Orbital Geometry
-
Upload
walt-sautter -
Category
Education
-
view
7.022 -
download
2
description
Transcript of Orbital Geometry

Copyright Sautter 2003

The next slide is a quick promo for my books after which the presentation will begin
Thanks for your patience!Walt S.
[email protected] stuff at: www.wsautter.com

Books available at:www.wsautter.com
www.smashwords.comwww.amazon.com
www.bibliotastic.comwww.goodreads.com
Walt’s Books for Free!


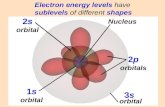
SPACITAL ORIENTATION OF ORBITALS ISDESIGNATED BY SUBSCRIPTS FOR THE
p, d ORBITALS AND f ORBITALS. S ORBITALSREQUIRE NO DESIGNATION SINCE THERE
IS ONLY ONE POSSIBLE S ORBITAL.
p ORBITALS OCCUR IN THREES, d ORBITALS IN FIVES AND f ORBITALS IN SEVENS.
THEREFORE IN THE CASES OF p, d AND fORBITALS EACH ONE REQUIRES SPECIFIC
IDENTIFICATION IN ORDER TO DIFFERENTIATE ONE ORBITAL OF THE SAME TYPE FROM
ANOTHER.

S ORBITALSHAPE
1S

P ORBITALSHAPES
PX
PY
PZ

D ORBITALSHAPES
dz2
dyz dxz
dxz dx2-y2


TYPES OF HYBRID ORBITALS & BONDS
S ORBITAL OVERLAPSP BONDINGSP2 BONDINGSP3 BONDING
LONE PAIR ELECTRONSMULTIPLE BONDS (DOUBLE & TRIPLE)
SIGMA BONDSPI BONDS

WHEN TWO HYDROGEN ATOM COMBINE TO FORMH2 THE 1S ATOMIC ORBITALS OVERLAP EACHOTHER.THIS OVERLAP OF ATOMIC ORBITALS GIVES A NEW
COMMON ORBITAL SPACE WHERE A PAIR OF BONDINGELECTRONS CAN RESIDE. THE GREATER THE OVERLAP
THE STRONGER THE BOND BETWEEN THE ATOMSAND THE ATOMS MOVE CLOSER TOGETHER. A SHORTER BOND LENGTH AND GREATER BOND ENERGY RESULTS.

1S ORBITALOVERLAP
IN H2
1S1S
H H

SP3 HYBRIDORBITALS IN
CH4

SP3 HYBRIDORBITALS IN
H2O
LONE ELECTRONS
PAIRS

END TO END OVERLAP OF HYBRID ORBITALS YIELDS SIGMA BONDS.
( = SIGMA BONDING)

SP3 HYBRIDORBITALS IN
C2H6
SIGMABOND

SIDEWAYS OVERLAP OF HYBRID ORBITALS IN THE FORMATION OF
DOUBLE AND TRIPLE BONDS YIELDS PI BONDS. THE PRIMARY BOND IN A
MULTIPLE BOND IS A SIGMA BOND AND THE SECONDARY BONDING IS PI BONDING. ( = PI BONDING)

SP2 HYBRIDORBITALS IN
C2H4
PI BOND
SIGMABOND

SP HYBRIDORBITALS IN
C2H2
PI BOND
SIGMABOND
PI BOND