Matter Classification Matter Flowchart Pure Substances Mixtures.
Mixtures solutions and pure substances
-
Upload
jose-pacheco -
Category
Documents
-
view
2.278 -
download
1
Transcript of Mixtures solutions and pure substances

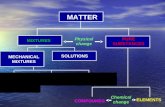
MIXTURES, SOLUTIONS AND PURE SUBSTANCES

Matter and its appearance
According to its appearance, there are two kinds of material systems:
Homogeneus systems: they have the same properties and compositions in all different parts of them.
Heterogeneus systems: different parts with different properties can be found in them.

Homogeneus Systems

Heterogeneus Systems

Heterogeneus Mixtures Systems with several different
substances that can be distinguished:
Oil and water. Granite is a rock that has three
different substances: quartz, mica and felspar.
Blood: using a microscope we can see a liquid (plasma) and different kinds of cells.
Smoke: has solid particles in a gas (air).

Heterogeneous Mixtures
How to separe their components:
– If the components are a solid and a liquid, we can separe them by filtering.
• E. g.: water and sand

Heterogeneous Mixtures
How to separe their components:
– Decantation: This method uses the different densities of the components to separe them:
• (E.g.: oil and water)

Homogeneous Systems
There are two kinds of homogeneous systems:
Pure substances:
• They have an only component• They have constant
composition and properties. Solutions:
• They have several components.• Their composition can be
changed

Solutions A solution has, at least, two components:
Solute: It is the component that is present in a lower
quantity in the solution. Solvent:
It is the component that is present in excess. Its state never changes when the solution is being
formed
A simple sample: salty water: There is much more water than salt Water is a liquid and the solution too.
SOLUTE = SALT and SOLVENT = WATER

SolutionsThey are not always liquids

Solutions
There three kinds of solutions: Solvent + a small amount of solute =
diluid solution. Solvent + a large amount of solute =
concentrate solution. The quantity of solute that can be
dissolved has a limit (solubility). In this case we have a satured solution.
If we try to dissolve more solute in a satured solution, the extra quantity of solute will fall onto the botton of the container. (precipitation)

Concentration of a solution:
– Expresses, in a numeric way, the quantity of soluto un a specific quantity of solution.
Solutions
concentration=amountofsolutoamountofsolution
There are many ways of express it, depending of the unit we use. We are going to study three of them.

Concentration: grams per liter of solution.
Solutions
concentration ( g /L )=massofsoluto (g )Volumenofsolution
Sample: we put 20 grams of sugar in a 500-mililiters flask and fill it in with water.
In the flask, there are:
20 g of soluto (sugar)Soluto and solvent with a total volume of 500 mL = 0,5 L
concentration (g/L) = 20 g / 0,5 L = 40 g/L

Concentration: percentage in mass
Solutions
concentration (mass )= massofsolutomassofsolution
·100
Sample: we add 25 grams of salt into a flask that contains 100 grams of water.
In the flask, there are:
25 g of soluto (salt)100 g + 25 g = 125 g of solution (salt + water)
% mass = (25 g / 125 g) · 100 = 20 % in mass

Concentration: percentage in volume
Solutions
concentration (volume )= volumeofsolutovolumeofsolution
·100
Sample: we mixure 20 mililiters of alcohol and 130 mililiters of water:
We have:
20 mL of alcohol (alcohol)130 mL + 20 mL = 150 mL of solution (alcohol + water)
% mass = (20 mL / 150 mL) · 100 = 13.3 % in volume

Solutions
How to separate its components:
– Evaporation and crystallization: a liquid evaporates at room temperature, but a solid doesn't.

Solutions How to separate its components:
– Distillation: this method is useful to separate:• Liquids whose boiling points are different
enough
• Liquids and solids

Pure SubstancesPure Substances
Pure substances: They have a constant composition. They can't be separated in simpler
substances using physical methods. There are two different kinds of pure
substances: Chemical compounds: pure substances
that can be separated in others using chemical methods.
Elements: pure substances that can't be separated in others

CompoundCompound
Pure substance composed of two or more different elements joined by chemical bonds.
Made of elements in a specific ratio that is always the same
Has a chemical formula Can only be separated by chemical
means, not physically There are millions of different compounds
in nature

CompoundCompound
¿Compound or Mixture?

ElementElement It is the simplest kind
of matter. Has a chemical
symbol. Can't be
separated in others substances
There are only about one hundred elements in natures. All of them are in the Periodic Table

Four kinds of matterFour kinds of matter

Four kinds of matterFour kinds of matter

Four kinds of matterFour kinds of matter