Mixture and Seperations
-
Upload
chevy-toni-lewin -
Category
Documents
-
view
221 -
download
0
Transcript of Mixture and Seperations
-
7/29/2019 Mixture and Seperations
1/38
1
Identify the different types of solutions
Investigate experimentally the effect of structure and
temperature on solubility of solids in water Distinguish among solutions, suspensions and
colloids Identify suitable separation techniques basedon difference in properties of the components ofmixtures
Describe the extraction of sucrose from sugar cane
Mixtures and SeparationsChapter 8
Learning Outcomes
-
7/29/2019 Mixture and Seperations
2/38
Copper(II) sulphate
Pure substances
A pure substance is a single substance not mixed
with anything else.
Sugar and table salt are examples of pure
substances.
Pure substances usually exist in crystal form.
For example, table salt, sugar and copper(II)
sulphate all exist as crystals. A pure substance can be an element or a
compound.
Sugar crystals
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
3/38
Pure and impure substances
A pure substance has afixed melting point andboiling point.For example, pure waterboils at 100 oC, and
melts at 0 oC.b.p. 100 oC
m.p. 0 oC
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
4/38
Impure substances or mixtures
A mixture on the other hand, is an impure substance. It
does not have a fixed melting point and boiling point. Itmelts or boils over a range of temperatures.
For example, sea water boils at about 102 oC and freezes
at around 2.5oC.
In general, an impurity lowers the melting point and
increases the boiling point of a substance.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
5/38
Impure substances or mixtures
In nature, most substances are impure. They consist of two
or more substances mixed together. Such substances are called mixtures.
Examples of mixtures are air, sea water, petroleum and
granite rock.
Mixtures can be purified orseparated by physical methods.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
6/38
6
A Mixture
Air is made up of different gases: nitrogen, oxygen, carbon dioxide andnoble gases such as argon, neon, and helium.
The gases in air can be easily separated by liquefaction followed byfractional distillation.
Air is an example of a mixture.
A mixture is made up of two or more substances
physically combined together.
Mixtures and SeparationsChapter 8
-
7/29/2019 Mixture and Seperations
7/38
7
Composition of a mixture
A mixture can be made up oftwo or more
elements.
E.g. Brass is a mixture of copper and zinc.
A mixture can be made up oftwo
or more elements or compounds.
E.g. Air is a mixture which containsboth elements and compounds.
Copper
atom
Zincatom
Air contains both elements and
compounds.
Chapter 8Mixtures and Separations
Brass
Element
Compound
-
7/29/2019 Mixture and Seperations
8/38
Mixtures
Homogenous
Mixtures
Heterogeneous
Solution
Suspension Colloid
Emulsion
-
7/29/2019 Mixture and Seperations
9/38
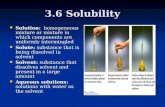
Homogeneous mixtures
Homogeneous mixture is uniform throughout no
difference. Characteristics of solutions:
When left to stand, the solute and solvent do not separate.
Solutes are not visible (particle size 1 nm).
All parts of the solution have the same chemical composition,chemical and physical properties.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
10/38
Homogeneous mixtures
Solubility of a solution refers to the amount of solute that isable to dissolve in a given quantity of solvent at a fixedtemperature and pressure.
The rate of dissolving of a solute depends on: Temperature (e.g. Solubility increases with temperature.)
Surface area of solute (e.g. Fine sugar dissolves faster than rock
sugar.) Rate of agitation (e.g. Stirring the solution increases the rate of
dissolving.)
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
11/38
Heterogeneous mixtures
Heterogeneous mixtures have non-uniform compositionswhich can be mechanically separated.
Examples of heterogeneous mixtures are colloids andsuspensions.
Colloids include fog, smoke, shaving cream, milk, blood,styrofoam, gelatin, and cheese.
Suspensions include muddy water, paint and chalk powdersuspended in water.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
12/38
Definitions
Solution- A solution is
formed when one
substance called a
solute dissolves inanother called the
solvent.
Types of solution
State ofsolute
State ofsolvent
Example
solid liquid Sodiumchlori
de and water
solid solid Metal alloys
eg brass
gas Liquid Carbon
dioxide inbeverages
liquid liquid Alcohol and
water
-
7/29/2019 Mixture and Seperations
13/38
When two liquids form a solution they are said to be
miscible.
Suspension- A suspension is a type of mixtureformed when the solute does not dissolve in a
solvent. These particles settle on standing.
Liquids that do not dissolve in each other are said to
be immiscible.
A colloid is a mixture where the solute particles are
bigger than that of a solution but smaller than those
in a suspension. These particle do not settle onstanding and can pass through a filter paper.
-
7/29/2019 Mixture and Seperations
14/38
Emulsion- These are type of colloids where one
liquid is finely dispersed in another liquid.
Eg mayonnaise, oil dispersed in water
Summary of separation Technique
Type of mixture Separation
Method
Principle of method Example
Solid/solid Sublimation
Solvent
extraction
One substance
sublimes the other
does not
Differing solubilityin a particular
solvent.
Ammonium
chloride/sodium
chloride
Iodine/sodiumchloride
-
7/29/2019 Mixture and Seperations
15/38
solid/liquid
suspension
Solution
colloid
Decantation and
filtration
Evaporation/
Crystallisation
Simple
distillation
chromatography
Differing particle
sizes
Widely differing
boiling points
Differing
solubilities in apraticular
solvent leading
to differing
speed of
movement on
chromatogram
Chalk/water
Copper
sulphate/water
Screened
methyl orange
Liquid/liquid
Miscible
immiscible
Fractional
distillation
Separatingfunnel
Slightly differing
boiling points
Differingdensities
Ethanol/water
Oil/water
-
7/29/2019 Mixture and Seperations
16/38
16
Quick check 1
1. Explain why air is a mixture but wateris a compound.
2. (a), State two reasons why a mixture of iron filingsand sulphur before heating is a mixture.(b) When the mixture of iron filingsand sulphur is heated strongly, statetwo reasons why the solid formed
is a compound.3. State whether the following diagrams on the rightrepresent elements, mixtures or compounds.
Solution
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
17/38
17
Solution to Quick check 1
1. Air is a mixture because it is made up of many gases such as oxygen,nitrogen, etc. not chemically combined together. The gases in air can beseparated by physical means. Water is a compound because it is made up
of hydrogen and oxygen chemically joined together. We cannot separatethe gases in water by physical means.
2. (a) It is a mixture because the iron and sulphur can be separated byphysical means e.g. by using a magnet. Also, no heat or light is given outwhen the iron filings and sulphur are mixed together.(b) When the mixture of iron filings and sulphur is heated strongly, a
compound is formed because a chemical reaction occurs e.g. heat andlight are given off. Also, the sulphur and iron in the compound can nolonger be separated by physical means such as by using a magnet.
3. (a) compound, (b) compound, (c) mixture, (d) mixture
Return
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
18/38
Methods of separating Mixtures
Filtration
Evaporation
Crystallisation
Sublimation
Simple distillation
Fractional distillation
Use of separating funnel
Chromatography
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
19/38
Filtration
Filtration is the method used to separate an insoluble solid from a liquid.
An example would be to separate a mixture of sand and water.
The liquid (water) that
has passed through
the filter paper is called
filtrate.
The solid (sand) left on thefilter paper is called residue.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
20/38
Evaporation
Evaporation is the method used to separate a solute from a solution.
It can only be used for solids which do not decompose under heat e.g.table salt (sodium chloride).
It is done by heating the solution in an evaporating dish until it is completelydry.
Crystals of salt remain after the solution is evaporated to dryness.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
21/38
Crystallisation Crystallisation is the method used to obtain pure crystals from a solution.
It is done by heating the solution in an evaporating dish until it is saturated.
The hot solution is then allowed to cool.
Crystals will be formed on cooling. They are then dried between sheets of filter
paper.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
22/38
Sublimation
Sublimation is the method used toseparate a substance which sublimesfrom a mixture.
Examples of substances whichsublime are: iodine, ammonium salts,naphthalene and dry ice.
The mixture is heated in anevaporating dish covered witha filter funnel.
The substance which sublimeswill be changed into a vapourand then formed back on theinside of the funnel.
To separate ammonium chloride
from sodium chloride, this
method can be employed.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
23/38
Simple distillation
Distillation is the method used toobtain a pure solvent from asolution. E.g. pure water from
seawater. It is done by heating the solution in
a distillation flask and collecting thevapour that boils off.
A condenseris used to condensethe hot vapour and change it to a
liquid. The condenser is cooled by the
flow of cold water (in thecondenser) from the tap.
The pure liquid collected from
distillation is called distillate.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
24/38
Fractional distillation
Fractional distillation is used to separatetwo or more miscible liquids with differentboiling points.
A fractionating column and acondenserare used.
Example: Mixture of ethanol and water
When the thermometer reaches a steady
temperature of78 oC,ethanol is collected.
Water is collected when thethermometer reaches a steadytemperature of 100 oC.
Chapter 8Mixtures and Separations
Separation of ethanol-water mixture
by fractional distillation
-
7/29/2019 Mixture and Seperations
25/38
Use of separating funnel
This method is used to separate a mixture ofimmiscible liquids.
The mixture is placed into a separating funneland allowed to settle into two layers.
Example: Mixture of oil and water
The tap is first opened to allow the water todrain out of the funnel.
After all the water has drained out, anotherbeaker is placed below the funnel and oil isnow drained out.
Separation of oil-water mixture
by using a separating funnel
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
26/38
Chromatography
Chromatography is a method used toseparate and identify small quantities ofsubstances.
There are different kinds of chromatographytechniques, such as gas chromatography,liquid chromatography and paperchromatography.
It is based on the principle thatdifferent substances have different
solubilities in the same solvent. The more soluble substance will get
carried along faster by the solventand move further ahead than theless soluble substances.
Chromatography can be used todetect and identify very smallquantities of substances. It is usedto detect banned dyes used infood colourings, and detectillegal drugs used by athletesduring major competitions.
Paper Chromatography
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
27/38
Setup for paper chromatography
-The starting line must be drawn in
pencil, not ink. This is because unlike
ink, pencil lead is insoluble in the
solvent and will not interfere with thechromatogram.
-The spots of mixtures must be placed
above the solvent level, so that they
will not immediately dissolve in the
solvent, and the solvent has time to
slowly move up.
starting line
solvent
solvent front
chromatographypaper
large
beaker
glass cover
-The solvent front must be allowed to move as far up the paper as possible to
ensure that all the dyes are separated.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
28/38
The Rfvalue of a substance
Rfvalue = distance moved by a substancedistance moved by solvent front
(7 cm)(10 cm)
E.g. Rfvalue of red dye = 7 cm = 0.7
10 cm
Sometimes the chromatogram needs to be sprayed with a locating
agent to make the colourless spots in the chromatogram appear.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
29/38
Paper chromatography
Worked example
The chromatogram shows 3 single dyes
red, green and blue, and also fourunknown samples P, Q, R and S.
Identify the dyes present in each of the
samples, P, Q, R, S.
SolutionSample P contains green dye and one unknown dye.
Sample Q contains only blue dye.
Sample R contains green, blue and red dyes.
Sample S contains green and red dyes.
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
30/38
30
Identify the various method separation methods used in the diagram below .
Chapter 8Mixtures and Separations
-
7/29/2019 Mixture and Seperations
31/38
Extraction of sucrose from sugarcane
The cane sugar stalks and harvested and brought tofactory for processing
The cane stalks are cleaned by passing then alongan agitated conveyer belt which passes strong jets
of water over the cane stalks to remove dust, smallrocks and leaves.
The stalks are passed into shredder which containsrevolving knives which cut stalks into chips
The chips are passed into crusher consisting of twoor three heavily grooved rollers. At the same time asthe chips are crushed , hot water is sprayed onto thechips to dissolve the sugar in the stalks.
e u ce t at s extracte s an ac c ar green qu
-
7/29/2019 Mixture and Seperations
32/38
e u ce t at s extracte s an ac c, ar green quwhich contains sediments.
The next step is clarification. The juice is neutralisation of
calcium hydroxide and the addition of heat, insoluble salts
are formed by the reaction between calcium hydroxide and
sediment impurities. The non- sugar impurities are
removed by continuous filtration producing a final clarifiedjuice, which is 85% of water.
The clarified juice goes into a series of three or four evaporators.
The evaporators are under successively lower pressure so that asthe juice passes from one to the next it boils at successively low
temperatures .In this way the juice is concentrated but not charred or
caramelized by the boiling process. The juice from the last
evaporator is a thick syrup containing only 35% water.
The next step is crystallisation which take place in a single stage
-
7/29/2019 Mixture and Seperations
33/38
The next step is crystallisation which take place in a single stage
vacuum pan .The syrup is evaporated until saturate with sugar .As
soon as the saturation point has been exceeded, small grains of
sugar (called seed) are added to the pan to serve as nuclei for theformation of sugar crystals .As the crystals form, the remaining syrup
becomes thick and viscous and is called molasses. The mixture of
crystals and molasses form massecuite.
Molasses and sugar in the massecuite are separated by spinning in
centrifuges. Each centrifuge contains a perforated basket .The
massecuite is placed in the basket and is spun at high speed .The
sugar crystals stay behind in the basket and the molasses is forcedout through the holes.
-
7/29/2019 Mixture and Seperations
34/38
Quick check
1. State the method you will use toseparate the following substances.(a) calcium carbonate from table salt(b) iodine from sodium chloride(c) table salt from seawater(d) sugar from sugar solution(e) pure water from sewage water(f) ethanol from beer(g) yellow dye from durian ice cream
2. Explain the following in chromatography.
(a) Why is the starting line not drawn with ink or a ball point pen?(b) The spots of samples on the start line should be small.(c) What is the biggest advantage of chromatography?
3. A sample of ink was analysed using paper chromatography (seediagram above). Identify the dyes present in the ink.
Solution
Chapter 8Mixtures and Separations
Ch t 8
-
7/29/2019 Mixture and Seperations
35/38
Solution to Quick check
1. State the method you will use to separate the following substances.(a) dissolution, followed byfiltration(b) sublimation(c) evaporation(d) crystallisation(e) distillation(f ) fractional distillation(g) chromatography
2. Explain the following in chromatography.(a) Ink contains dyes which could dissolve in the solvent and
interfere with the chromatogram.(b) So that they would not smudge the paper.(c) It can detect and identify very small amounts of substances.
3. Blueandyellow dyes
Return
Chapter 8Mixtures and Separations
Ch t 8
-
7/29/2019 Mixture and Seperations
36/38
36
3. Hold a magnet over the mixture of
iron filings and sulphur.
The iron filings are attracted by the magnetand can be separated from the sulphur.
4. Heat the mixture of sulphur and iron
filings strongly in an evaporating dish.
Examine the substance formed carefully. A black solid is formed. It does not look
like iron filings or sulphur.
Compound of iron and sulphur
Mixture of iron and sulphur
Experiment
Chapter 8Mixtures and Separations
Ch t 8
-
7/29/2019 Mixture and Seperations
37/38
37
5. Hold a magnet over the black solid. What happens?
The black solid is not attracted by the magnet.
The iron can no longer be separated from the sulphur in the black
solid.
6. When iron filings and sulphur are heated,
a compound called iron(II)sulphide is formed.
The equation for the reaction is:
Fe(s) + S(s) FeS(s)
Experiment
Chapter 8Mixtures and Separations
Ch t 8
-
7/29/2019 Mixture and Seperations
38/38
1. http://orgchem.colorado.edu/hndbksupport/filt/filtration.html
2. http://en.wikipedia.org/wiki/Distillation
3. http://orgchem.colorado.edu/hndbksupport/dist/dist.html
4. http://en.wikipedia.org/wiki/Fractional_distillation
5. http://www.chemistrydaily.com/chemistry/Chromatography
To find out more about Methods of Purification and
Analysis, click on the links below!
Chapter 8Mixtures and Separations
http://orgchem.colorado.edu/hndbksupport/filt/filtration.htmlhttp://en.wikipedia.org/wiki/Distillationhttp://orgchem.colorado.edu/hndbksupport/dist/dist.htmlhttp://en.wikipedia.org/wiki/Fractional_distillationhttp://www.chemistrydaily.com/chemistry/Chromatographyhttp://www.chemistrydaily.com/chemistry/Chromatographyhttp://en.wikipedia.org/wiki/Fractional_distillationhttp://orgchem.colorado.edu/hndbksupport/dist/dist.htmlhttp://en.wikipedia.org/wiki/Distillationhttp://orgchem.colorado.edu/hndbksupport/filt/filtration.html