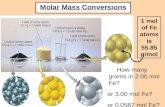
mgng molecules ftoI¥e x atoms - Open Computing Facility · I mol H atoms H: (11.19g) 1.008 I mol O...
1
by Remi Leano | Fall 2019 ocf.berkeley.edu/~rleano CHEM001 NOTESHEET : CHAPTERS 7 & 8 CHAPTER 7 EMPIRICAL FORMULAS TELL YOU THE MOLECULAR FORMULAS ELL YOU HOW MANY LAW OF MULTIPLE PROPORTIONS : TWO SIMPLEST OR MOST REDUCED RATIO OF ATOMS OF EACH ELEMENT ARE IN THE COMPOUNDS CAN HAVE IDENTICAL ELEMENTS IN A COMPOUND . COMPOUND . EMPIRICAL FORMULAS AND DIFFERENT MOLECULAR FORMULAS . AVOGADRO 'S NUMBER × ( # ATOMS PER ÷ molar mass MOLECULE ) GRAMS - MOLES # MOLECULES - # ATOMS ng x mg = my x AVOzmomfY = molecules x ftoI¥e = atoms CHAPTER 8 =
Transcript of mgng molecules ftoI¥e x atoms - Open Computing Facility · I mol H atoms H: (11.19g) 1.008 I mol O...

by Remi Leano | Fall 2019 ocf.berkeley.edu/~rleano
CHEM001 NOTESHEET : CHAPTERS 7 & 8
CHAPTER 7
EMPIRICAL FORMULAS TELL YOU THE MOLECULAR FORMULAS ELL YOU HOW MANY LAW OF MULTIPLE PROPORTIONS : TWO
SIMPLEST OR MOST REDUCED RATIO OF ATOMS OF EACH ELEMENT ARE
IN THE COMPOUNDS CAN HAVE
IDENTICAL
ELEMENTS IN A COMPOUND . COMPOUND .EMPIRICAL FORMULAS AND DIFFERENT
MOLECULAR FORMULAS .
AVOGADRO 'S NUMBER
× (# ATOMS PER÷molar mass MOLECULE )
GRAMS - MOLES # MOLECULES - # ATOMS
ngxmg = myxAVOzmomfY = molecules xftoI¥e = atoms
CHAPTER 8
=