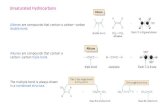
Reactions of Alkenes Section 10.3. Introduction Alkenes are unsaturated The double bond in ethene,...
-
Upload
bernard-goodman -
Category
Documents
-
view
221 -
download
0
description
Transcript of Reactions of Alkenes Section 10.3. Introduction Alkenes are unsaturated The double bond in ethene,...

Reactions of Alkenes
•Section 10.3

Introduction Alkenes are unsaturated The double bond in ethene,
for example, has one sigma bond and one pi bond (2nd bond)
• The pi bond is weaker than the sigma bond and can be broken without the molecule falling apart

Types of Reaction Addition reactions occur. There are 5 types:• 1. hydrogenation• 2. halogenation• 3. reaction with hydrogen halides• 4. hydration• 5. polymerization

Hydrogenation Addition of H across C=C bond to produce an alkane A nickel catalyst is used and a temp. of 180° C See examples for you on notes in class Hydrogenation is used to convert vegetable oil that
contains unsaturated hydrocarbon chains into more saturated compounds with higher melting points
This will make margarine be a solid at room temperature

Halogenation Halogens reacting with alkenes Occurs at room temperature Halogen loses color Halogens are added across the double bond so
they are on adjacent C atoms Use the examples on class notes

Reaction of Symmetrical Alkenes with Hydrogen Halides
React with alkenes to produce halogenoalkanes
Occurs rapidly in solution at room temperature See the examples from class notes

Hydration Alkene is converted to an alcohol Water does not react directly with the alkene Concentrated H2SO4 is used to form an addition
product An intermediate product as H+ and HSO4
- are added across the double bond
Cold water is added, hydrolysis takes place, and the sulfuric acid is re-formed
See the example from class

Industrial Process of Synthesizing Ethanol by Hydration of Ethene
Pass ethene and steam at high pressure over a catalyst of phosphoric acid at 300° C
Phosphoric acid is adsorbed on SiO2 pellets Equilibrium is achieved and there is a conversion to
ethanol of about 5% Unconverted ethene is recycled in the process until
all is reacted High degree of purity is achieved Ethanol is an important solvent in other processes

Test for Saturation Add the compound to bromine water Alkane produces no reaction (saturated) Alkene reacts and the bromine water discolors
(unsaturated)

Polymerization Alkenes can join together in addition reactions to
form long chains called polymers Beginning alkene is called a monomer Chemical nature of the monomer determines the
properties of the polymer Typically are thousands of molecules long Major product of the organic chemical industry Most common and useful plastics are polymers of
alkenes

Continued Remember the video from class (You can
access MIT open course ware, solid state chemistry lectures with Donald Sadoway, lectures 27, 28 and 29 deal with organic)
Poly(chloroethene) is PVC Poly (tetrafluoroethene) is PTFE, known as
Teflon See the examples from class notes.



Source: Oil and Gas Journalhttp://www.ogj.com/articles/print/vol-110/issue-07/special-report-ethylene-report/global-ethylene-capacity.html

Ethene Products Polystyrene
PVC
polyethene