresses. During its developmental cycle in the mammalian and insect hosts, T.
cruzi faces critical environmental challenges and ones that are especially dramatic are the
changes in osmolarity. Trypomastigotes need to resist osmolarities of 1,400 mOsm/kg
and return to isosmotic conditions (300 mOsm/kg) when circulating through the renal
medulla [80]. Amastigotes reproduce in some tissues that have higher osmolarity than
serum (330 in lymphoid tissues vs 300 mOsm/kg) [81], and epimastigotes need to resist
high osmolarities (~1,000 mOsm/kg) in the rectal content of the insect vector [82].
TcRab11 appears to have a role in the resistance to these changes.
In summary, we describe a new unconventional pathway of GPI-APs to the plasma
membrane that includes their traffic through the contractile vacuole complex. TcTS
requires the participation of TcRab11 to reach the plasma membrane, while TcTSSA II
and other mucins do not. This traffic of proteins through the CVC appears to be specific
for GPI-APs, since other membrane proteins do not follow the same pathway.

64
Materials and Methods
Cell culture
Human foreskin fibroblasts (HFF) were grown in DMEM Low Glucose medium
supplemented with 10% Cosmic CalfTM
serum and 0.1% L-glutamine. Vero cells were
grown in RPMI supplemented with 10% fetal bovine serum. L6E9 myoblasts were grown
in DMEM High Glucose medium supplemented with 10% fetal bovine serum. Host cells
were maintained at 37°C with 5% CO2. Tissue culture cell-derived trypomastigotes were
obtained from L6E9 myoblasts infected with metacyclic trypomastigotes from stationary
cultures of GFP-TcRab11OE and GFP-TcRab11DN parasites. T. cruzi amastigote and
trypomastigote forms were collected from the culture medium of infected host cells,
using a modification of the method of Schmatz and Murray [83] as described previously
[84]. Epimastigotes from T. cruzi were cultured in liver infusion tryptose (LIT) medium
containing 10% newborn serum at 28°C [10]. T. cruzi epimastigotes transfected with
GFP-TcRab11OE and GFP-TcRab11DN were maintained in the presence of 250 µg/ml
geneticin (G418).
Chemicals and reagents
Fetal bovine serum, newborn calf serum, Dulbecco’s phosphate buffer saline (PBS) and
Hank’s solution, 4’,6-diamidino-2-phenylindole (DAPI), DMEM and RPMI media,
paraformaldehyde, bovine serum albumin, and protease inhibitors were purchased from
Sigma (St. Louis, MO). Restriction enzymes, were from New England BioLabs (Ipswich,
MA). pCR2.1-TOPO cloning kit, 1 kb plus DNA ladder, rabbit GFP antibodies and Gene
Tailor Site-Directed Mutagenesis System were from Invitrogen (Life Technologies,
Grand Island, NY). Hybond-N nylon membranes were obtained from PerkinElmer

65
(Waltham, MA). TbRab11 purified antibodies were a gift from Mark Field (University of
Dundee, Scotland). Monoclonal antibody 2B10 was a gift from Nobuko Yoshida (Federal
University of São Paulo, Brazil), Chagasic α-Gal antibodies were a gift from Igor de
Almeida (University of Texas, El Paso), antibody against TcTSSA II was a gift from
Carlos Buscaglia (National University of San Martin, Argentina), monoclonal antibody
FCaBP was a gift from David Engman (Northwestern University, Evanston, IL). Rabbit
and goat GFP antibodies were from Abcam (Cambridge, MA). Recombinant active TcTS
and inactive TcTS (iTS) were obtained as described (65-67). BCA Protein Assay Reagent
was from Pierce (Thermo Fisher Scientific, Rockford, IL). All other reagents were
analytical grade. The oligonucleotides were ordered from Sigma or IDT (Coralville, IA).
Metacyclogenesis
We followed the protocol described by Bourguignon et al. [85] with some modifications.
Epimastigotes were obtained after 4 days in LIT medium and submitted to a stress
(incubation for 2 h in a medium containing 190 mM NaCl, 17 mM KCl, 2 mM MgCl2, 2
mM CaCl2, 0.035% sodium bicarbonate, 8 mM phosphate, pH 6.9 at room temperature;
triatome artificial urine (TAU) medium). After this stress, parasites were incubated for 96
h in TAU 3AAG medium (which consists of the previously described TAU medium
supplemented with 10 mM L-proline, 50 mM sodium L-glutamate, 2 mM sodium L-
aspartate, and 10 mM glucose). To increase the number of metacyclic forms, the contents
of the flask were collected and resuspended in media containing fresh fetal bovine serum
and incubated at 37°C for 20 h. The complement in the FBS kills epimastigotes while
metacyclic trypomastigotes survive. Samples were harvested from the TAU 3AAG +
FBS-containing medium at days 5 and 10 of cultivation.

66
In vitro infection assay
HFF or irradiated myoblasts (6 x 105
cells per well) were equally distributed in a 12-well
plate on a sterile coverslip in their respective growth media (as mentioned above) and
were incubated for 24 h at 37°C in a 5% CO2 atmosphere. The following day, the cells
were washed once with Dulbecco’s Hank’s solution, and 6 x 106 wild type, TcGFP, GFP-
TcRab11OE, or GFP-TcRab11DN trypomastigotes were added to each well (10
trypomastigotes per myoblast or HFF), and they were incubated for 4 h at 37°C in a 5%
CO2 atmosphere. To decrease the chances of contamination of cell derived-
trypomastigotes with extracellular amastigotes, collections of parasites were centrifuged
and incubated at 37°C for 2 h to allow trypomastigotes to swim to the surface. The
supernatant was collected and used for subsequent invasion assays. Next, the parasites
were removed from the plate, and the infected cells were washed extensively with
Dulbecco’s Hank’s solution and fixed for immunofluorescence assays. For rescue
experiments the same number of trypomastigotes were incubated with PBS, pH 7.4, in
the absence of serum, and with fetuin or asialofetuin (solutions made in PBS, pH 7.4, and
sterilized by filtration) at a final concentration of 10 g/ml, and with 200 ng of active
(TcTS) or inactive (iTS) trans-sialidase for 30 min at room temperature before infecting
host cells. For attachment/internalization assays, recently internalized parasites, and
parasites caught in the process of invasion, were considered and manually counted in at
least 200 DAPI-stained cells in 3 independent experiments. The percentage of infected
cells and the average number of parasites per infected cell were determined.

67
Immunofluorescence and western blot analyses
For immunofluorescence microscopy, parasites were fixed in PBS, pH 7.4, with 4%
paraformaldehyde, adhered to poly-lysine coverslips, and permeabilized for 3 min with
PBS, pH 7.4, containing 0.3% Triton X-100. Permeabilized cells were quenched for 30
min at room temperature with 50 mM NH4Cl and blocked overnight with 3% BSA in
PBS, pH 8.0. Both primary and secondary antibodies were incubated for 1 h at room
temperature. Coverslips were mounted by using a mounting medium containing DAPI at
5 µg/ml for staining DNA-containing organelles. For imaging of intracellular parasites,
mammalian cells were seeded onto sterile coverslips in 12-well culture plates and
allowed to grow for 24 h. To semi-synchronize the infection, we added the parasites at a
ratio of 10:1 (parasite/host cell) for 4 hours, washed the cells to eliminate extracellular
parasites and fixed in cold methanol for 30 min. Infected cells were prepared for
immunofluorescence analyses as described above for extracellular parasites, except for
the permeabilization that was performed for 10 min with Triton X-100 in PBS, pH 7.4.
The dilution used for primary antibodies were as follows: rabbit anti-TcAQP1, 1:50 [3];
rabbit anti-TbRab11 [24] 1:200; rabbit polyclonal anti-GFP, 1:500; rabbit anti-TcTS [57],
1:2,000; rabbit anti-TcTSSA II [42], 1:200; rabbit anti-H+ATPase [63], 1:100.
Differential interference contrast (DIC) and direct fluorescence images were obtained by
using an Olympus IX-71 inverted fluorescence microscope with a
PhotometrixCoolSnapHQ charge-coupled device camera driven by Delta Vision
softWoRx3.5.1 (Applied Precision, Issaquah, WA). Images were deconvolved for 10
cycles using the same software and applying the “noise filter” at “medium” mode. This is
an automatic deconvolution software and was applied to all channels; brightness and

68
contrast were the same in all channels. The figures were built by using Adobe Photoshop
10.0.1 (Adobe System, Inc., San Jose, CA).
For western blot analysis, ~108 T. cruzi epimastigotes, amastigotes or trypomastigotes
were collected by centrifugation at 1,600 x g for 10 min, washed twice in PBS, pH 7.4,
and resuspended in modified radioimmunoprecipitation analysis (RIPA) buffer (150 mM
NaCl, 20 mM Tris-Cl pH 7.5, 1 mM EDTA, 1% SDS and 0.1% Triton X-100) containing
protease inhibitor cocktail (2 mM EDTA, 2 mM phenylmethylsulfonyl fluoride (PMSF),
2 mM tosylphenylalanylchloromethyl ketone (TPCK), 0.1 mM trans-epoxysuccinyl-L-
leucylamido(4-guanidino) butane (E64) and Sigma P8340 protease inhibitor cocktail,
1:250). Cells were mechanically fragmented by passing lysates through a 20-gauge
needle five times. The protein concentration was estimated by spectrophotometry, using
the BCA Protein Assay Reagent. Twenty micrograms of protein from each total cell
lysate was mixed with 2X Laemmli sample buffer, boiled for 5 min, and total
homogenate of each sample were separated by SDS-PAGE. Proteins were transferred
onto nitrocellulose membranes and blocked overnight with 5% nonfat dry milk in PBS-
0.1% Tween 20 (PBS-T). The following primary antibodies were applied at room
temperature for 1 hr: rabbit anti-GFP at 1:1000, mFCaBP at 1:50, and rabbit anti-TcTS at
1:5000. Densitometric analysis of 3 independent experiments was performed with Alfa-
Imager software.
Flow cytometry
Tissue culture-derived trypomastigotes (106
cells) were fixed in 4% paraformaldehyde in
PBS, pH 7.4, and washed in blocking solution (3% BSA in PBS). After washing, cells
were incubated with the anti-TcTS (1:2,000 dilution) in blocking solution for 1 hr on ice.

69
Parasites were washed and incubated in Alexa Fluor 633 goat anti-rabbit for one hour on
ice. After washing, parasites were resuspended in PBS and samples were sorted on a
MoFlo cytometer (Cytomation, Fort Collins, CO) using a 633 nm argon laser for
excitation and an emission filter of 632/647 nm band pass. Samples were manually gated
to eliminate debris and dead parasites or cells. Data were analyzed using Summit version
3.1 (Cytomation) and prepared for publication using Flowjo version 4.0.2 (Treestar, San
Carlos, CA)
Generation of TcRab11 dominant negative mutant and transfection
Dominant negative forms of Rab11 were constructed via site directed mutagenesis by the
use of Gene Tailor Site-Directed Mutagenesis System. This method involved methylating
the TOPO blunt end vector containing the coding sequence for TcRab11 with DNA
methylase at 37°C for 1 hour, followed by amplification of the plasmid in a mutagenesis
reaction with two overlapping primers, forward, 5’-
GCGATAGTGGCGTCGGCAAGAACAACCTCATGACG-3’ and reverse, 5’-
CTTGCCGACGCCACTATCGCCGATGATGACAAC-3’ of which the forward primer
had the target mutation, resulting in the mutation of amino acid serine to asparagine.
Mutations were confirmed by sequencing (Yale DNA Analysis Facility, Yale University,
New Haven, Connecticut). After transformation the resulting plasmid TcRab11S21N in
TOPO was digested with restriction enzymes BamHI and HindIII. The circular pTEX-N-
GFP vector was linearized by the corresponding restriction enzymes. Finally,
TcRab11S21N insert was ligated to pTEX-N-GFP followed by transformation. The
plasmid pTEX-N-GFPTcRab11S21N was sequenced to confirm that the correct reading
frame was used. T. cruzi CL strain epimastigotes were transfected in cytomix (120 mM

70
KCl, 0.15 mM CaCl2, 10 mM K2HPO4, 2 mM EDTA, 5 mM MgCl2, pH 7.6) containing
50 μg of the plasmid construct in a 4 mm cuvette. The cuvette was cooled on ice for 10
min and pulsed 3 times (1.5 kV, 25 μF) with a Gene Pulser Xcell™ (Bio-Rad), and
expression of GFP-fusion proteins was verified by western blot analyses. Stable cell lines
were established under drug selection with G418 at 250 μg/ml. Enrichment of GFP
fluorescent parasites was performed with a high-speed cell sorter when needed (MoFlo
Legacy; Beckman-Coulter, Hialeah, FL).
Cryo-immunoelectron microscopy
HFF containing intracellular GFP-TcRab11OE expressing amastigotes were detached by
treating the T25 flasks with 0.25% trypsin at 96 h and 106 h post-infection. The contents
of the flask were collected and amastigotes were isolated from the host cells by passing
them through a 20-gauge needle. The released amastigotes (with ~5% contamination of
trypomastigotes) were fixed in 4% paraformaldehyde in 0.1 M cacodylate buffer, pH 7.3
for 1 h on ice. Epimastigotes were collected as described above and submitted to
hyposmotic conditions. Hyposmotic stress was induced by addition of hyposmotic buffer
(64 mM NaCl, 4 mM KCl, 1.8 mM CaCl2, 0.53 mM MgCl2, 5.5 mM glucose, 50 mM D-
mannitol, 5 mM Hepes-Na, pH 7.4) to a final osmolarity of 177 mosmol/L for 2 min and
then fixed with 0.1 % glutaraldehyde and 4% paraformaldehyde in 0.1 M cacodylate
buffer, pH 7.3 for 1 h on ice. The samples were processed for cryo-immunoelectron
microscopy at the Molecular Microbiology Imaging Facility, Washington University
School of Medicine. The antibodies used were: goat anti-GFP (1:500), rabbit anti-GFP
(1:50), rabbit anti-TcTS (1:250), donkey anti-goat 18 nm colloidal gold, donkey anti-
rabbit 18 nm colloidal gold, donkey anti-rabbit 12 nm colloidal gold.

71
Cell volume measurements
T. cruzi epimastigotes (GFP-TcRab11OE, GFP-TcRab11DN and wild-type) at log phase
of growth (3 days) were collected at 1,600 g for 10 min (at a density of 1 x 108/ml),
washed twice in PBS and resuspended in isosmotic buffer (64 mM NaCl, 4 mM KCl, 1.8
mM CaCl2, 0.53 mM MgCl2, 5.5 mM glucose, 150 mM D-mannitol, 5 mM Hepes-Na,
pH 7.4, to a final osmolarity of 282 mosmol/L, as determined using an Advanced
Instruments 3D3 osmometer. Relative cell volume changes after osmotic stress were
measured by light scattering. Aliquots of parasites were distributed in 96 well plates such
that each well had 1 x 107 cells and an appropriate volume of the corresponding buffer
was added for osmotic stress. Hyposmotic stress was induced by dilution of the isosmotic
cell suspension with deionized water to a final osmolarity of 150 mOsm at time zero.
Hyperosmotic stress was induced by addition of hyperosmotic buffer (64 mM NaCl, 4
mM KCl, 1.8 mM CaCl2, 0.53 mM MgCl2, 5.5 mM glucose, 500 mM D-mannitol, 5 mM
Hepes-Na, pH 7.4) to a final osmolarity of 650 mosmol/L. Absorbance at 550 nm was
monitored every 10 sec for 10 min using a SpectraMax M2e plate reader (Molecular
Devices) [10]. A decrease in absorbance corresponds to an increase in cell volume. The
results were normalized respect to the value of a 3 min pre-reading under isosmotic
conditions.
Video microscopy
Epimastigotes (1 x 108 cells) in logarithmic phase of growth were collected by
centrifugation, washed 3 times in PBS and resuspended in isosmotic buffer (composition
mentioned above). GFP-TcRab11 overexpressing epimastigotes were immobilized with
poly-L-lysine on coverslips in MatTek glass bottom dishes for 30 min at room

72
temperature. Unattached cells were washed with PBS. To induce hyposmotic stress the
isosmotic buffer was diluted by 1:1 with deionized water. Hyperosmotic stress was
induced by bathing the chamber with hyperosmotic buffer (as described above). Time
lapse photographic data were collected at 1 sec intervals with a 60X objective and a 1024
X 1024 field with a Delta Vision Elite system (Applied Precision). Video sequences were
reconstructed using Quicktime software.
Lipid raft isolation
An Optiprep gradient centrifugation (sucrose float) procedure was used to isolate lipid
rafts from T. cruzi epimastigotes wild type Y strain and those expressing GFP, GFP-
TcRab11 and GFP-TcSNARE2.1 fusion proteins using lysates equivalent to 2.5 x 108
mid log phase epimastigotes for each sample. The procedure was as described before [86]
with minor modifications. Briefly, tubes were centrifuged continuously at 4 °C in a
Beckman Coulter OptimaTM
L-100XP ultracentrifuge with a Beckman SW32Ti rotor at
35,000 rpm (210,000 x g) for 5 h and then 25,000 rpm (107,000 x g) for 8 h. After
collecting the fractions, a 24 µl aliquot of each fraction was mixed with 6 µl of 5X SDS-
PAGE loading buffer, boiled for 10 min, and processed for SDS-PAGE and western blot
analysis as above. The procedure for temperature-dependent Triton X-100 extraction for
GFP-TcRab11- and GFP-expressing epimastigotes was as described [66].

73
REFERENCES
1. Allen RD, Naitoh Y (2002) Osmoregulation and contractile vacuoles of protozoa.
International review of cytology 215: 351-394.
2. Docampo R, Jimenez V, Lander N, Li ZH, Niyogi S (2013) New insights into roles of
acidocalcisomes and contractile vacuole complex in osmoregulation in protists.
International review of cell and molecular biology 305: 69-113.
3. Montalvetti A, Rohloff P, Docampo R (2004) A functional aquaporin co-localizes with
the vacuolar proton pyrophosphatase to acidocalcisomes and the contractile
vacuole complex of Trypanosoma cruzi. The Journal of biological chemistry 279:
38673-38682.
4. Rohloff P, Montalvetti A, Docampo R (2004) Acidocalcisomes and the contractile
vacuole complex are involved in osmoregulation in Trypanosoma cruzi. The
Journal of biological chemistry 279: 52270-52281.
5. Figarella K, Uzcategui NL, Zhou Y, LeFurgey A, Ouellette M, et al. (2007)
Biochemical characterization of Leishmania major aquaglyceroporin LmAQP1:
possible role in volume regulation and osmotaxis. Molecular microbiology 65:
1006-1017.
6. Nishihara E, Yokota E, Tazaki A, Orii H, Katsuhara M, et al. (2008) Presence of
aquaporin and V-ATPase on the contractile vacuole of Amoeba proteus. Biology
of the cell / under the auspices of the European Cell Biology Organization 100:
179-188.
7. Komsic-Buchmann K, Stephan LM, Becker B (2012) The SEC6 protein is required for
contractile vacuole function in Chlamydomonas reinhardtii. Journal of cell
science 125: 2885-2895.
8. Clark TB (1959) Comparative morphology of four genera of trypanosomatidae. J
Protozool 6: 227-232.

74
9. Girard-Dias W, Alcantara CL, Cunha ESN, de Souza W, Miranda K (2012) On the
ultrastructural organization of Trypanosoma cruzi using cryopreparation methods
and electron tomography. Histochemistry and cell biology 138: 821-831.
10. Li ZH, Alvarez VE, De Gaudenzi JG, Sant'Anna C, Frasch AC, et al. (2011)
Hyperosmotic stress induces aquaporin-dependent cell shrinkage, polyphosphate
synthesis, amino acid accumulation, and global gene expression changes in
Trypanosoma cruzi. The Journal of biological chemistry 286: 43959-43971.
11. Patel S, Docampo R (2010) Acidic calcium stores open for business: expanding the
potential for intracellular Ca2+ signaling. Trends in cell biology 20: 277-286.
12. Moniakis J, Coukell MB, Janiec A (1999) Involvement of the Ca2+-ATPase PAT1
and the contractile vacuole in calcium regulation in Dictyostelium discoideum.
Journal of cell science 112 ( Pt 3): 405-414.
13. Malchow D, Lusche DF, Schlatterer C, De Lozanne A, Muller-Taubenberger A
(2006) The contractile vacuole in Ca2+-regulation in Dictyostelium: its essential
function for cAMP-induced Ca2+-influx. BMC developmental biology 6: 31.
14. Ladenburger EM, Korn I, Kasielke N, Wassmer T, Plattner H (2006) An Ins(1,4,5)P3
receptor in Paramecium is associated with the osmoregulatory system. Journal of
cell science 119: 3705-3717.
15. Ludlow MJ, Durai L, Ennion SJ (2009) Functional characterization of intracellular
Dictyostelium discoideum P2X receptors. The Journal of biological chemistry
284: 35227-35239.
16. Sivaramakrishnan V, Fountain SJ (2012) A mechanism of intracellular P2X receptor
activation. The Journal of biological chemistry 287: 28315-28326.
17. Heuser J, Zhu Q, Clarke M (1993) Proton pumps populate the contractile vacuoles of
Dictyostelium amoebae. The Journal of cell biology 121: 1311-1327.
18. Sesaki H, Wong EF, Siu CH (1997) The cell adhesion molecule DdCAD-1 in
Dictyostelium is targeted to the cell surface by a nonclassical transport pathway
involving contractile vacuoles. The Journal of cell biology 138: 939-951.
19. Heuser J (2006) Evidence for recycling of contractile vacuole membrane during
osmoregulation in Dictyostelium amoebae--a tribute to Gunther Gerisch.
European journal of cell biology 85: 859-871.

75
20. Sriskanthadevan S, Lee T, Lin Z, Yang D, Siu CH (2009) Cell adhesion molecule
DdCAD-1 is imported into contractile vacuoles by membrane invagination in a
Ca2+- and conformation-dependent manner. The Journal of biological chemistry
284: 36377-36386.
21. Hasne MP, Coppens I, Soysa R, Ullman B (2010) A high-affinity putrescine-
cadaverine transporter from Trypanosoma cruzi. Molecular microbiology 76: 78-
91.
22. Macro L, Jaiswal JK, Simon SM (2012) Dynamics of clathrin-mediated endocytosis
and its requirement for organelle biogenesis in Dictyostelium. J Cell Sci 125:
5721-5732.
23. Kelly EE, Horgan CP, McCaffrey MW (2012) Rab11 proteins in health and disease.
Biochemical Society transactions 40: 1360-1367.
24. Jeffries TR, Morgan GW, Field MC (2001) A developmentally regulated rab11
homologue in Trypanosoma brucei is involved in recycling processes. Journal of
cell science 114: 2617-2626.
25. Harris E, Yoshida K, Cardelli J, Bush J (2001) Rab11-like GTPase associates with
and regulates the structure and function of the contractile vacuole system in
dictyostelium. Journal of cell science 114: 3035-3045.
26. Ulrich PN, Jimenez V, Park M, Martins VP, Atwood J, 3rd, et al. (2011)
Identification of contractile vacuole proteins in Trypanosoma cruzi. PloS one 6:
e18013.
27. Pal A, Hall BS, Jeffries TR, Field MC (2003) Rab5 and Rab11 mediate transferrin
and anti-variant surface glycoprotein antibody recycling in Trypanosoma brucei.
The Biochemical journal 374: 443-451.
28. Grunfelder CG, Engstler M, Weise F, Schwarz H, Stierhof YD, et al. (2003)
Endocytosis of a glycosylphosphatidylinositol-anchored protein via clathrin-
coated vesicles, sorting by default in endosomes, and exocytosis via RAB11-
positive carriers. Molecular biology of the cell 14: 2029-2040.
29. El-Sayed NM, Myler PJ, Bartholomeu DC, Nilsson D, Aggarwal G, et al. (2005) The
genome sequence of Trypanosoma cruzi, etiologic agent of Chagas disease.
Science 309: 409-415.

76
30. Freitas LM, dos Santos SL, Rodrigues-Luiz GF, Mendes TA, Rodrigues TS, et al.
(2011) Genomic analyses, gene expression and antigenic profile of the trans-
sialidase superfamily of Trypanosoma cruzi reveal an undetected level of
complexity. PloS one 6: e25914.
31. Di Noia JM, Buscaglia CA, De Marchi CR, Almeida IC, Frasch AC (2002) A
Trypanosoma cruzi small surface molecule provides the first immunological
evidence that Chagas' disease is due to a single parasite lineage. The Journal of
experimental medicine 195: 401-413.
32. Cremona ML, Campetella O, Sanchez DO, Frasch AC (1999) Enzymically inactive
members of the trans-sialidase family from Trypanosoma cruzi display beta-
galactose binding activity. Glycobiology 9: 581-587.
33. Tomlinson S, Pontes de Carvalho LC, Vandekerckhove F, Nussenzweig V (1994)
Role of sialic acid in the resistance of Trypanosoma cruzi trypomastigotes to
complement. Journal of immunology 153: 3141-3147.
34. Buscaglia CA, Campo VA, Frasch AC, Di Noia JM (2006) Trypanosoma cruzi
surface mucins: host-dependent coat diversity. Nature reviews Microbiology 4:
229-236.
35. Pereira-Chioccola VL, Acosta-Serrano A, Correia de Almeida I, Ferguson MA,
Souto-Padron T, et al. (2000) Mucin-like molecules form a negatively charged
coat that protects Trypanosoma cruzi trypomastigotes from killing by human anti-
alpha-galactosyl antibodies. Journal of cell science 113 ( Pt 7): 1299-1307.
36. Schenkman S, Jiang MS, Hart GW, Nussenzweig V (1991) A novel cell surface trans-
sialidase of Trypanosoma cruzi generates a stage-specific epitope required for
invasion of mammalian cells. Cell 65: 1117-1125.
37. Buschiazzo A, Muia R, Larrieux N, Pitcovsky T, Mucci J, et al. (2012) Trypanosoma
cruzi trans-sialidase in complex with a neutralizing antibody: structure/function
studies towards the rational design of inhibitors. PLoS pathogens 8: e1002474.
38. Rubin-de-Celis SS, Uemura H, Yoshida N, Schenkman S (2006) Expression of
trypomastigote trans-sialidase in metacyclic forms of Trypanosoma cruzi
increases parasite escape from its parasitophorous vacuole. Cellular microbiology
8: 1888-1898.

77
39. Tribulatti MV, Mucci J, Van Rooijen N, Leguizamon MS, Campetella O (2005) The
trans-sialidase from Trypanosoma cruzi induces thrombocytopenia during acute
Chagas' disease by reducing the platelet sialic acid contents. Infection and
immunity 73: 201-207.
40. Mucci J, Hidalgo A, Mocetti E, Argibay PF, Leguizamon MS, et al. (2002)
Thymocyte depletion in Trypanosoma cruzi infection is mediated by trans-
sialidase-induced apoptosis on nurse cells complex. Proceedings of the National
Academy of Sciences of the United States of America 99: 3896-3901.
41. Freire-de-Lima L, Alisson-Silva F, Carvalho ST, Takiya CM, Rodrigues MM, et al.
(2010) Trypanosoma cruzi subverts host cell sialylation and may compromise
antigen-specific CD8+ T cell responses. J Biol Chem 285: 13388-13396.
42. Canepa GE, Degese MS, Budu A, Garcia CR, Buscaglia CA (2012) Involvement of
TSSA (trypomastigote small surface antigen) in Trypanosoma cruzi invasion of
mammalian cells. The Biochemical journal 444: 211-218.
43. Almeida IC, Ferguson MA, Schenkman S, Travassos LR (1994) Lytic anti-alpha-
galactosyl antibodies from patients with chronic Chagas' disease recognize novel
O-linked oligosaccharides on mucin-like glycosyl-phosphatidylinositol-anchored
glycoproteins of Trypanosoma cruzi. Biochem J 304 ( Pt 3): 793-802.
44. Almeida IC, Krautz GM, Krettli AU, Travassos LR (1993) Glycoconjugates of
Trypanosoma cruzi: a 74 kD antigen of trypomastigotes specifically reacts with
lytic anti-alpha-galactosyl antibodies from patients with chronic Chagas disease. J
Clin Lab Anal 7: 307-316.
45. Almeida IC, Milani SR, Gorin PA, Travassos LR (1991) Complement-mediated lysis
of Trypanosoma cruzi trypomastigotes by human anti-alpha-galactosyl antibodies.
J Immunol 146: 2394-2400.
46. Di Noia JM, D'Orso I, Aslund L, Sanchez DO, Frasch AC (1998) The Trypanosoma
cruzi mucin family is transcribed from hundreds of genes having hypervariable
regions. J Biol Chem 273: 10843-10850.
47. Yoshida N, Mortara RA, Araguth MF, Gonzalez JC, Russo M (1989) Metacyclic
neutralizing effect of monoclonal antibody 10D8 directed to the 35- and 50-

78
kilodalton surface glycoconjugates of Trypanosoma cruzi. Infect Immun 57:
1663-1667.
48. Fujita M, Kinoshita T (2012) GPI-anchor remodeling: potential functions of GPI-
anchors in intracellular trafficking and membrane dynamics. Biochimica et
biophysica acta 1821: 1050-1058.
49. Fukuda M (2010) How can mammalian Rab small GTPases be comprehensively
analyzed?: Development of new tools to comprehensively analyze mammalian
Rabs in membrane traffic. Histology and histopathology 25: 1473-1480.
50. Docampo R (2011) Molecular parasitology in the 21st century. Essays in
biochemistry 51: 1-13.
51. Feig LA (1999) Tools of the trade: use of dominant-inhibitory mutants of Ras-family
GTPases. Nature cell biology 1: E25-27.
52. Stenmark H (2009) Rab GTPases as coordinators of vesicle traffic. Nature reviews
Molecular cell biology 10: 513-525.
53. Rohloff P, Rodrigues CO, Docampo R (2003) Regulatory volume decrease in
Trypanosoma cruzi involves amino acid efflux and changes in intracellular
calcium. Molecular and biochemical parasitology 126: 219-230.
54. Pereira ME (1983) A developmentally regulated neuraminidase activity in
Trypanosoma cruzi. Science 219: 1444-1446.
55. Frevert U, Schenkman S, Nussenzweig V (1992) Stage-specific expression and
intracellular shedding of the cell surface trans-sialidase of Trypanosoma cruzi.
Infection and immunity 60: 2349-2360.
56. Giorgi ME, de Lederkremer RM (2011) Trans-sialidase and mucins of Trypanosoma
cruzi: an important interplay for the parasite. Carbohydrate research 346: 1389-
1393.
57. Buscaglia CA, Campetella O, Leguizamon MS, Frasch AC (1998) The repetitive
domain of Trypanosoma cruzi trans-sialidase enhances the immune response
against the catalytic domain. The Journal of infectious diseases 177: 431-436.
58. Souto-Padron T, Reyes MB, Leguizamon S, Campetella OE, Frasch AC, et al. (1989)
Trypanosoma cruzi proteins which are antigenic during human infections are
located in defined regions of the parasite. Eur J Cell Biol 50: 272-278.

79
59. Ellgaard L, Molinari M, Helenius A (1999) Setting the standards: quality control in
the secretory pathway. Science 286: 1882-1888.
60. Parodi AJ, Pollevick GD, Mautner M, Buschiazzo A, Sanchez DO, et al. (1992)
Identification of the gene(s) coding for the trans-sialidase of Trypanosoma cruzi.
EMBO J 11: 1705-1710.
61. Vanderheyden N, Benaim G, Docampo R (1996) The role of a H(+)-ATPase in the
regulation of cytoplasmic pH in Trypanosoma cruzi epimastigotes. The
Biochemical journal 318 ( Pt 1): 103-109.
62. Luo S, Scott DA, Docampo R (2002) Trypanosoma cruzi H+-ATPase 1 (TcHA1) and
2 (TcHA2) genes complement yeast mutants defective in H+ pumps and encode
plasma membrane P-type H+-ATPases with different enzymatic properties. The
Journal of biological chemistry 277: 44497-44506.
63. Vieira M, Rohloff P, Luo S, Cunha-e-Silva NL, de Souza W, et al. (2005) Role for a
P-type H+-ATPase in the acidification of the endocytic pathway of Trypanosoma
cruzi. The Biochemical journal 392: 467-474.
64. Yoshida N (2006) Molecular basis of mammalian cell invasion by Trypanosoma
cruzi. An Acad Bras Cienc 78: 87-111.
65. Tyler KM, Fridberg A, Toriello KM, Olson CL, Cieslak JA, et al. (2009) Flagellar
membrane localization via association with lipid rafts. J Cell Sci 122: 859-866.
66. Maric D, McGwire BS, Buchanan KT, Olson CL, Emmer BT, et al. (2011) Molecular
determinants of ciliary membrane localization of Trypanosoma cruzi flagellar
calcium-binding protein. J Biol Chem 286: 33109-33117.
67. Campetella OE, Uttaro AD, Parodi AJ, Frasch AC (1994) A recombinant
Trypanosoma cruzi trans-sialidase lacking the amino acid repeats retains the
enzymatic activity. Mol Biochem Parasitol 64: 337-340.
68. Oppezzo P, Obal G, Baraibar MA, Pritsch O, Alzari PM, et al. (2011) Crystal
structure of an enzymatically inactive trans-sialidase-like lectin from
Trypanosoma cruzi: the carbohydrate binding mechanism involves residual
sialidase activity. Biochim Biophys Acta 1814: 1154-1161.

80
69. Cremona ML, Sanchez DO, Frasch AC, Campetella O (1995) A single tyrosine
differentiates active and inactive Trypanosoma cruzi trans-sialidases. Gene 160:
123-128.
70. Todeschini AR, Dias WB, Girard MF, Wieruszeski JM, Mendonca-Previato L, et al.
(2004) Enzymatically inactive trans-sialidase from Trypanosoma cruzi binds
sialyl and beta-galactopyranosyl residues in a sequential ordered mechanism. J
Biol Chem 279: 5323-5328.
71. Todeschini AR, Girard MF, Wieruszeski JM, Nunes MP, DosReis GA, et al. (2002)
trans-Sialidase from Trypanosoma cruzi binds host T-lymphocytes in a lectin
manner. J Biol Chem 277: 45962-45968.
72. Silverman JS, Bangs JD (2012) Form and function in the trypanosomal secretory
pathway. Current opinion in microbiology 15: 463-468.
73. Soares MJ, Souto-Padron T, De Souza W (1992) Identification of a large pre-
lysosomal compartment in the pathogenic protozoon Trypanosoma cruzi. Journal
of cell science 102 ( Pt 1): 157-167.
74. Chiribao ML, Libisch MG, Osinaga E, Parodi-Talice A, Robello C (2012) Cloning,
localization and differential expression of the Trypanosoma cruzi TcOGNT-2
glycosyl transferase. Gene 498: 147-154.
75. Bayer-Santos E, Aguilar-Bonavides C, Rodrigues SP, Cordero EM, Marques AF, et
al. (2013) Proteomic analysis of Trypanosoma cruzi secretome: characterization
of two populations of extracellular vesicles and soluble proteins. Journal of
proteome research 12: 883-897.
76. Maeda Y, Kinoshita T (2011) Structural remodeling, trafficking and functions of
glycosylphosphatidylinositol-anchored proteins. Progress in lipid research 50:
411-424.
77. Cordero EM, Nakayasu ES, Gentil LG, Yoshida N, Almeida IC, et al. (2009)
Proteomic analysis of detergent-solubilized membrane proteins from insect-
developmental forms of Trypanosoma cruzi. Journal of proteome research 8:
3642-3652.

81
78. Gabernet-Castello C, Dubois KN, Nimmo C, Field MC (2011) Rab11 function in
Trypanosoma brucei: identification of conserved and novel interaction partners.
Eukaryotic cell 10: 1082-1094.
79. Essid M, Gopaldass N, Yoshida K, Merrifield C, Soldati T (2012) Rab8a regulates
the exocyst-mediated kiss-and-run discharge of the Dictyostelium contractile
vacuole. Molecular biology of the cell 23: 1267-1282.
80. Lang F, Busch GL, Volkl H (1998) The diversity of volume regulatory mechanisms.
Cellular physiology and biochemistry : international journal of experimental
cellular physiology, biochemistry, and pharmacology 8: 1-45.
81. Go WY, Liu X, Roti MA, Liu F, Ho SN (2004) NFAT5/TonEBP mutant mice define
osmotic stress as a critical feature of the lymphoid microenvironment.
Proceedings of the National Academy of Sciences of the United States of America
101: 10673-10678.
82. Kollien AH, Grospietsch T, Kleffmann T, Zerbst-Boroffka I, Schaub GA (2001) Ionic
composition of the rectal contents and excreta of the reduviid bug Triatoma
infestans. Journal of insect physiology 47: 739-747.
83. Schmatz DM, Murray PK (1982) Cultivation of Trypanosoma cruzi in irradiated
muscle cells: improved synchronization and enhanced trypomastigote production.
Parasitology 85 (Pt 1): 115-125.
84. Moreno SN, Silva J, Vercesi AE, Docampo R (1994) Cytosolic-free calcium
elevation in Trypanosoma cruzi is required for cell invasion. The Journal of
experimental medicine 180: 1535-1540.
85. Bourguignon SC, de Souza W, Souto-Padron T (1998) Localization of lectin-binding
sites on the surface of Trypanosoma cruzi grown in chemically defined
conditions. Histochemistry and cell biology 110: 527-534.
86. de Paulo Martins V, Okura M, Maric D, Engman DM, Vieira M, et al. (2010)
Acylation-dependent export of Trypanosoma cruzi phosphoinositide-specific
phospholipase C to the outer surface of amastigotes. J Biol Chem 285: 30906-
30917.

82
FIGURES
Figure 3.1 Fluorescence microscopy analysis of TcRab11 in different stages of T.
cruzi. (A-C) GFP fusion protein of TcRab11 was detected in the contractile vacuole
bladder of epimastigotes (Epi, A), trypomastigotes (Trypo, B), and intracellular
amastigotes (Ama, C) using antibodies against GFP. Upper panels show differential
interference contrast microscopy (DIC) images merged with DAPI staining of DNA (in
blue) and GFP-TcRab11 (in green). Lower panels show fluorescence images. (D) GFP-
TcRab11 (green) co-localizes with antibodies against T. cruzi aquaporin 1 (-AQP, red),
a marker for the contractile vacuole, under hyposmotic conditions. (E) Antibodies against
TbRab11 (-Rab11, red) co-localize with GFP-TcRab11 (green). (F) Antibodies against
TbRab11 (red) localize to a compartment that resembles the contractile vacuole in (E).

83
DAPI staining is in blue. Arrowheads in D-F show co-localization between antibodies
against TcAQP1 and GFP (D), TbRab11 antibody and GFP (E) and labeling with
antibodies against TbRab11 (F), respectively. Bars in A-F = 10 µm. (G) Western blot
analyses with TbRab11 antibody of lysates of epimastigotes overexpressing GFP-
TcRab11 (E-OE), or wild-type epimastigotes (E), trypomastigotes (T) and amastigotes
(A) showing bands (arrows) corresponding to the endogenous TcRab11 (24 kDa) and to
GFP-TcRab11 (50 kDa). The blots were sequentially probed with TbRab11 and anti-
tubulin antibodies, used as loading control.

84
Figure 3.2 GFP-TcRab11DN localizes to the cytoplasm of different life cycle stages.
(A-C) GFP-TcRab11DN, mimicking the GDP–bound state of the protein has a cytosolic
punctate localization in epimastigotes, (A), trypomastigotes (B), and intracelular
amastigotes (C), as detected using antibodies against GFP. DNA was stained with DAPI.
Bars = 10 µm.

85
Figure 3.3 Regulatory volume changes of epimastigotes (A-B) Cells were pre-
incubated in isosmotic buffer for 3 min and then subjected to hyposmotic (final
osmolarity = 150 mOsm) (A) or hyperosmotic (final osmolarity = 650 mOsm) (B) stress.
Relative change in cell volume was followed by monitoring absorbance at 550 nm by
light scattering. As compared to wild-type cells (WT), cells expressing GFP-TcRab11DN
(DN) failed to fully recover their volume after hyposmotic stress and shrank less after
hyperosmotic stress, while cells overexpressing GFP-TcRab11 (OE) recovered their

86
volume faster after hyposmotic stress and shrank more after hyperosmotic stress. Values
are means ± SD of three different experiments. Asterisks indicate statistically significant
differences, p < 0.05, (Bonferroni’s multiple comparison “a posteriori” test of one-way
ANOVA) at all time points after induction of osmotic stress. (C-D) Epimastigotes were
immobilized on glass slides with poly-lysine and diluted with deionized water to a final
osmolarity of 150 mOsm (C) or bathed with hyperosmotic (650 mOsm) buffer (D). Video
microscopy data were collected and selected frames are shown. Times indicated in each
frame represent 1 second apart after induction of stress. Arrowheads show different
dilated compartments that transform into larger bladders at a later time. Results are
representative of those obtained from at least three independent experiments. Bars = 10
µm.

87
Figure 3.4 Co-localization of GFP-TcRab11 and TcTS during amastigote
differentiation in human foreskin fibroblasts. (A) Expression of TcTS becomes
apparent at 48 h p.i., when antibodies against TcTS (red) co-localize with GFP-TcRab11,
as detected with antibodies against GFP (green). Co-localization progresses to close to
80% of cells by 96 h, and after 106 h co-localization starts to decrease and surface
labeling of TcTS is more evident. Scale bars = 10 µm. Insets shows co-localization at
high magnification (double). (B) Percentage of amastigotes showing co-localization of
TcTS and GFP-Rab11 with time. Two hundred amastigotes were counted in each
experiment and results are expressed as means ± SEM (n = 3).

88
Figure 3.5 Localization of TcTS during differentiation to cell-derived and
metacyclic trypomastigotes. (A) Co-localization of TcTS and antibodies against GFP
in intermediate stages (epimastigote-like) obtained from tissue culture supernatants. (B)
TcTS localizes to patches of the plasma membrane in fully differentiated trypomastigotes
while GFP-TcRab11 remains in the CVC. (C) Co-localization of TcTS with GFP-
TcRab11 in epimastigotes during transformation into metacyclic stages. Scale bars (C-E)
= 10 µm.

89
Figure 3.6 Cryo-immunoelectron microscopy localization of GFP-TcRab11 and
TcTS in amastigotes. Amastigotes were isolated from HFF at different times p.i., as
described under Materials and Methods. GFP-TcRab11 and TcTS were detected with
goat anti-GFP, and rabbit anti-TcTS antibodies, and donkey anti-goat 18 nm colloidal
gold and donkey anti-rabbit 12 nm colloidal gold, respectively. (A-D) Amastigotes
obtained after 96 h p.i. Co-localization of antibodies against GFP (arrows) and TcTS
(small dots) is evident in the CV bladder (CV) and spongiome (Sp), while TcTS also

90
localizes to the flagellar pocket (FP) and in patches of the plasma membrane. Note in (B)
a collapsed bladder and intense labeling of the spongiome. (E-F) Amastigotes obtained
106 h p.i. GFP-TcRab11 localizes to the CV bladder while TcTS localizes to vesicles (V,
small arrows) close to the plasma membrane and in patches in the plasma membrane.
Scale bars = 500 nm. Note that the patchy appearance of the cytoplasm is due to the
absence of glutaraldehyde in the fixative because it abolished labeling of TcTS.

91
Figure 3.7 Overexpression of GFP-TcRab11DN reduces the surface expression of
TcTS. Tissue culture-derived wild type, and GFP-TcRab11DN-expressing
trypomastigotes and intermediate forms were fixed, permeabilized and stained with
antibodies against TcTS (A), or both TcTS and GFP (B and C). Labeling of TcTS (red) in
fully differentiated trypomastigotes was predominantly in surface patches (A). Labeling
of GFP-TcRab11DN (green) was predominantly cytosolic while labeling of TcTS was
punctated but did not reach the cell surface in intermediate forms (B) or fully
differentiated trypomastigotes (C). (D) The fluorescence intensity of TcTS in the cell

92
surface of tissue culture-derived GFP-TcRab11DN-expressing trypomastigotes was
measured in 200 cells in each experiment and expressed as percentage of control (wild-
type trypomastigotes). Values are means ± SEM of 3 independent experiments. **p <
0.05. (E) FACS analysis of fixed GFP-TcRab11DN trypomastigotes reveals a decrease in
the surface expression of TcTS as depicted by their lesser fluorescence intensity (DN) in
comparison to that of wild type cells (WT). The negative control were unstained wild
type trypomastigotes (US) showing background fluorescence. Wild type cells have two
peaks of TcTS, suggesting the presence of intermediate stages in these asynchronously
growing cultures. Data is representative of the profile analysis of 20,000 cells from 3
independent experiments.
Figure 3.8 Localization of surface proteins in GFP-TcRab11OE and GFP-
TcRab11DN-expressing parasites. Antibodies against TcTSSA II (red) co-localize with

93
antibodies against GFP (green) in intermediate forms (A) and amastigotes (B) but not in
trypomastigotes expressing GFP-TcRab11, where they localize to the plasma membrane
(C). Antibodies against TcTSSA II (D) still localize to the plasma membrane in GFP-
TcRab11DN-expressing cells, while antibodies against the H+-ATPase (E) maintain their
intracellular and plasma membrane localization in GFP-Rab11DN-expressing cells. In
(D) and (E) GFP staining localizes to the cytosol. Scale bars = 10 µm.
Figure 3.9 Localization of anti-Gal antibodies. (A) GFP-TcRab11 co-localizes with the
anti-Gal antibodies in the CVC of the intermediate forms as detected by polyclonal
antibody against GFP (green arrow) and anti-α-galactosyl antibodies from patients with
chronic Chagas disease (red arrow), respectively. (B) Anti-Gal antibodies strongly label
the surface of fully differentiated tissue culture derived trypomastigotes while GFP-
TcRab11 labels the CVC. (C) GFP-TcRab11DN mutants show a punctated cytosolic

94
localization (green) while anti-Gal antibodies (red) localize to the plasma membrane in
intermediate stages. Scale bars (A-C) = 10 µm
Figure 3.10 Association of CVC proteins with lipid rafts and reduced infectivity of
TcRab11DN trypomastigote. (A) Parasite extracts were loaded at the bottom (fraction
9) of a discontinuous Optiprep density gradient and subjected to ultracentrifugation.
Fractions were collected and analyzed by anti-GFP and anti-FCaBP immunoblotting.
Fractions 2 and 3 contain the lipid raft interface. The TcSNARE2.1GFP (SNARE), GFP-
TcRab11 (Rab11), and FCaBP floated to the lipid raft interface. Lanes 6-9 represent the
heavier fractions of the GFP and FCaBP derivatives and GFP alone was detected in these
fractions. A whole cell lysate (WCL) is included in each panel as a control of loading.
Total protein in lysates of GFP-TcSNARE2.1-, GFP-TcRab11- and GFP-expressing
epimastigotes were 1.41, 1.3 and 1.39 mg/ml, respectively. (B) T. cruzi expressing GFP-
TcRab11 or GFP were solubilized in Triton X-100 at 4°C or 37°C and separated into

95
soluble (S) and insoluble (P) fractions and analyzed by western blotting with anti-GFP.
Rab11-GFP partitions in the pellet fraction at 4°C, but is solubilized at 37°C, whereas
GFP is only detected in the soluble fraction. (C-D) Effect of TcRab11 overexpression
(OE) or mutation (DN) on trypomastigote invasion of host cells. In vitro infection assays
were carried out as described under Materials and Methods. (E-F) Partial rescue of the
infectivity of DN trypomastigotes by their incubation in the presence of active TcTS and
sialofetuin, whereas inactive trans-sialidase (iTS) does not rescue the infectivity of GFP-
TcRab11DN mutants. Fetuin was present in all samples. Other conditions under
Materials and Methods. Values in C-F are mean ± SD (n = 3). *, ** and *** indicate that
differences are statistically significant compared with respective controls, p < 0.05
(Ordinary one way ANOVA with Bonferroni post-test).

96
Supplementary figures
Figure 3.11 Cryo-immunoelectron microscopy localization of GFP-TcRab11 in
epimastigotes. Epimastigotes were isolated and submitted to hyposmotic stress as
described under Materials and Methods. GFP-TcRab11 was detected with rabbit anti-
GFP, and anti-rabbit 18 nm colloidal gold. GFP-TcRab11 localizes mainly to the CV
bladder. Arrows in C show labeling of the dilated spongiome (Sp) tubules. CV;
contractile vacuole bladder; Sp: spongiome; Fl, flagellum; K, kinetoplast. Scale bars =
100 nm.

97
Figure 3.12 Growth rate, and western blot analyses of overexpressed TcRab11. (A)
Growth rate of epimastigotes overexpressing (OE, blue) or expressing the dominant
negative (DN, green) mutant of TcRab11, as compared to controls (C, red). (B) Western
blot analyses of GFP-TcRab11OE (OE), GFP-TcRab11DN (DN) and GFP-expressing
(GFP) epimastigotes. Membranes were stripped and re-incubated with anti-tubulin
antibody as a loading control (bottom panel). (C) Densitometry analysis of western blots
of lysates from TcRab11 overexpressing epimastigotes (OE) as compared to those of
control cells. Values in arbitrary units (AU) correspond to mean ± SD from 3
independent experiments. (D) Western blot analyses of GFP-TcRab11OE (OE), GFP-
TcRab11DN (DN) and GFP-expressing (GFP) trypomastigotes. Membranes were
stripped and re-incubated with anti-tubulin antibody as a loading control (bottom panel).

98
Figure 3.13 TcAQP1 localization is not affected in GFP-TcRab11DN mutants and
western blot analysis of wild type and GFP-TcRab11DN shows specificity of anti-
SAPA antibodies. (A) Co-localization of GFP-TcRab11, as detected with antibodies
against GFP (green arrow), with antibodies against TcAQP1 (-TcAQP, red arrow) in
epimastigotes. (B) GFP-TcRab11DN mutants show a punctated cytosolic localization as
detected with anti-GFP (green), while antibodies against TcAQP1 still localize to the
CVC (red arrows). Co-localization is indicated in Merge images (yellow arrows). Bars =
10 µm. (C) Western blot analyses of GFP-TcRab11DN (DN), and wild type (WT)
trypomastigotes using anti-SAPA antibodies. Membranes were stripped and re-incubated
with anti-tubulin antibody as a loading control (bottom panel).

99
Figure 3.14 Localization of GFP-TcRab11 and gp35/50 mucins during
metacyclogenesis. (A) GFP-TcRab11 co-localizes with gp35/50 mucins in the CVC of
intermediate forms, as detected with polyclonal antibody against GFP (green arrow), and
monoclonal antibody 2B10 (red arrow), respectively. Surface localization of gp35/50 is
also evident (red). (B) GFP-TcRab11 (green arrows) does not co-localize with gp35/50
mucins, which have a surface localization in metacyclic trypomastigotes (red). (C) GFP-
TcRab11DN mutants show a punctated cytosolic localization of TcRab11DN (green)
while gp35/50 mucins (red) localize to the plasma membrane in intermediate stages.
Scale bars (A-C) = 10 µm.

100
Figure 3.15 Infections of host cells by trypomastigotes overexpressing TcRab11. A-
B. TcRab11 overexpression (OE) does not cause significant changes in trypomastigote
invasion of host cells as compared to wild type trypomastigotes. In vitro infection assays
were carried out as described under Materials and Methods. (C-D). Recombinant active
trans-sialidase rescues the infectivity of GFP-TcRab11DN mutants in the presence of
fetuin (F) but not in the presence of asialofetuin (A). Other conditions under Materials
and Methods.

101
CHAPTER 4
RAB32 IS ESSENTIAL FOR MAINTAINING FUNCTIONAL
ACIDOCALCISOMES AND FOR GROWTH AND VIRULENCE OF
TRYPANOSOMA CRUZI
Sayantanee Niyogi, Veronica Jimenez and Roberto Docampo. (To be submitted to PLoS
Pathogens)

102
Abstract
We recently reported that the contractile vacuole complex (CVC) of Trypanosoma cruzi,
the etiologic agent of Chagas disease, is involved in the transfer of GPI-anchored proteins
to the plasma membrane of the parasite during its differentiation to trypomastigotes and
that the CVC-located small GTPase TcRab11 is essential for the specific transfer of
trans-sialidase. Here we report that another CVC-located small GTPase, TcRab32, is
important for acidocalcisome function, suggesting its involvement in trafficking of
membrane proteins to these organelles. TcRab32 is geranylgeranylated and localizes to
the CVC. A dominant negative (DN) mutant tagged with GFP (GFP-TcRab32DN)
localizes to the cytosol, and epimastigotes expressing this dominant negative mutant are
less responsive to osmotic stress. Mutant parasites are still able to differentiate into
metacyclic forms and infect host cells but they are less virulent than wild type cells.
Parasites expressing GFP-TcRab32DN have a reduced number of acidocalcisomes, which
are deficient in pyrophosphate (PPi) and polyphosphate (polyP), and are less electron-
dense as compared to acidocalcisomes in wild type cells. Taking together these results
reveal roles of TcRab32 in osmoregulation and trafficking of membrane proteins to
acidocalcisomes and indicate that the CVC is a trafficking hub in these parasites.
Author Summary
The contractile vacuole complex (CVC) consists of a large vacuole or bladder and a loose
network of tubules known as the spongiome. In addition to its role in osmoregulation, the
CVC of Trypanosoma cruzi has a role in trafficking of GPI-anchored proteins to the
plasma membrane of differentiating cells. In this work we reveal that its role is not
limited to the traffic of GPI-anchored proteins to the plasma membrane but also includes

103
the traffic of membrane proteins to acidocalcisomes. Expression of dominant negative
mutants of the CVC-located GFP-TcRab32 results in acidocalcisomes of altered
morphology and content and less virulent parasites revealing the similarity of its role to
that of early/recycling endosomes.
Introduction
Trypanosoma cruzi [1], the etiologic agent of Chagas disease, together with Leishmania
spp. [2], and a number of monogenetic trypanosomes [3,4], possess a contractile vacuole
complex (CVC) involved in osmoregulation. In T. cruzi, the CVC was shown to be
important for regulatory volume decrease (RVD) after hyposmotic stress [5], and for
shrinking of the cells when submitted to hyperosmotic stress [6]. In addition, we recently
reported a role for the CVC in trafficking glycosylphosphatidylinositol(GPI)-anchored
proteins to the plasma membrane [7]. Previous studies in T. cruzi [8] and Dictyostelium
discoideum [9-12] suggested that other soluble [9,10] and membrane [8,11,12] proteins
can also be transported through the CVC to the plasma membrane. The presence of
Rab11, a small GTPase that localizes in recycling endosomes in most cells, including T.
brucei [13], in the CVC of T. cruzi [14] and D. discoideum [15], suggested that the CVC
could be an evolutionary precursor to the recycling endosomal system in other eukaryotes
[15,16].
In a previous proteomic and bioinformatics study of the CVC of T. cruzi we identified a
number of proteins involved in trafficking roles, among them SNAREs 2.1 and 2.2,
VAMP1 (VAMP7 homolog), AP180, and the small GTPases Rab11 and Rab32 [14]. It
was verified by immunofluorescence that indeed these proteins localize to the CVC. Rab
proteins mediate tethering of incoming vesicles to the correct target organelle through

104
cycling between a GDP-bound inactive and a GTP-active form [17]. They have also been
implicated in vesicle budding and in the interaction with cytoskeletal elements [17].
Different Rab GTPases are localized to different organelles and this represents an
important determinant of each organelle identity [18-20]. Rab32 and its close homolog
Rab38 are predominantly expressed in lysosome-related organelles-(LROs)-producing
cells such as melanocytes, and platelets [21], and it has been suggested that these Rabs
could be the specificity factors that work in concert with the ubiquitous trafficking
machinery for transport toward LROs [21]. It has been proposed that LROs arise by
delivery of specific cargoes from the early endosomal network, comprising sorting and
recycling endosomes [22,23].
T. cruzi possesses organelles with similarities to LROs of mammalian cells, known as
acidocalcisomes [24-26]. As LROs of human platelets [27,28] and mast cells [29],
acidocalcisomes have rounded morphology, are acidic, and rich in calcium,
pyrophosphate (PPi) and polyphosphate (polyP). In addition, adaptor protein complex-3
(AP-3), the system known to be involved in transport of membrane proteins to LROs of
mammalian cells [30], is also involved in the biogenesis of acidocalcisomes [31,32].
Interestingly, electron microscopy evidences of fusion of acidocalcisomes to the CVC of
T. cruzi [33], and D. discoideum [34] have been reported. Also, under hyposmotic stress
acidocalcisomes fuse to the CVC and results in translocation of an aquaporin (TcAQP1)
[5]. In this work we demonstrate that the expression of dominant-interfering TcRab32
mutants altered osmoregulation, acidocalcisome number and content, and parasite
infectivity. The results suggest that the CVC and TcRab32 are involved in trafficking
membrane proteins involved in acidocalcisome biogenesis, and reaffirm the role of the

105
CVC as a trafficking hub.
Results
Localization of TcRab32 in different T. cruzi stages:
We reported that N-terminal tagging of T. cruzi Rab32 (TcRab32; TcCLB.506289.80)
with green fluorescent protein (GFP) resulted in labeling of the CVC of epimastigotes
and an additional punctated staining [14]. We confirmed this localization by indirect
immunofluorescence analysis using an affinity purified TcRab32 antibody raised in
mouse against the recombinant protein. For the generation of antibody against the
protein, we expressed TcRab32 in Escherichia coli as a fusion protein with a C-terminal
polyhistidine tag. Recombinant Rab32 proteins was purified by affinity chromatography
using His-Bin cartridges and fractions were verified by SDS PAGE. Fig. 4.1A shows that
the bacterially expressed rTcRab32 protein (including the His-tag) appears as a strong
single band with an approximate molecular mass of 42 kDa. Fig. 4.1B-D shows that
αTcRab32 localizes to the bladder of the CVC of wild type epimastigotes,
trypomastigotes, and amastigotes (arrows), with additional punctated staining especially
in epimastigotes and trypomastigotes. This antibody was shown to predominantly react
with a protein of 26 kDa in all T. cruzi stages (Fig. 4.1E).
In vitro prenylation studies of TcRab32
TcRab32 possesses the sequence CXC at the carboxyl terminus (Fig. 4.2A; denoted by
red overlap) and it is known that Rab prenylation at Cys residues of the carboxyl
terminus retain Rabs at membranes [35]. Previous studies using recombinant T. cruzi
protein geranylgeranyl transferase I (GGTI) using a panel of mammalian and yeast
protein substrates reported that two mammallian Rab family GTPases containing the C-

106
terminal CXC sequence did not serve as substrates for this enzyme, as expected [36].
Accordingly, Prenylation Prediction Suite (PrePS) predicts that geranylgenanyl
transferase II (GGTII) is the enzyme involved in the prenylation of this protein. To
examine whether TcRab32 is geranylgeranylated, we performed in vitro prenylation
experiments (Fig. 4.2B) using recombinant Rab32 (rRab32) as substrate in the presence
of a cytosolic epimastigote extract as the source of prenyltransferases. When tritiated
geranylgeranyl pyrophosphate ([3H]GGPP) was used as the isoprenoid donor, His-tagged
TcRab32 was efficiently geranylgeranylated as shown by the labeled band of 42 kDa
detected, corresponding to the His-tagged protein. The intensity of the prenylated band
was strongest at 30 min, the optimum incubation time. Conversely, when tritiated
farnesyl pyrophosphate ([3H]FPP) was used as donor, we were unable to detect
prenylation of rTcRab32, even after exposure of the gel for more tan 2 weeks at -80°C
(data not shown). Therefore, TcRab32 is specifically geranylgeranylated.
Localization of TcRab32 mutants
To examine the role of the prenylation motif in targeting of TcRab32 to cell membranes,
we generated mutants in which the prenylation motif was mutated and we studied the
effect of this mutation on the localization of the protein. An N-terminal GFP epitope tag
was fused to TcRab32 in which the C-terminal Cys residues were mutated to Ala. In
transfected T. cruzi epimastigotes GFP-TcRab32C241A/243A had a cytosolic
localization (Fig. 4.3A). We also constructed an expression plasmid encoding a TcRab32
mutant that mimics the GDP-bound form (dominant-negative; TcRab32T24N) (Fig.
4.3B) or the GTP-bound form (dominan-positive; TcRab32Q71L) (Fig. 4.3C). In
transfected T. cruzi epimastigotes, GFP-TcRab32DN have a punctated cytosolic

107
localization while GFP-TcRab32DP have a preferential localization in the CVC.
Together, these results suggest that TcRab32 localizes to the membrane of the CVC in a
GTP-dependent manner with the COOH-terminal cysteines.
Lack of co-localization of GFP-TcRab32 with a mitochondrial marker
It has been reported that mammalian Rab32 functions as an A-kinase anchoring protein
(AKAP), interacting with the type II regulatory subunit (RII) of protein kinase A (PKA)
and associating to the mitochondria [37]. An Ala at position 185 in the α5 helix acts as an
anchoring determinant and introduction of a phenylalanine, which is conserved in this
position in most Rab family members, prevents binding to RII. Interestingly, TcRab32
possesses a phenylalanine (F) at the equivalent position (Fig. 4.2A; denoted by blue
asteriks), and, as expected, GFP-TcRab32 does not co-localize with the mitocondrial
marker Mitotracker (Fig. 4.4A), and neither GFP-TcRab32DP (Fig. 4.4B) nor GFP-
TcRab32DN (Fig. 4.4C) affects mitocondrial labeling.
Co-localization of GFP-TcRab32 with TcVP1 under osmotic stress
It has been reported that mammalian [38] and Xenopus [39] Rab32 partially localizes to
melanosomes, which are LROs. We therefore investigated whether TcRab32 partially co-
localizes with the acidocalcisome marker vacuolar proton pyrophosphatase (VP1) [40].
We did not observe any significant overlap between antibodies to TbVP1 and against
GFP-TcRab32 under isosmotic conditions (Fig. 4.5A). However, under hyposmotic
conditions (Fig. 4.5B) we observed that TcVP1 staining overlaps with GFP staining at the
CVC region, in agreement with the reported fusion of these organelles under hyposmotic
stress [5,33].

108
TcRab32DN mutants have a decreased content of PPi and polyP
Most PPi and polyP in trypanosomes are accumulated in acidocalcisomes [25,26]. It is
not known whether PPi is taken up from the cytosol or synthesized inside
acidocalcisomes while synthesis of polyP is through the activity of polyP kinases such as
that formed by the vacuolar transporter chaperone (VTC) complex [41,42]. This is a
complex of at least two subunits in trypanosomatids, VTC1 and VTC4, both localized to
the membrane of acidocalcisomes and of which VTC4 is the catalytic subunit [41,42].
We hypothesized that if TcRab32 was important for the biogenesis of acidocalcisomes
these organelles would have a reduced ability to synthesize these compounds and that
was exactly the case. Expression of the dominant-negative form of TcRab32 (GFP-
TcRab32DN) led to a significant reduction in the levels of PPi (~50%) (4.6A) and short-
chain polyP (~80%) (4.6B) in comparison to GFP and wild type Rab32 expressing
epimastigotes. There was, however, no significant change in the expression of long chain
polyP (>300 up to 700–800 phosphate units) (4.6C), suggesting that only the activity of
TcVTC complex, which is mainly involved in the synthesis of short-chain polyP [41,42],
is affected in these mutants. The results were further verified by visualization of short
chain poly P extracted from the above cell lines, resolved by Urea-PAGE and stained
with toluidine blue (4.6D).
Changes in acidocalcisome electron-density and number in GFP-TcRab32DN
mutants
In previous work [43,44], electron microscopy techniques were used to demonstrate that
treatment of fixed trypanosomes with yeast pyrophosphatase resulted in loss of the
electron-density of acidocalcisomes, as observed in whole unstained cells, suggesting that

109
PPi (complexed with cations) was the main electron dense material of these organelles. In
agreement with the considerable decrease in PPi and short chain polyP content of GFP-
TcRab32DN mutants (Fig. 4.6), we detected by transmission electron microscopy on
intact unstained GFP-TcRab32DN (DN) expressing epimastigotes the presence of empty
vacuoles in comparison to wild type epimastigotes (WT) (Fig. 4.7A) and that there was a
significant reduction in the number of acidocalcisomes per cell (B). 84% of these DN
cells have acidocalcisomes that were not electron-dense (C) with an average of ~10
empty vacuole per cell expressing GFP-TcRab32DN (D).
Cells deficient in Rab32 display no defect in the traffic of trans-sialidase to the
plasma membrane
To investigate whether TcRab32 affects traffic of trans-sialidase (TcTS) to the cell
surface of trypomastigotes, or this is a process specific for TcRab11 [7], we infected Vero
cells with metacyclic trypomastigotes from stationary cultures of GFP-TcRab32DN
parasites and obtained cell culture-derived trypomastigotes. GFP-TcRab32DN
trypomastigotes were used to infect fibroblasts and labeling of TcTS was detected by
indirect immunofluorescence analysis using antibodies against the SAPA repeats of TcTS
after a full cycle of differentiation into trypomastigotes. Traffic of trans-sialidase to the
surface was not affected in TcRab32-DN trypomastigotes, further demonstrating that the
CVC-dependent trafficking pathway of trans-sialidase is specifically TcRab11-mediated
(Fig. 4.8).
GFP-TcRab32DN mutants have reduced growth and response to osmotic stress
The growth rate of the epimastigotes expressing GFP-TcRab32DN (DN) mutants was
significanlty reduced as compared to that of control epimastigotes expressing GFP alone

110
(C) (Fig. 4.9A). Wild-type, GFP-TcRab32-overexpressing (GFP-TcRab32OE), GFP-
TcRab32DP, and GFP-TcRab32DN-expressing epimastigotes were submitted to
hyposmotic stress and their regulatory volume decrease (RVD) measured using a light-
scattering technique, as described previously [7]. This technique measures the changes in
volume of the cells under hyposmotic (swelling and recovery) and hyperosmotic
conditions (shrinking and partial recovery). After recovery the cells recuperate their
normal morphology (Fig. 4.9B). DN mutants were less able to recover their volume after
hyposmotic stress than wild type cells, while recovery was faster in GFP-TcRab32OE
cells (OE). The response in TcRab32DP cells was similar to that of wild type cells. In
addition, when submitted to hyperosmotic stress (Fig. 4.9C), DN mutants shrank less
while GFP-TcRab32OE and GFP-TcRab32DP cells shrank more than control cells, and
in all cases they did not recover their volume during the time of the experiment. It has
been shown before that when epimastigotes are submitted to hyperosmotic stress the
parasites do not regain their normal volume at least during the subsequent two hours [6].
TcRab32 is required for infection
To study the effect of reduced polyP, and PPi levels and the effect of reduced cell
viability on the rate of invasion of the GFP-TcRab32DN mutants, we fully differentiated
them into cell derived trypomastigotes as described under Materials and Methods.
Invasion was significantly reduced in GFP-TcRab32DN and GFP-TcRab32OE mutants
as compared with controls transfected with GFP alone or with wild type parasites (Fig
4.10A and 4.10B). Cytosolic localization of GFP-TcRab32DN mutants was maintained
when epimastigotes were differentiated into trypomastigotes and intracellular amastigotes
(Fig. 4.10C).

111
Discussion
We show here that expression of dominant-negative form of the GTPase TcRab32 results
in alterations in the number and content of acidocalcisomes, and in deficient response to
osmotic stress, growth in vitro, and invasion of host cells. The results suggest that the
CVC, where TcRab32 is located, is involved in trafficking membrane proteins involved
in the synthesis/transport of phosphorus compounds to acidocalcisomes.
We reported before that GFP-tagged TcRab32 localizes to the CVC of epimastigotes of
T. cruzi [42]. We now confirm those observations using antibodies against the protein
and found it distributed in the CVC of different stages of the life cycle of the parasite and
with a punctate staining in epimastigotes and amastigotes. Mutants deficient in the
prenylation motif and dominant negative GFP-TcRab32, however, have a cytosolic
localization indicating that CVC localization is geranylgeranylation- and GTP-dependent.
Dominant negative TcRab32 mutants might be acting by blocking or reducing the
function of endogenous TcRab32, by competing or sequestering Rab32 effector proteins.
TcRab32, like other Rab32 proteins, contains amino acid sequences that are shared with
only a small number of other Rab sequences [45]. For example, threonine in the
WDTAGQE sequence (GTP binding site), which is conserved in almost all Rab proteins,
is replaced by isoleucine. A similar replacement is found in Rab38, Rab29, and
Rab7L1/29 of mammalian cells, and in RabE from Dictyostelium discoideum [45], but
there are no orthologs to any of these other Rabs in T. cruzi [46]. TcRab32 also possesses
three amino acids, the Gly at amino acid position 75, Asn-76, and Val-80 that are only
conserved in the switch II region of Rab32 and Rab38 alone and not of any of the other
58 Rabs of mammalian cells [47]. Val-80 is required for binding of mammalian Rab32 to

112
its effector VPS9-ankyrin-repeat protein/Ankrd27 (Varp) and this interaction is important
for trafficking of tyrosinase-related protein to melanosomes [47]. In contrast, TcRab32
has a phenylalanine at amino acid position 194 instead of an alanine in mammalian
Rab32. This Ala is an anchoring determinant for regulatory subunit II (RII subunit) of
protein kinase A and responsible for mammalian Rab32 interaction with mitochondria
[37]. In agreement with those studies we found that TcRab32 does not associate with
mitochondria. Interestingly, other authors were also unable to confirm the association of
human Rab32 with COS cells mitochondria [45]
Traffic of trans-sialidase to the plasma membrane of trypomastigotes is not affected in
GFP-TcRab32DN expressing parasites suggesting that the trafficking pathways of
membrane proteins to the plasma membrane and acidocalcisomes are independent of
each other.
Rab proteins participate in membrane trafficking events involving membrane fusion,
fission, and motility. Although our data do not distinguish between these events, the
localization of TcRab32 in the CVC and the deficient morphology and content of
acidocalcisomes upon expression of its dominant-negative form suggests that the CVC
acts equivalent to the recycling/early endosomes of mammalian cells where TcRab32
functions as tether facilitating cargo loading into fused vesicles [48]. The fusion of CVC
with acidocalcisomes would facilitate exchange of membrane proteins between the
organelles such as translocation of TcAQP1 from acidocalcisomes to the CVC [5] or of
membrane enzymes/transporters involved in the synthesis of phosphorus compounds
from the CVC to the acidocalcisomes. This model would be consistent with the known
interaction between Rab32 effector proteins and VAMP7 [38,47], which is a vesicle

113
SNARE protein involved in vesicle fusion, and its known interaction with the delta
subunit of adaptor complex-3 (AP-3) [49]. An ortholog to VAMP7 is present in the CVC
of T. cruzi [14] and AP-3 is known to be involved in the biogenesis of acidocalcisomes
[31,32].
In conclusion, we propose that the CVC is a trafficking hub involved not only in the
transfer of GPI-anchored proteins to the plasma membrane but also as a specialized
endosomal system that can be used to deliver membrane proteins important for the
biogenesis of acidocalcisomes.
Materials and Methods
Cell culture
Epimastigotes from T. cruzi were cultured in liver infusion tryptose (LIT) medium
containing 10% newborn serum at 28°C. T. cruzi epimastigotes transfected with GFP-
TcRab32OE, GFP-TcRab32DN, GFP-TcRab32DP and GFP-TcRab32C241A/243A were
maintained in the presence of 250 µg/ml geneticin (G418). Human foreskin fibroblasts
(HFF) were grown in DMEM Low Glucose medium supplemented with 10% Cosmic
Calf serum and 0.1% L-glutamine. Vero cells were grown in RPMI supplemented with
10% fetal bovine serum. L6E9 myoblasts were grown in DMEM High Glucose medium
supplemented with 10% fetal bovine serum. Host cells were maintained at 37°C with 5%
CO2. Tissue culture cell-derived trypomastigotes were obtained from Vero cells infected
with metacyclic trypomastigotes from stationary cultures of GFP-TcRab32OE and GFP-
TcRab32DN parasites. T. cruzi amastigote and trypomastigote forms were collected from
the culture medium of infected host cells, using a modification of the method of Schmatz
and Murray [50] as described previously [51].

114
Chemicals and reagents
Fetal bovine serum, newborn calf serum, Dulbecco’s phosphate buffer saline (PBS) and
Hank’s solution, 4’,6-diamidino-2-phenylindole (DAPI), DMEM and RPMI media,
paraformaldehyde, bovine serum albumin, and protease inhibitors were purchased from
Sigma (St. Louis, MO). Restriction enzymes, were from New England BioLabs (Ipswich,
MA). pCR2.1-TOPO cloning kit, 1 kb plus DNA ladder, rabbit GFP antibodies and Gene
Tailor Site-Directed Mutagenesis System were from Invitrogen (Life Technologies,
Grand Island, NY). Hybond-N nylon membranes were obtained from PerkinElmer
(Waltham, MA). Pierce ECL Western blotting substrate and BCA Protein Assay Reagent
was from Pierce (Thermo Fisher Scientific, Rockford, IL). All other reagents were
analytical grade. The oligonucleotides were ordered from Sigma or IDT (Coralville, IA).
Vector pET32 Ek/LIC, Benzonase® Nuclease, anti-Histidine tag antibodies, and S-
protein HRP conjugate were from Novagen (EMD Millipore, Billerica, MA). Farnesyl
Pyrophosphate, [1-3H(N)]-, Triammonium Salt, 1mCi (37MBq), Geranylgeranyl
Pyrophosphate, Triammonium Salt,[1-3H(N)]-, 50µCi (1.85MBq) and EN3HANCE were
from Perkin Elmer.
In vitro infection assay
HFF or irradiated myoblasts (6 x 105 cells per well) were equally distributed in a 12-well
plate on a sterile coverslip in their respective growth media (as mentioned above) and
were incubated for 24 h at 37°C in a 5% CO2 atmosphere. The following day, the cells
were washed once with Dulbecco’s Hank’s solution, and 6 x 106
wild type, TcGFP, GFP-
TcRab32OE, or GFP-TcRab32DN trypomastigotes were added to each well (10
trypomastigotes per myoblast or HFF), and they were incubated for 4 h at 37°C in a 5%

115
CO2 atmosphere. To decrease the chances of contamination of cell derived-
trypomastigotes with extracellular amastigotes, collections of parasites were centrifuged
and incubated at 37°C for 2 h to allow trypomastigotes to swim to the surface. The
supernatant was collected and used for subsequent invasion assays. Next, the parasites
were removed from the plate, and the infected cells were washed extensively with
Dulbecco’s Hank’s solution and fixed for immunofluorescence assays. For
attachment/internalization assays, recently internalized parasites, and parasites caught in
the process of invasion, were considered and manually counted in at least 200 DAPI-
stained cells in 3 independent experiments. The percentage of infected cells and the
average number of parasites per infected cell were determined.
Immunofluorescence and western blot analyses
To determine if there are domains of contact between TcRab32 with the mitochondria in
T. cruzi epimastigotes, live cells were labelled for 30 min with Mitotracker Red CMXRos
(Invitrogen) at 50 nM in LIT medium and then fixed and processed for
immunofluorescence. For immunofluorescence microscopy, parasites were fixed in PBS,
pH 7.4, with 4% paraformaldehyde, adhered to poly-lysine coverslips, and permeabilized
for 3 min with PBS, pH 7.4, containing 0.3% Triton X-100. Permeabilized cells were
quenched for 30 min at room temperature with 50 mM NH4Cl and blocked overnight
with 3% BSA in PBS, pH 8.0. Both primary and secondary antibodies were incubated for
1 h at room temperature. Coverslips were mounted by using a mounting medium
containing DAPI at 5 µg/ml for staining DNA-containing organelles. For imaging of
intracellular parasites, mammalian cells were seeded onto sterile coverslips in 12-well
culture plates and allowed to grow for 24 h. To semi-synchronize the infection, we added

116
the parasites at a ratio of 10:1 (parasite/host cell) for 4 hours, washed the cells to
eliminate extracellular parasites and fixed in cold methanol for 30 min. The dilution used
for primary antibodies were as follows: mouse anti-Rab32 (1:200), rabbit polyclonal anti-
GFP (1:500); rabbit anti-TcTS (1:2000), polyclonal rabbit anti-TbVP1 (1:250) [40].
Differential interference contrast (DIC) and direct fluorescence images were obtained by
using an Olympus IX-71 inverted fluorescence microscope with a Photometrix
CoolSnapHQ charge-coupled device camera driven by Delta Vision softWoRx3.5.1
(Applied Precision, Issaquah, WA). Images were deconvolved for 10 cycles using the
same software and applying the “noise filter” at “medium” mode. This is an automatic
deconvolution software and was applied to all channels; brightness and contrast were the
same in all channels. The figures were built by using Adobe Photoshop 10.0.1 (Adobe
System, Inc., San Jose, CA).
Generation of TcRab32 dominant negative, dominant positive and prenylation-
motif mutant and transfection
Dominant negative (GFP-TcRab32T24N), dominant positive (GFP-TcRab32Q71L) and
prenylation-motif mutant (GFP-TcRab32C241A/243A) forms of TcRab32 were
constructed via site directed mutagenesis by the use of Gene Tailor Site-Directed
Mutagenesis System. This method involved methylating the TOPO blunt end vector
containing the coding sequence for TcRab32 with DNA methylase at 37°C for 1 hour,
followed by amplification of the plasmid in a mutagenesis reaction with two overlapping
primers, of which the forward primer had the target mutation, resulting in the mutation of
amino acid threonine to asparagine (dominant negative), glutamine to leucine (dominant
positive), or Cysteine to Alanine (prenylation-motif mutant). Mutations were confirmed

117
by sequencing (Yale DNA Analysis Facility, Yale University, New Haven, Connecticut).
After transformation the resulting plasmids in TOPO was digested with restriction
enzymes BamHI and HindIII. The circular pTEX-N-GFP vector was linearized by the
corresponding restriction enzymes. Finally, TcRab32T24N, TcRab32Q71L and
TcRab32C241A/243A inserts were ligated to pTEX-N-GFP followed by transformation.
The plasmid pTEX-GFPTcRab32T24N/Q71L/C241A/243A were sequenced to confirm
that the correct reading frame was used. T. cruzi Y strain epimastigotes were transfected
in cytomix (120 mM KCl, 0.15 mM CaCl2, 10 mM K2HPO4, 2 mM EDTA, 5 mM
MgCl2, pH 7.6) containing 50 μg of the plasmid construct in a 4 mm cuvette. The cuvette
was cooled on ice for 10 min and pulsed 3 times (1.5 kV, 25 μF) with a Gene Pulser
Xcell™ (Bio-Rad), and expression of GFP-fusion proteins was verified by western blot
analyses. Stable cell lines were established under drug selection with G418 at 250 μg/ml.
Enrichment of GFP fluorescent parasites was performed with a high-speed cell sorter
when needed (MoFlo Legacy; Beckman-Coulter, Hialeah, FL).
Cell volume measurements
T. cruzi epimastigotes (GFP-TcRab32OE, GFP-TcRab32DN, GFP-TcRab32DP and wild-
type) at log phase of growth (3 days) were collected at 1,600 g for 10 min (at a density of
1 x 108/ml) and volume measurement experiments after stress were done exactly as
described in [7].
Recombinant protein expression, purification and antibody generation
DNA sequence corresponding to the entire open reading frame of TcRab32 was PCR-
amplified from T. cruzi Y strain gDNA
(Forward primer: 5' GACGACGACAAGATGTCATACTCGAA -3', Reverse primer:

118
5' GAGGAGAAGCCCGGTTTAACAGGAGCAGCCCGAC-3') and ligation-
independent cloned into vector pET32 Ek/LIC for heterologous expression in bacteria.
The sequence of several recombinant clones was verified and they were transformed by
heat shock into E. coli BL21 Codon Plus (DE3)-RIPL chemically competent cells.
Expression of recombinant protein was obtained by induction in 0.5 mM isopropyl-β-
Dthiogalactopyranoside (IPTG) in LB broth overnight at 37° C. His-tagged recombinant
protein was purified under denaturing conditions with His-Bind cartridges (Novagen).
Recombinant TcRab32 was used as immunogen for production of polyclonal antibody in
mice. This antibody was generated at the Monoclonal Antibody Facility of the College of
Veterinary Medicine, University of Georgia (Athens, GA).
In-vitro prenylation
In vitro prenylation reactions were done as described in [52] and [53] with minor
modifications. A total of 2 µCi of [3H] FPP or [
3H] GGPP was used as isoprenoid
donors. The assay reaction was carried out at 30°C for 30 min, 1 hour and 3 hour and 30
min was the optimum reaction time for this assay and resolved by SDS–10% PAGE. The
gel was incubated in En3Hance, dried, and exposed to film at -80°C for 2 weeks.
Short chain and long chain polyphosphate quantification
Cells (2 x 108) in log phase were harvested and washed twice with buffer A. The PPi and
short-chain polyP were extracted using 0.5 M perchloric acid (HClO4) [54], and the long-
chain polyP was extracted using glass milk (Molecular Probes) as described [55]. PPi
level was determined by the amount of Pi released upon treatment with an excess of
Saccharomyces cerevisiae inorganic pyrophosphatase (catalog no. I-1891, Sigma). The
free Pi (released) amount was determined by using a standard curve. Briefly, the

119
enzymatic reaction was performed on 96-well plates with 50 mM Tris-HCl (pH 7.4), 6
mM MgCl2, inorganic pyrophosphatase, and extracted PPi samples at a final volume of
100µl. After incubation at 30 °C for 10 min, the reaction was immediately stopped by the
addition of an equal amount of the fresh mixture of 3 parts of 0.045% malachite green
with 1 part of 4.2% ammonium molybdate (Sigma), which was filtered prior to use. The
absorbance at 660 nm was read using a SpectraMax M2e plate reader (Molecular
Devices, Sunnyvale, CA).
Short-chain and long-chain polyP levels were determined by the amount of Pi released
upon treatment with an excess of the purified recombinant exopolyphosphatase of S.
cerevisiae (rScPPX1) freshly purified in our laboratory (Ruiz et al., 2001).
Short chain PolyP extracted from 5 x 108 cells were mixed with 6X Dye (0.01% Orange
G; 30% glycerol; 10 mM TrisHCl pH 7.4; 1 mM EDTA) and resolved on 20% TBE
PAGE. Samples were run at 600 V 6 mA overnight at 4°C until the Orange G had run
through 2/3 of the gel. Gels were stained with 0.1% Toluidine blue.
Transmission electron microscopy
For imaging whole epimastigote forms, cells were washed with filtered buffer A [116
mM NaCl, 5.4 mM KCl, 0.8 mM MgSO4, 50 mM Hepes (pH 7.2) and 5.5 mM glucose]
twice, and directly applied to Formvar-coated copper grids, allowed to adhere for 10 min,
carefully blotted dry, and observed in JEM-1210 electron microscope operating at 80 kV.
Whole unfixed epimastigotes of wild type and TcRab32DN were randomly selected and
the number of acidocalcisome per cell was counted in 50 cells from 2 different
preparations.

120
Cell growth measurement
Measured optical density at 600 nm as a measure of concentration of epimastigotes in
suspensión in the Gilford spectrophotometer with a starting culture of 4.5 x 106
epimastigotes and monitored cell density for the next 7 days.

121
REFERENCES
1. Clark TB (1959) Comparative morphology of four genera of trypanosomatidae. J
Protozool 6: 227-232.
2. Figarella K, Uzcategui NL, Zhou Y, LeFurgey A, Ouellette M, et al. (2007)
Biochemical characterization of Leishmania major aquaglyceroporin LmAQP1:
possible role in volume regulation and osmotaxis. Molecular microbiology 65:
1006-1017.
3. Linder JC, Staehelin LA (1979) A novel model for fluid secretion by the
trypanosomatid contractile vacuole apparatus. The Journal of cell biology 83:
371-382.
4. Baqui MM, De Moraes N, Milder RV, Pudles J (2000) A giant phosphoprotein
localized at the spongiome region of Crithidia luciliae thermophila. The Journal of
eukaryotic microbiology 47: 532-537.
5. Rohloff P, Montalvetti A, Docampo R (2004) Acidocalcisomes and the contractile
vacuole complex are involved in osmoregulation in Trypanosoma cruzi. The
Journal of biological chemistry 279: 52270-52281.
6. Li ZH, Alvarez VE, De Gaudenzi JG, Sant'Anna C, Frasch AC, et al. (2011)
Hyperosmotic stress induces aquaporin-dependent cell shrinkage, polyphosphate
synthesis, amino acid accumulation, and global gene expression changes in
Trypanosoma cruzi. The Journal of biological chemistry 286: 43959-43971.
7. Niyogi S, Mucci J, Campetella O, Docampo R (2014) Rab11 Regulates Trafficking of
Trans-sialidase to the Plasma Membrane through the Contractile Vacuole
Complex of Trypanosoma cruzi. PLoS Pathog 10: e1004224.
8. Hasne MP, Coppens I, Soysa R, Ullman B (2010) A high-affinity putrescine-
cadaverine transporter from Trypanosoma cruzi. Molecular microbiology 76: 78-
91.

122
9. Sesaki H, Wong EF, Siu CH (1997) The cell adhesion molecule DdCAD-1 in
Dictyostelium is targeted to the cell surface by a nonclassical transport pathway
involving contractile vacuoles. The Journal of cell biology 138: 939-951.
10. Sriskanthadevan S, Lee T, Lin Z, Yang D, Siu CH (2009) Cell adhesion molecule
DdCAD-1 is imported into contractile vacuoles by membrane invagination in a
Ca2+- and conformation-dependent manner. The Journal of biological chemistry
284: 36377-36386.
11. Heuser J, Zhu Q, Clarke M (1993) Proton pumps populate the contractile vacuoles of
Dictyostelium amoebae. The Journal of cell biology 121: 1311-1327.
12. Moniakis J, Coukell MB, Janiec A (1999) Involvement of the Ca2+-ATPase PAT1
and the contractile vacuole in calcium regulation in Dictyostelium discoideum.
Journal of cell science 112 ( Pt 3): 405-414.
13. Jeffries TR, Morgan GW, Field MC (2001) A developmentally regulated rab11
homologue in Trypanosoma brucei is involved in recycling processes. Journal of
cell science 114: 2617-2626.
14. Ulrich PN, Jimenez V, Park M, Martins VP, Atwood J, 3rd, et al. (2011)
Identification of contractile vacuole proteins in Trypanosoma cruzi. PloS one 6:
e18013.
15. Harris E, Yoshida K, Cardelli J, Bush J (2001) Rab11-like GTPase associates with
and regulates the structure and function of the contractile vacuole system in
dictyostelium. Journal of cell science 114: 3035-3045.
16. Docampo R, Jimenez V, Lander N, Li ZH, Niyogi S (2013) New insights into roles of
acidocalcisomes and contractile vacuole complex in osmoregulation in protists.
International review of cell and molecular biology 305: 69-113.
17. Zerial M, McBride H (2001) Rab proteins as membrane organizers. Nat Rev Mol Cell
Biol 2: 107-117.
18. Pfeffer SR (2001) Rab GTPases: specifying and deciphering organelle identity and
function. Trends Cell Biol 11: 487-491.
19. Munro S (2002) Organelle identity and the targeting of peripheral membrane proteins.
Curr Opin Cell Biol 14: 506-514.

123
20. Seabra MC, Wasmeier C (2004) Controlling the location and activation of Rab
GTPases. Curr Opin Cell Biol 16: 451-457.
21. Bultema JJ, Ambrosio AL, Burek CL, Di Pietro SM (2012) BLOC-2, AP-3, and AP-1
proteins function in concert with Rab38 and Rab32 proteins to mediate protein
trafficking to lysosome-related organelles. J Biol Chem 287: 19550-19563.
22. Raposo G, Marks MS (2007) Melanosomes--dark organelles enlighten endosomal
membrane transport. Nat Rev Mol Cell Biol 8: 786-797.
23. Delevoye C, Hurbain I, Tenza D, Sibarita JB, Uzan-Gafsou S, et al. (2009) AP-1 and
KIF13A coordinate endosomal sorting and positioning during melanosome
biogenesis. The Journal of cell biology 187: 247-264.
24. Docampo R, Scott DA, Vercesi AE, Moreno SN (1995) Intracellular Ca2+ storage in
acidocalcisomes of Trypanosoma cruzi. The Biochemical journal 310 ( Pt 3):
1005-1012.
25. Docampo R, de Souza W, Miranda K, Rohloff P, Moreno SN (2005)
Acidocalcisomes - conserved from bacteria to man. Nature reviews Microbiology
3: 251-261.
26. Docampo R, Moreno SN (2011) Acidocalcisomes. Cell calcium 50: 113-119.
27. Ruiz FA, Lea CR, Oldfield E, Docampo R (2004) Human platelet dense granules
contain polyphosphate and are similar to acidocalcisomes of bacteria and
unicellular eukaryotes. The Journal of biological chemistry 279: 44250-44257.
28. Smith SA, Mutch NJ, Baskar D, Rohloff P, Docampo R, et al. (2006) Polyphosphate
modulates blood coagulation and fibrinolysis. Proceedings of the National
Academy of Sciences of the United States of America 103: 903-908.
29. Moreno-Sanchez D, Hernandez-Ruiz L, Ruiz FA, Docampo R (2012) Polyphosphate
is a novel pro-inflammatory regulator of mast cells and is located in
acidocalcisomes. The Journal of biological chemistry 287: 28435-28444.
30. Theos AC, Tenza D, Martina JA, Hurbain I, Peden AA, et al. (2005) Functions of
adaptor protein (AP)-3 and AP-1 in tyrosinase sorting from endosomes to
melanosomes. Mol Biol Cell 16: 5356-5372.

124
31. Besteiro S, Tonn D, Tetley L, Coombs GH, Mottram JC (2008) The AP3 adaptor is
involved in the transport of membrane proteins to acidocalcisomes of Leishmania.
Journal of cell science 121: 561-570.
32. Huang G, Fang J, Sant'Anna C, Li ZH, Wellems DL, et al. (2011) Adaptor protein-3
(AP-3) complex mediates the biogenesis of acidocalcisomes and is essential for
growth and virulence of Trypanosoma brucei. The Journal of biological chemistry
286: 36619-36630.
33. Montalvetti A, Rohloff P, Docampo R (2004) A functional aquaporin co-localizes
with the vacuolar proton pyrophosphatase to acidocalcisomes and the contractile
vacuole complex of Trypanosoma cruzi. The Journal of biological chemistry 279:
38673-38682.
34. Marchesini N, Ruiz FA, Vieira M, Docampo R (2002) Acidocalcisomes are
functionally linked to the contractile vacuole of Dictyostelium discoideum. The
Journal of biological chemistry 277: 8146-8153.
35. Jean S, Kiger AA (2012) Coordination between RAB GTPase and phosphoinositide
regulation and functions. Nat Rev Mol Cell Biol 13: 463-470.
36. Yokoyama K, Gillespie JR, Van Voorhis WC, Buckner FS, Gelb MH (2008) Protein
geranylgeranyltransferase-I of Trypanosoma cruzi. Mol Biochem Parasitol 157:
32-43.
37. Alto NM, Soderling J, Scott JD (2002) Rab32 is an A-kinase anchoring protein and
participates in mitochondrial dynamics. J Cell Biol 158: 659-668.
38. Tamura K, Ohbayashi N, Maruta Y, Kanno E, Itoh T, et al. (2009) Varp is a novel
Rab32/38-binding protein that regulates Tyrp1 trafficking in melanocytes. Mol
Biol Cell 20: 2900-2908.
39. Park M, Serpinskaya AS, Papalopulu N, Gelfand VI (2007) Rab32 regulates
melanosome transport in Xenopus melanophores by protein kinase a recruitment.
Current biology : CB 17: 2030-2034.
40. Rodrigues CO, Scott DA, Docampo R (1999) Characterization of a vacuolar
pyrophosphatase in Trypanosoma brucei and its localization to acidocalcisomes.
Mol Cell Biol 19: 7712-7723.

125
41. Lander N, Ulrich PN, Docampo R (2013) Trypanosoma brucei vacuolar transporter
chaperone 4 (TbVtc4) is an acidocalcisome polyphosphate kinase required for in
vivo infection. The Journal of biological chemistry 288: 34205-34216.
42. Ulrich PN, Lander N, Kurup SP, Reiss L, Brewer J, et al. (2014) The acidocalcisome
vacuolar transporter chaperone 4 catalyzes the synthesis of polyphosphate in
insect-stages of Trypanosoma brucei and T. cruzi. The Journal of eukaryotic
microbiology 61: 155-165.
43. Urbina JA, Moreno B, Vierkotter S, Oldfield E, Payares G, et al. (1999)
Trypanosoma cruzi contains major pyrophosphate stores, and its growth in vitro
and in vivo is blocked by pyrophosphate analogs. The Journal of biological
chemistry 274: 33609-33615.
44. Mendoza M, Mijares A, Rojas H, Rodriguez JP, Urbina JA, et al. (2002)
Physiological and morphological evidences for the presence acidocalcisomes in
Trypanosoma evansi: single cell fluorescence and 31P NMR studies. Mol
Biochem Parasitol 125: 23-33.
45. Hirota Y, Tanaka Y (2009) A small GTPase, human Rab32, is required for the
formation of autophagic vacuoles under basal conditions. Cell Mol Life Sci 66:
2913-2932.
46. Berriman M, Ghedin E, Hertz-Fowler C, Blandin G, Renauld H, et al. (2005) The
genome of the African trypanosome Trypanosoma brucei. Science 309: 416-422.
47. Tamura K, Ohbayashi N, Ishibashi K, Fukuda M (2011) Structure-function analysis
of VPS9-ankyrin-repeat protein (Varp) in the trafficking of tyrosinase-related
protein 1 in melanocytes. J Biol Chem 286: 7507-7521.
48. Bultema JJ, Di Pietro SM (2013) Cell type-specific Rab32 and Rab38 cooperate with
the ubiquitous lysosome biogenesis machinery to synthesize specialized
lysosome-related organelles. Small GTPases 4: 16-21.
49. Martinez-Arca S, Rudge R, Vacca M, Raposo G, Camonis J, et al. (2003) A dual
mechanism controlling the localization and function of exocytic v-SNAREs. Proc
Natl Acad Sci U S A 100: 9011-9016.

126
50. Schmatz DM, Murray PK (1982) Cultivation of Trypanosoma cruzi in irradiated
muscle cells: improved synchronization and enhanced trypomastigote production.
Parasitology 85 (Pt 1): 115-125.
51. Moreno SN, Silva J, Vercesi AE, Docampo R (1994) Cytosolic-free calcium
elevation in Trypanosoma cruzi is required for cell invasion. J Exp Med 180:
1535-1540.
52. Cuevas IC, Rohloff P, Sanchez DO, Docampo R (2005) Characterization of
farnesylated protein tyrosine phosphatase TcPRL-1 from Trypanosoma cruzi.
Eukaryot Cell 4: 1550-1561.
53. Nepomuceno-Silva JL, Yokoyama K, de Mello LD, Mendonca SM, Paixao JC, et al.
(2001) TcRho1, a farnesylated Rho family homologue from Trypanosoma cruzi:
cloning, trans-splicing, and prenylation studies. J Biol Chem 276: 29711-29718.
54. Ruiz FA, Rodrigues CO, Docampo R (2001) Rapid changes in polyphosphate content
within acidocalcisomes in response to cell growth, differentiation, and
environmental stress in Trypanosoma cruzi. J Biol Chem 276: 26114-26121.
55. Ault-Riche D, Fraley CD, Tzeng CM, Kornberg A (1998) Novel assay reveals
multiple pathways regulating stress-induced accumulations of inorganic
polyphosphate in Escherichia coli. J Bacteriol 180: 1841-1847.

127
FIGURES
Figure 4.1 TcRab32 localization in different life stages of T. cruzi. (A) Eluted and
desalted fractions (E1 to E9) obtained during recombinant TcRab32 purification from
E.coli as analyzed by SDS PAGE showing a band of correct size (42 KDa) corresponding
to the His-tagged protein. The 10% SDS PAGE gel was stained with Coommassie blue.

128
(B-D) TcRab32 was detected in the contractile vacuole bladder of epimastigotes (Epi),
trypomastigotes (Trypo), and intracellular amastigotes (Ama) with additional punctated
staining using specific antibodies against TcRab32 raised in mouse. (E) Western blot
analyses with TcRab32 antibody of lysates of wild-type trypomastigotes (T), amastigotes
(A) and epimastigotes (E), showing bands (arrows) corresponding to the endogenous
TcRab32 (26 kDa). The blots were sequentially probed with anti-tubulin antibodies, used
as loading control.

129
Figure 4.2 TcRab32 is digeranylated in vitro. (A) Comparison of the deduced amino
acid sequence of T. cruzi Rab32 with human Rab32. The presence of the “WDIAGQE”
and C terminal “CSC” domain is boxed in red and the presence of phenylalanine “F” at
position 194 is denoted in blue asterisk. (B)Radiolabelled proteins were analyzed by
SDS-PAGE on a 15% gel followed by autoradiography. Lane 1 in the presence of all
reactants; rTcRab32, epimastigote extract and (3H) GGPP, lanes 2 and 3 are negative

130
controls. Enzymatic assay performed for 30 min. A radioactive band of 42 KDa was
observed corresponding to the His-tagged protein.
Figure 4.3 Localization of GFP-TcRab32 mutants. GFP-TcRab32 prenylation-motif
mutants have a cytosolic localization. GFP-TcRab32DN mutants which mimic the GDP-
bound state of the protein have a punctated cytosolic localization. GFP-TcRab32DP
mutants that mimic the GTP-bound state of the proteins localize mainly to the membrane
of the CVC.

131
Figure 4.4 Lack of colocalization between GFP-TcRab32 and mitochondrial marker
and localization of mitochondrial marker is not affected in TcRab32 mutants. (A)
There is no colocalization between mitotracker (red) with GFP-TcRab32, as detected
with antibodies against GFP (green). Mitotracker (red) labels the mitochondria in GFP-
TcRab32 DP (B) and GFP-TcRab32DN (C) epimastigotes. Labeling of the GFP-
TcRab32DP and GFP-TcRab32DN was detected with anti-GFP (green).

132
Figure 4.5 Colocalization of GFP-TcRab32 and VP1 under osmotic stress. (A) There
is no colocalization between TbVP1 (red) with GFP-TcRab32 as detected with antibodies
against GFP (green) under isosmotic conditions. (B) Overlap between signals for TcVP1
(red) and GFP-TcRab32 (green) in the CVC occur under hyposmotic stress conditions.

133
Figure 4.6 Reduced short chain poly P and PPi levels in TcRab32DN epimastigotes
in comparison to wild type epimastigotes. Extracts from TcRab32DN epimastigotes
(DN) showed a 50% reduction in (A) PPi levels and an 80% reduction in (B) short chain
poly P levels with no significant changes in (C) long chain poly P levels in comparison to
GFP (C) and TcRab32 (OE) epimastigotes. TcRab32OE epimastigotes (OE) have levels

134
of PPi and short chain poly P slightly more than control epimastigotes. Values are means
± SD of three different experiments. *Differences are statistically significant as compared
to respective controls, p < 0.05 (Student’s t test). (D) Extracts of short chain PolyP
produced by OE, GFP and DN resolved by Urea PAGE and visualized by toluidine blue.
ORG represents migration of orange G dye. Levels of short chain poly P lower in lanes
labelled DN in comparison to lanes labelled OE and C.
Figure 4.7 Reduction in electron dense acidocalcisomes and considerable increase in
empty vacuole in TcRab32DN epimastigotes in comparison to wild type. (A)
Transmission electron microscopy (TEM) image from whole unstained and unfixed
TcRab32DN epimastigotes (DN) show the presence of numerous empty vacuoles with
complete loss of electron density of acidocalcisomes in comparison to wild type (WT)

135
epimastigotes. Scale bars, 2 µm (B) The number of acidocalcisomes per cell were
counted in 70 random cells from 2 independent experiments and the numeric distribution
of acidocalcisomes showed that majority of TcRab32DN epimastigotes had <10 or
between 11-20 electron-dense acidocalcisomes. (C and D) In order to quantitate the
phenotype of empty vacuoles we counted 50 random parasites in WT and DN after TEM
and found that there is a significant increase in parasites with empty vacuoles in DN w.r.t
WT. * indicates differences are statistically significant compared with respective
controls, p<0.05.
Figure 4.8 Traffic of trans-sialidase is not affected in TcRab32DN mutant
trypomastigotes. GFP-TcRab32 DN mutants show a punctated cytosolic localization as

136
detected with anti-GFP (green), while antibodies against TcTS still localize to the
surface.
Figure 4.9 Effect of TcRab32 mutations on the cell growth of epimastigotes and
their response to hyposmotic and hyperosmotic stress conditions. (A) Growth rate of
of epimastigotes expressing the dominant negative (DN) TcRab32 in comparison to GFP-
0
20000000
40000000
60000000
80000000
100000000
120000000
140000000
160000000
0 2 4 6 8 10
CE
LL
DE
NS
ITY
/ml
TIME (DAYS)
C
DN

137
expressing (C) epimastigotes. (B) Cells were pre-incubated in isosmotic buffer for 3 min
and then subjected to hyposmotic (final osmolarity = 150 mOsm) (C) or hyperosmotic
(final osmolarity = 650 mOsm) stress. Relative change in cell volume was followed by
monitoring absorbance at 550 nm by light scattering. As compared to wild-type cells
(WT), cells expressing GFPTcRab32DN (DN) failed to fully recover their volume after
hyposmotic stress and shrank less after hyperosmotic stress, while cells overexpressing
GFP-TcRab32 (OE) recovered their volume faster after hyposmotic stress and shrank
more after hyperosmotic stress. The response of the GFPTcRab32DP (DP) was very
similar to the wild-type cells. Values are means ± SD of three different experiments.
Asterisks indicate statistically significant differences, p<0.05, (Bonferroni’s multiple
comparison ‘‘a posteriori’’ test of one-way ANOVA) at all time points after induction of
osmotic stress.
Figure 4.10 Reduced infectivity of TcRab32 mutant trypomastigotes. (A and B)
Effect of TcRab32 overexpression (OE) or mutation (DN) on in vitro invasion by

138
trypomastigotes on host cell in comparison with control; wild type trypomastigotes (WT)
and GFP-expressing trypomastigotes (GFP). Values are mean ±SD (n = 3). * indicate that
differences are statistically significant compared with respective controls, p<0.05
(Ordinary one way ANOVA with Bonferroni post-test). (C) Punctated cytosolic
localization of GFP-TcRab32DN trypomastigotes and amastigotes as detected using
antibodies against GFP. DNA was stained with DAPI. Bars = 10 µm.

139
CHAPTER 5
CONCLUSION
Summary of key findings
At the beginning of my thesis work the role of the contractile vacuole complex (CVC) in
osmoregulation was well established in T. cruzi. As T. cruzi is exposed to different host
environments, the need for osmoregulation is critical in these parasites. Work from this
laboratory has shown the role of CVC not only in regulating volume under conditions of
hyposmotic stress [1], but also in shrinking of the parasites, under hyperosmotic stress
[2]. I got interested in studying the role of two Rab proteins: Rab11 (published in [3] and
Rab32 which were identified in a proteomic study of the CVC [4]. This study revealed
the presence of a cohort of proteins which usually have function in regulation of
intracellular traffic, vesicle fusion and protein secretion. Only a few Rab proteins:
TcRab7, TcRab5, and TcRab4 had been studied in T. cruzi, and molecular events of
vesicle trafficking were still poorly understood [5] [6] [7]. Although the localization of
some of these Rabs has been demonstrated, there was no functional study conducted with
them. Our study provides the first experimental evidence of the mechanistic role of two
Rab GTPases in T. cruzi: TcRab11 and TcRab32.
Mutation of Rab proteins has been of major value in determining the physiological role of
Rabs in other cell systems. The approach is based upon the ability to make GTP- and
GDP-locked forms of the proteins, with dominant positive and negative effects,

140
respectively, over the endogenous Rab protein. We applied this same approach for
studying TcRab32 and TcRab11.
Results with the dominant negative TcRab11 (TcRab11DN) were clear: there was a
defect in osmoregulation in the Rab11 mutant parasites suggesting that volume regulation
requires Rab11-dependent processes, such as membrane fusion. There was also a defect
in traffic of GPI-anchored trans-sialidase to the surface and as a result, an inability to
invade host cells properly in comparison to wild type parasites. Proper trafficking of
surface proteins is important for evading host immune defenses and to support host
invasion. The observed phenotype of perturbation in traffic was not a side effect of
mutating a contractile vacuole complex resident protein as the morphology of the CVC
was not affected in the mutant parasites. The observed phenotype was specific for the
traffic of trans-sialidase because the membrane traffic of other proteins (both GPI- and
non GPI-anchored) was not affected at all. The traffic of trans-sialidase was also
unaffected in the Rab32 mutant trypomastigotes, further suggesting the uniqueness of
each of these Rab proteins. Our study provides support for a role of the CVC as a
trafficking hub in addition to its role in osmoregulation. TcRab11 is developmentally
regulated and our results show that it has specific roles in different life cycle stages of T.
cruzi, suggesting different requirements of the protein for survival in the insect and
mammalian host.
The hypothesis that the CVC is a trafficking hub and an equivalent to early/recycling
endosomes of other eukaryotes was additionally supported by the phenotypic changes
occurring after mutation of the CVC-located TcRab32. Acidocalcisomes were less
numerous and electron-dense, and deficient in PPi and polyP, suggesting a deficient

141
traffic of proteins involved in the biogenesis of these organelles. This phenotype
suggested the traffic of proteins from the CVC to the acidocalcisomes. This ultimately
resulted in a reduced ability of these parasites to invade host cells.
The purpose of my thesis was two-fold: to investigate the function of the CVC as a
trafficking hub and to provide a detailed analysis of the roles of TcRab11 and TcRab32 in
this process. The CVC is a fascinating complex product of evolution. Much remains to be
studied about how this organelle evolved. It is clear that this organelle is central to
parasite growth, development and pathogenesis.
Future work
Does the Contractile Vacuole Complex (CVC) in T. cruzi have a role in calcium
homeostasis?
A role of the CVC in calcium homeostasis has been proposed in the amoeba D.
discoideum and in Paramecium tetraurelia on the basis of the presence in these
organelles of different calcium transporters, such as a Ca2+
-ATPase [8,9] and an inositol
1,4,5-trisphosphate receptor (IP3R) [10]. Ca2+
-ATPase PAT1, usually present in the
membrane of the CVC moves to the plasma membrane when cells are incubated at high
Ca2+
concentrations. The contractile vacuole membranes in Dictyostelium are also
extremely rich in calmodulin [11]. The identification of peptides corresponding to a
calcium channel (Tc00.1047053504105.130) and an IP3/ryanodine receptor
(Tc00.1047053509461.90) in subcellular fractions of T. cruzi enriched in the CVC
implies that the CVC could have a possible role in Ca2+
signaling. Besides, fusion events
may require a local calcium signal, as Ca2+
controls priming steps and prepares vesicles
for fusion [12].

142
The role of the CVC as a trafficking hub remains to be fully characterized
The presence of SNAREs, such as SNARE2.1 (TcCLB.507625.183), SNARE2.2
(TcCLB.53506715.50), and VAMP1 (TcCLB.53511627.60) in the proteomic data of T.
cruzi CVC and their localization to the CVC, suggests the existence of several membrane
to membrane interactions that facilitate vesicle docking for subsequent fusion. The
vesicle associated membrane proteins (VAMPs) belong to the R-SNAREs group.
Paramecium tetraurelia RSNARE PtSyb2-2 has been shown to localize to the entire
contractile vacuole complex [13], and its orthologue in T. cruzi (VAMP1,
TcCLB.53511627.60) was detected in the CV bladder of epimastigotes submitted to
hyposmotic stress. This could be indicative of a possible role of these contractile vacuole
complex-localized SNAREs in fusion/fision events between the bladder and the
spongiome or between the CVC and acidocalcisomes during swelling or collapse of the
CVC. In this regard, a study in Dictyostelium found that there is an interaction between
adaptor protein AP180 (present in clathrin-coated vesicles on contractile vacuole
bladders) and the contractile vacuole-localized SNARE, Vamp7, especially during fusion
events of the CVC [14]. Orthologues of both AP180 (TcCLB.53503449.30) and VAMP7
(TcCLB.53511627.60) are present in the CVC of T. cruzi. All this reflects that a
multitude of vesicle fusion and membrane interaction events probably exists in the CVC
of T. cruzi.
To identify the interaction partners of TcRab32 and the study of their cell-type
specific regulation
Presently we are immunoprecipitating proteins from the GFP-TcRab32-expressing cell
line and doing mass spectrometric analysis to identify potential interaction partners of

143
TcRab32. Candidates of interest are VAMP7 and AP-3, which were identified in other
eukaryotes as interaction partners for Rab32 [15]. VAMP7, which is a vesicle SNARE
protein, is highly conserved across eukaryotes and the identification of an interaction
between CVC-resident TcRab32 and VAMP7 in T. cruzi would further suggest a link
between higher and lower eukaryotes. The delta subunit of the AP-3 complex, whose role
in acidocalcisome biogenesis has been studied by our lab [16], is found to interact with
VAMP7 and direct its traffic along the endocytic pathway in mammalian cells. A
possible interaction between TcRab32, VAMP7 and AP-3 might serve as a pre-requisite
for the correct traffic of cargo to acidocalcisomes.
Precise regulation of Rab-GTPases requires activity and binding with Rab GAP (GTPase
Activating Protein), Rab GEF (guanine nucleotide exchange factor) and Rab GDI
(guanosine nucleotide dissociation inhibitor). Rab GEFs or Rab GAPs are activated at the
right place and time. It is probable that the CVC-resident TcRab32 gets recruited at
domains of endosomes. The failure to detect TcRab32 in the acidocalcisome membrane
could be attributed to sensitivity issues of the immunofluorescence technique used in our
experiments; or it can be that TcRab32 remain bound to vesicles (corresponding to the
punctate staining detected) but they do not fuse with the acidocalcisomes. Understanding
how the sequential activation of Rab GTPases is achieved during vesicle trafficking is a
central question in the study of the cell biology of these parasites.
Possible changes in the acidocalcisome proteome in the Rab32 mutant parasites?
Experiments can be conducted to demonstrate the direct effect of TcRab32 mutation in
these parasites and its effect (direct or indirect) on the membrane composition of the
acidocalcisome. As the TcRab32DN parasites suffer a loss in the level of short chain

144
polyP (~80% reduction), without any change in long chain polyP it seems that the
TcVTC complex (Vacuolar Transporter Chaperone Complex), which is mainly involved
in the synthesis of short chain polyP [17,18] is affected. The use of antibodies against
components of the VTC complex will be appropriate to investigate if there is a loss or
reduction in any of the components. An alternative will be to do proteomic analysis of
acidocalcisomes of TcRab32 mutant parasites to study whether there is an altered content
of membrane proteins. A detailed analysis has to be made while interpreting the data with
appropriate controls to minimize false positive data and to subtract background. These
data are often biased against proteins that are expressed in low abundance.
To investigate how cargo destined to the acidocalcisome is sorted out from lysosomal
cargo
The acidocalcisome is a lysosome-related organelle (LRO) whose biogenesis is
apparently dependent on protein sorting from the Golgi [19]. How cargo destined for the
acidocalcisome is sorted from the lysosomal cargo at the trans-Golgi should be an
interesting area to explore further. Most of the LROs coexist with conventional
lysosomes as distinct organelles in the same cells [20]. What is the signal that avoids their
undesirable fusion with lysosomes? Lysosomal hydrolases and membrane proteins follow
the same route that comprises of the ER, Golgi and trans-Golgi network and endosomes
to the lysosome (reviewed in [21]. The identification of specific receptors or individual
adaptor proteins or lipid components, or dissection of the molecular machinery on the
membrane of the acidocalcisome to look for particular traffic mediators would be
relevant. To ensure that the TcRab32 mediates sorting to the acidocalcisome and has no
effect on the lysosomal traffic we can investigate whether the localization of the

145
membrane glycoprotein p67 (a marker of the lysosome) [22] is unaltered in the Rab32
mutant parasites. Preliminary work from our lab has shown, with Trypanosoma brucei
AP-3, that the lysosomal traffic route is separated from that of the LRO (acidocalcisome
in this case) route [19].
CVC as a potential drug target?
Despite T. cruzi trans-sialidase (TcTS) being known for several years our understanding
of its intracellular trafficking is still limited. TS has been identified as a potential target
for drug discovery and design. Besides having key role in host cell invasion,
pathogenesis, and host immune system evasion, trans-sialidase is not present in the
mammalian host, thus making it a potential drug candidate. The identification of the
specific role of TcTS in infection has been difficult to demonstrate in the past because of
the impossibility of doing knockouts of the considerable number of gene copies encoding
this protein scattered through the genome of this parasite. The mechanistic details now
known, through this work, regarding the traffic of TcTS in these parasites can be used in
rational drug design experiments aiming at effective treatments of Chagas disease.
As sorting of GPI-anchored surface proteins responsible for invasion occurs at the CVC,
disrupting the integrity of the CVC may act as a potential block to the surface traffic of
the antigens, hence providing a mechanism for control of T. cruzi invasion. A deeper
understanding of the intracellular traffic in T. cruzi will potentially open the door to new
rational therapeutics. Besides, it will be helpful to dissect the information regarding key
biological processes of these parasites and its effect on pathogenesis.

146
Hopefully this will just be the start in understanding the multi-faceted role of the
contractile vacuole complex and unravelling its potential as a drug target, thus opening
doors to new therapeutics.

147
REFERENCES
1. Rohloff P, Docampo R (2008) A contractile vacuole complex is involved in
osmoregulation in Trypanosoma cruzi. Exp Parasitol 118: 17-24.
2. Li ZH, Alvarez VE, De Gaudenzi JG, Sant'Anna C, Frasch AC, et al. (2011)
Hyperosmotic stress induces aquaporin-dependent cell shrinkage, polyphosphate
synthesis, amino acid accumulation, and global gene expression changes in
Trypanosoma cruzi. J Biol Chem 286: 43959-43971.
3. Niyogi S, Mucci J, Campetella O, Docampo R (2014) Rab11 Regulates Trafficking of
Trans-sialidase to the Plasma Membrane through the Contractile Vacuole
Complex of Trypanosoma cruzi. PLoS Pathog 10: e1004224.
4. Ulrich PN, Jimenez V, Park M, Martins VP, Atwood J, 3rd, et al. (2011) Identification
of contractile vacuole proteins in Trypanosoma cruzi. PLoS One 6: e18013.
5. Araripe JR, Cunha e Silva NL, Leal ST, de Souza W, Rondinelli E (2004)
Trypanosoma cruzi: TcRAB7 protein is localized at the Golgi apparatus in
epimastigotes. Biochem Biophys Res Commun 321: 397-402.
6. Araripe JR, Ramos FP, Cunha e Silva NL, Urmenyi TP, Silva R, et al. (2005)
Characterization of a RAB5 homologue in Trypanosoma cruzi. Biochem Biophys
Res Commun 329: 638-645.
7. Ramos FP, Araripe JR, Urmenyi TP, Silva R, Cunha e Silva NL, et al. (2005)
Characterization of RAB-like4, the first identified RAB-like protein from
Trypanosoma cruzi with GTPase activity. Biochem Biophys Res Commun 333:
808-817.
8. Plattner H (2013) Contractile vacuole complex--its expanding protein inventory. Int
Rev Cell Mol Biol 306: 371-416.
9. Bohme R, Bumann J, Aeckerle S, Malchow D (1987) A high-affinity plasma
membrane Ca2+-ATPase in Dictyostelium discoideum: its relation to cAMP-
induced Ca2+ fluxes. Biochim Biophys Acta 904: 125-130.

148
10. Ladenburger EM, Korn I, Kasielke N, Wassmer T, Plattner H (2006) An Ins(1,4,5)P3
receptor in Paramecium is associated with the osmoregulatory system. J Cell Sci
119: 3705-3717.
11. Heuser J, Zhu Q, Clarke M (1993) Proton pumps populate the contractile vacuoles of
Dictyostelium amoebae. J Cell Biol 121: 1311-1327.
12. Neher E (2012) Introduction: regulated exocytosis. Cell Calcium 52: 196-198.
13. Schilde C, Wassmer T, Mansfeld J, Plattner H, Kissmehl R (2006) A multigene
family encoding R-SNAREs in the ciliate Paramecium tetraurelia. Traffic 7: 440-
455.
14. Wen Y, Stavrou I, Bersuker K, Brady RJ, De Lozanne A, et al. (2009) AP180-
mediated trafficking of Vamp7B limits homotypic fusion of Dictyostelium
contractile vacuoles. Mol Biol Cell 20: 4278-4288.
15. Schafer IB, Hesketh GG, Bright NA, Gray SR, Pryor PR, et al. (2012) The binding of
Varp to VAMP7 traps VAMP7 in a closed, fusogenically inactive conformation.
Nat Struct Mol Biol 19: 1300-1309.
16. Huang G, Bartlett, P.D., Thomas, A.P., Moreno, S.N.J., Docampo, R. (2013)
Acidocalcisomes of Trypanosoma brucei have an inositol 1,4,5-trisphosphate
receptor that is required for growth and infectivity. Proc Natl Acad Sci USA in
press.
17. Lander N, Ulrich PN, Docampo R (2013) Trypanosoma brucei vacuolar transporter
chaperone 4 (TbVtc4) is an acidocalcisome polyphosphate kinase required for in
vivo infection. The Journal of biological chemistry 288: 34205-34216.
18. Ulrich PN, Lander N, Kurup SP, Reiss L, Brewer J, et al. (2014) The acidocalcisome
vacuolar transporter chaperone 4 catalyzes the synthesis of polyphosphate in
insect-stages of Trypanosoma brucei and T. cruzi. The Journal of eukaryotic
microbiology 61: 155-165.
19. Huang G, Fang J, Sant'Anna C, Li ZH, Wellems DL, et al. (2011) Adaptor protein-3
(AP-3) complex mediates the biogenesis of acidocalcisomes and is essential for
growth and virulence of Trypanosoma brucei. J Biol Chem 286: 36619-36630.
20. Marks MS, Seabra MC (2001) The melanosome: membrane dynamics in black and
white. Nat Rev Mol Cell Biol 2: 738-748.

149
21. Braulke T, Bonifacino JS (2009) Sorting of lysosomal proteins. Biochim Biophys
Acta 1793: 605-614.
22. Tazeh NN, Silverman JS, Schwartz KJ, Sevova ES, Sutterwala SS, et al. (2009) Role
of AP-1 in developmentally regulated lysosomal trafficking in Trypanosoma
brucei. Eukaryot Cell 8: 1352-1361.






![Glycosylphosphatidylinositol (GPI) Modi cation · Glycosylphosphatidylinositol (GPI) Modification Serves as a Primary Plasmodesmal Sorting Signal1[OPEN] Raul Zavaliev*, Xinnian Dong,](https://static.fdocuments.us/doc/165x107/5ff02a33b6b8f86a7036ad17/glycosylphosphatidylinositol-gpi-modi-glycosylphosphatidylinositol-gpi-modiication.jpg)