Properties of Water
-
Upload
aimee-terrell -
Category
Documents
-
view
21 -
download
1
description
Transcript of Properties of Water
Why does ice float?A.Because it evaporates at
100°CB.Because it forms pockets of
empty space when frozenC.Because it is a universal
solventD.Because it melts at 0°C
Why does ice float?A.Because it evaporates at
100°CB.Because it forms pockets of
empty space when frozenC.Because it is a universal
solventD.Because it melts at 0°C
Which best describes the movement of water molecules in a glass of liquid water?A. The molecules all move at the
same speedB. The molecules all move at
different speedsC. The molecules are not movingD. The molecules only move when
you move the glass of water.
Which best describes the movement of water molecules in a glass of liquid water?A. The molecules all move at the
same speedB. The molecules all move at
different speedsC. The molecules are not movingD. The molecules only move when
you move the glass of water.
Think pair shareThink pair share
Do you know what the term polar means? How do you think polar relates to
molecules?
Do you know what the term polar means? How do you think polar relates to
molecules?
What is water? What is water? Think-Pair-Share with your table mates
to answer the questions below
What is the chemical formula for water?
What does the formula tell us about water?
What kind of chemical bond do you think water has?
Think-Pair-Share with your table mates to answer the questions below
What is the chemical formula for water?
What does the formula tell us about water?
What kind of chemical bond do you think water has?
Video notesVideo notes
Electrolysis breaks water apart into
2 parts hydrogen and 1 part oxygen
Electrolysis breaks water apart into
2 parts hydrogen and 1 part oxygen
Water’s covalent bondsWater’s covalent bonds Sometimes in covalent bonds there is
an unequal sharing of electrons in a molecule.
When this happens, atoms pull more strongly on the shared electrons than other atoms do.
As a result the electrons move closer
to one atom, causing atoms to have slight electrical charges.
One side of the molecule is slightly negative and the other is slightly positive
Sometimes in covalent bonds there is an unequal sharing of electrons in a molecule.
When this happens, atoms pull more strongly on the shared electrons than other atoms do.
As a result the electrons move closer
to one atom, causing atoms to have slight electrical charges.
One side of the molecule is slightly negative and the other is slightly positive
Polar MoleculesPolar Molecules- Molecules with unequal electron sharing
are called polar molecules
- Polar molecules have a strong attraction to one another because of their slightly charged sides. - (just like opposite ends of a magnet)
- Molecules with unequal electron sharing are called polar molecules
- Polar molecules have a strong attraction to one another because of their slightly charged sides. - (just like opposite ends of a magnet)
Are all covalent bonds polar?
Are all covalent bonds polar?
Not all covalently bonded elements are polar, only the ones with unequal sharing.
A molecule can be nonpolar if… it contains positive and negative charges
that cancel each other out
Not all covalently bonded elements are polar, only the ones with unequal sharing.
A molecule can be nonpolar if… it contains positive and negative charges
that cancel each other out
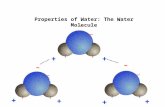
A polar moleculeA polar moleculeWater is a polar molecule and looks
like this…Water is a polar molecule and looks
like this…
Electrons are pulled more towards the nucleus of the oxygen
Video notesVideo notes
The attraction atoms have for electrons varies.
Polar molecules have positive and negative poles like a battery or a magnet.
The attraction atoms have for electrons varies.
Polar molecules have positive and negative poles like a battery or a magnet.
Why does it matter that water is polar?
Why does it matter that water is polar?
Water’s charged sides attract other elements and compounds with their charges. For example: a negative ion like chlorine would be
attracted to the positive end of a water molecule (the H or hydrogen side)
Water’s charged sides attract other elements and compounds with their charges. For example: a negative ion like chlorine would be
attracted to the positive end of a water molecule (the H or hydrogen side)
Water the Universal solvent
Water the Universal solvent
The polar charge that develops across the water molecule helps make it an excellent solvent. Solvent- a liquid that allows solids to break
apart to become a well mixed solution.
This is why water is called the Universal Solvent. Water can dissolve more substances than
any other liquid
The polar charge that develops across the water molecule helps make it an excellent solvent. Solvent- a liquid that allows solids to break
apart to become a well mixed solution.
This is why water is called the Universal Solvent. Water can dissolve more substances than
any other liquid
Salt (NaCl) dissolved in water
Salt (NaCl) dissolved in water
Salt contains sodium ions Na+ and chlorine ions Cl-
Sodium is attracted to oxygen end of H2OChlorine is attracted to hydrogen end of H2O
this can also be seen on pg 180 of your textbookCLICK HERE FOR SALT WATER ANIMATION
























![The International Association for the Properties of Water ... · The International Association for the Properties of Water ... for the Thermodynamic Properties of Water and ... [2].](https://static.fdocuments.us/doc/165x107/5b8f203709d3f2103e8bebc7/the-international-association-for-the-properties-of-water-the-international.jpg)







