MDACC MR Research Edward F. Jackson, PhD Department of Imaging Physics Challenges in Quantification...
-
Upload
juniper-flynn -
Category
Documents
-
view
222 -
download
0
Transcript of MDACC MR Research Edward F. Jackson, PhD Department of Imaging Physics Challenges in Quantification...

MDACC MR Research
Edward F. Jackson, PhD
Department of Imaging Physics
Challenges in Quantification of DCE-MRI
ACRIN DCE-MRI Workshop – October 2, 2009

MDACC MR Research
Outline
• General MR quantification challenges
– Lack of standards • Varying measurement results between vendors
– Highly variable quality control procedures• Varying measurement results between centers
• DCE-MRI quantification challenges
– Data acquisition
– Data processing
– Quality control
– Vendor specific issues
– Multicenter trial issues

MDACC MR Research
General Challenges in MR Quantification
Arbitrary (and spatially- / temporally-dependent) signal intensity units
– Magnitude and homogeneity of Bo
– Magnetic field gradient nonlinearity and/or miscalibration
– RF coil dependency: RF coil type, B1 sensitivity profiles, subject positioning within the coil
– Slice profile variations (with RF pulse shape, flip angle, etc.)
– System stability issues (RF & gradient subsystems, Bo, RF coils, etc.)

Bo Homogeneity
•In general: Larger B0 => higher signal-to-noise
•B0 inhomogeneity yields spatially variant signal intensities in general and spatially variant fat suppression when chemically selective saturation methods are utilized.

MDACC MR Research
Arbitrary (and spatially- / temporally-dependent) signal intensity units
– Magnitude and homogeneity of Bo
– Magnetic field gradient nonlinearity and/or miscalibration
– RF coil dependency: RF coil type, B1 sensitivity profiles, subject positioning within the coil
– Slice profile variations (with RF pulse shape, flip angle, etc.)
– System stability issues (RF & gradient subsystems, Bo, RF coils, etc.)
General Challenges in MR Quantification

MDACC MR Research
Gradient Field Nonlinearity Effects
Reference: Sumanaweera TS et al., Neurosurgery 35(4):696-703, 1994.
In-Plane Through-Plane Through-Plane
Slice at isocenter

MDACC MR Research
Isocenter
Errormax with correction: < 1 mm @ 10 cm
Errormax w/o correction: ~ 4.5mm @ 10 cm
20 cm off isocenter
Errormax with correction: < 2 mm @ 10 cm
Errormax w/o correction: ~ 5.5cm @ 10 cm
20 cm FOV, white: w/correction, black: w/o correction
Gradient Field Nonlinearity Effects

MDACC MR Research
Primary Limits on Spatial Accuracy
• System Limitations– Poor Bo homogeneity
– Linear scale factor errors in the gradient fields
– Field distortion due to induced eddy currents
– Nonlinearities of the gradient fields
• Object-Induced– Chemical shift effects (fat / water displacement, in-plane and slice)
– Intravoxel magnetic susceptibility differences (particularly air-tissue)
– Effects are minimized with non-vendor specific appropriate acquisition parameters (increased BW, smaller FOV), but at the expense of SNR. [Importance of Technique!]

MDACC MR Research
Arbitrary (and spatially- / temporally dependent) signal intensity units
– Magnitude and homogeneity of Bo
– Magnetic field gradient nonlinearity and/or miscalibration
– RF coil dependency: RF coil type, B1 coil sensitivity profiles, subject positioning within the coil
– Slice profile variations (with RF pulse shape, flip angle, etc.)
– System stability issues (RF & gradient subsystems, Bo, RF coils, etc.)
General Challenges in MR Quantification

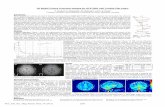
B1 Coil Response Non-Uniformity
B1 response non-uniformity
1.5T
3.0T

MDACC MR Research
Arbitrary (and spatially- / temporally-dependent) signal intensity units
– Magnitude and homogeneity of Bo
– Magnetic field gradient nonlinearity and/or miscalibration
– RF coil dependency: RF coil type, B1 sensitivity profiles, subject positioning within the coil
– Slice profile variations (with RF pulse shape, flip angle, etc.)
– System stability issues (RF & gradient subsystems, Bo, RF coils, etc.)
General Challenges in MR Quantification

MDACC MR Research
Slice profile variations (with RF pulse shape, flip angle, etc.)
5 mm SE 5 mm fast GRE
5.78 mm5.07 mm
Typically, faster imaging sequences use increasingly truncated RF pulses resulting in thicker slice profiles for a given prescribed slice thickness. This gives rise to increased partial volume averaging effects and a concomitant loss of spatial resolution.
Flip angle calibrations can also be negatively affected.
General Challenges in MR Quantification

MDACC MR Research
Arbitrary (and spatially- / temporally-dependent) signal intensity units
– Magnitude and homogeneity of Bo
– Magnetic field gradient nonlinearity and/or miscalibration
– RF coil dependency: RF coil type, B1 sensitivity profiles, subject positioning within the coil
– Slice profile variations (with RF pulse shape, flip angle, etc.)
– System stability issues (RF & gradient subsystems, Bo, RF coils, etc.)
General Challenges in MR Quantification

MDACC MR Research
System stability issues (RF & gradient subsystems, Bo, RF coils, etc.)
For quantitative imaging, particularly in longitudinal studies, a rigorous quality control program is critical.
Key components of frequent QC tests:• Geometric accuracy
• Slice thickness
• Signal-to-noise ratio (or low contrast object detectability)
• Uniformity
• High contrast spatial resolution
• Center frequency
• Transmit gain
• Contrast response
General Challenges in MR Quantification

ADNI
• Multicenter, multivendor study
• Optimized pulse sequence / acquisition parameters for each platform
• MagPhan/ADNI phantom scan at each measurement point
• Access to vendor gradient correction parameters
• With full correction for gradient nonlinearities and optimized acquisition strategies, spatial accuracies of ~0.3 mm can be obtained over a ~180 mm spherical volume

MDACC MR Research
DCE-MRI Quantification Challenges

MDACC MR Research
Outline
• General MR quantification challenges
– Lack of standards • Varying measurement results between vendors
– Highly variable quality control procedures• Varying measurement results between centers
• DCE-MRI quantification challenges
– Data acquisition
– Data processing
– Quality control
– Vendor specific issues
– Multicenter trial issues

MDACC MR Research
Dynamic Contrast Enhanced-MRI
3.0 T 3D FSPGR
20 3.5-mm sections every 4.5 s
0.86-mm in-plane resolution
Ktrans Parametric Map

Two Compartment Pharmacokinetic Model
CP = [Gd] in plasma (mM) = Cb / (1-Hct)
CEES = [Gd] in extravascular, extracellular space (mM)Ktrans = endothelial transfer constant (min-1)
kep = reflux rate (min-1)
vP = fractional plasma volume, ve = fractional EES volume (= Ktrans / kep)
Standardized parameters as proposed by Tofts et al., J Magn Reson Imaging, 10:223-232, 1999.
Ktrans
Plasma Flow
PlasmaCP, vP
EESCEES, ve
Endothelium
kep
( ')
0( ) ( ') 'ep
t k t ttransEES PC t K C t e dt CL(t) = vP CP(t) + CEES(t)
0 2 4 6 8 100
1
2
3
Time (min)
[GdD
TP
A]
(mM
)
CP(t)
CL(t)
MDACC MR Research
Measured Measured

MDACC MR Research
Signal intensity data from tumor
and vascular ROIs
'
0'
1
ept k t ttrans
P P P
L
K C t e v C tC t
Hct
0,1
0
01
0,10,1
0,10,1
1cos1
1coscos1
ln1
Re
SS
e
eSS
e
TRR
RTRRTR
RTRRTR
1
1
1
0,11][r
R
r
RRGd
Determine vP, Ktrans, and kep, from non-linear Marquardt-Levenberg fitting of CL(t) and CP(t) data
DCE-MRI

MDACC MR Research
[GdDTPA] (mM)
(S -
S0)
/ S
0 (%
)
Spin-Echo: TE/TR = 10/400 ms
FSPGR: TR/TR = 2/10 ms, =250
r1/r2 = 4.5 / 5.5 mM-1 s-1
T1,0 =1250 ms ; T2,0 = 125 ms
FSPGR
SE
DCE-MRI – Pulse Sequences
Several different pulse sequences and variations can be used– Most common: fast spoiled gradient echo, with or without magnetization
preparation pulses

MDACC MR Research
DCE-MRI Data Acquisition Challenges
• Pulse sequence
– Contrast response must be well characterized
• Temporal resolution
– Must match choice of pharmacokinetic model and parameters of interest• Must be rapid (~4-6 s) for generalized kinetic model with estimation of vp
• Recommended to be ≤15 s for any pharmacokinetic model
• Spatial resolution
– Must be adequate for target lesion size and application
• Anatomic coverage
– Should fully cover target lesion(s) and include an appropriate vascular structure

MDACC MR Research
DCE-MRI Data Acquisition Challenges
• Effects of motion
– Liver / lung / breast• Respiratory motion
• Cardiac motion
– Head & neck• Swallowing
• Tongue / jaw movement
– Brain and other “fixed” anatomy• Involuntary motion, e.g., when contrast agent is injected
• Effects should be prospectively mitigated, if possible, or retrospectively addressed (rigid body or deformable registration)

MDACC MR Research
DCE-MRI Data Acquisition Challenges
• Vascular input function (VIF)
– Needed to account for variations in cardiac output and contrast agent delivery parameters (i.v. access location, gauge of needle, position of arm, injection rate for contrast agent and flush, etc.)
– Requires good temporal resolution
– Acquisition strategy should:
• Minimize motion artifacts due to pulsatile flow
• Minimize the impact of flow related enhancement (in axial acquisition transverse to major vessel) on VIF, e.g., use in-plane reference vessel, spatial saturation pulse (at a cost of temporal resolution), etc.

MDACC MR Research
DCE-MRI Data Acquisition Challenges
• T1 measurement
– Required if contrast agent concentration, as opposed to signal intensity change, is used in the pharmacokinetic modeling
– Must be accomplished in a reasonable acquisition time
– Options widely available on scanners:• Inversion recovery --- accurate, but too slow
• Saturation recovery --- also slow
• Variable flip angle fast gradient echo techniques --- can be acquired in acceptable scan times, but can yield widely varying T1 estimates depending on specific pulse sequence and acquisition parameters
– T1 measurement “noise” is propagated into estimates of Ktrans and other pharmacokinetic model parameters.

MDACC MR Research
VFA T1 MeasurementsMDACC VFA MDACC VFA
Region 20090907* Flip + 5** Average Diff T1
1 738.22 1124.59 931.41 386.37
2 422.02 563.15 492.59 141.13
3 556.35 770.53 663.44 214.18
4 1356.10 1406.91 1381.51 50.81
5 468.94 639.36 554.15 170.42
6 704.67 1052.80 878.74 348.13
7 955.35 1234.30 1094.83 278.95
8 405.00 513.88 459.44 108.88
9 490.49 659.58 575.04 169.09781.24 110.936 <=stdev
27.8 217.43 <=1.96*stdev207.55 <=mean difference
*Flip angles: 2, 5, 10, 15, 20, 25, 30 - per QIBA protocol
**Flip angles: 7, 10, 15, 20, 25, 30, 35
3D FSPGR TE=1ms, TR=5.1ms

MDACC MR Research
Ktrans Simulations – T1 Dependence
Ktrans vs Calculated Lesion T1
0.00
0.04
0.08
0.12
0.16
0.20
1000 1100 1200 1300 1400
T1 (ms)
Ktr
an
s (
min
-1)

MDACC MR Research
DCE-MRI Data Acquisition Challenges• Quality Control
– To reduce system-level variance in longitudinal measurements, a robust quality control program is needed
– The ACR MR Accreditation Program is the most widely implemented MR QC program, but does not address all issues important in DCE-MRI, e.g., contrast response and signal stability.
• Use of multiple “matched” scanners
• “Upgrade Dilemma”
– If a system is upgraded during an ongoing longitudinal study, a thorough evaluation of the impact(s) of such an upgrade on DCE-MRI sequences and acquisition parameters (and contrast response, signal stability, etc.) must be performed.

MDACC MR Research
From Single- to Multi-Vendor Studies
• Major challenges:
– Protocol matching
• Pulse sequence and acquisition parameter selection for matched:– contrast response– temporal resolution– spatial resolution– anatomic coverage
• Can be achieved, but requires effort at start up and, subsequently, constant monitoring for changes in hardware/software (need for ongoing quality control)
– Vendors focused on “competitive advantage” in diagnostic radiology, not on quantitative imaging applications

MDACC MR Research
From Single- to Multi-Center Studies
• Major challenges:
– Matched protocols
– Distribution and activation of protocols• Distribute/load electronically (ADNI)
• Provide expert training and initial protocol load/test
• Rely on local expertise
– Compliance with protocol• Local radiologists, technologists
– Widely varying quality control• Ranging from specific for DCE-MRI, to ACR MRAP, to none
– “Scanner upgrade dilemma”
– Data management / submission

MDACC MR Research
• Data analysis implementation strategies are as variable as acquisition strategies
• Currently, DCE-MRI analysis tools based upon pharmacokinetic modeling are developed by the users as no such tools are currently commercially available from the MR vendors
• Choice of model must match data acquisition strategy, e.g., temporal resolution of the acquired data
• To facilitate testing/validation of various analysis packages, readily available, standardized test / retest data and analysis results (Ktrans, kep, ve, vp) are needed.
DCE-MRI Data Analysis Challenges

MDACC MR Research
• Many choices to be made:
– Vascular input selection
• Manual ROI, automated identification vascular structure pixels, etc.
• Reproducibility
– Lesion region of interest(s)
• Definition criteria
• Reproducibility
– Fits of single averaged pixel uptake curve or pixel-by-pixel fits
– Modeling of: [Gd](t) (requiring T1 mapping) or SI(t) data
DCE-MRI Data Analysis Challenges

MDACC MR Research
Quantitative Imaging Biomarkers – Who’s Interested?
NIST USMS Workshop 2006
Representative Agencies / Organizations

MDACC MR Research
General As Yet Unmet Needs
To move MR imaging biomarkers from exploratory / secondary endpoints to primary endpoints / surrogate markers:
– There exists a need for standardized acquisition pulse sequences and analysis techniques for MR “imaging biomarker” studies.
– Validated phantoms and test data need to be available to users in order to test new releases of pulse sequences and analysis software.
– Specific phantoms should be available to validate each vendor’s acquisition techniques for the particular MR biomarker technique (lesion morphology, perfusion, diffusion, MR spectroscopy, etc.).

MDACC MR Research
General As Yet Unmet Needs
To move MR imaging biomarkers from exploratory / secondary endpoints to primary endpoints / surrogate markers:
– Imaging biomarker to tissue-based and outcome measure comparisons are needed for validation.
– Reproducibility (test/retest) studies are lacking in several key areas.

MDACC MR Research
Multi-center trial design = Variations in imaging
Hardware & Software algorithms
• Standardize imaging techniques across multi-centers • Cross-consistency - within & across imaging systems
• Don’t assume – assess!– Discover & adjust for differences
George Mills, MD, MBA (former Director FDA/CDER Division of Medical Imaging & Radiopharmaceutical Drug Products)
Need for Standardized Imaging Techniques

MDACC MR Research
• “Precision is the goal”
• Implement the same, detailed imaging acquisition protocols at all clinical sites
• Optimize image processing & reconstruction software– Avoid manual techniques as possible– Select & develop semi-automated => automated– Reduce human interactions
• “Established” vs. “cutting edge”
Need for Standardized Imaging Techniques
George Mills, MD, MBA (former Director FDA/CDER Division of Medical Imaging & Radiopharmaceutical Drug Products)

MDACC MR Research
Quantitative MR Efforts
• NCI: RIDER / SAIC Contracts
• IRAT: MR Committee / Phantom Development
• RSNA:Quantitative Imaging Biomarker Alliance DCE-MRI Cmte
• ISMRM: Ad Hoc Committee for Standards for Quantitative MR
• AAPM: Working Group for Standards for Quantitative MR Measures

MDACC MR Research
NCI RIDER
• NCI Cancer Imaging Program RIDER – Reference Image Database to Evaluate Response*
Collaborative project for development and implementation of a caBIG public resource
Series of manuscripts in Translational Oncology in Dec 2009 and
data made publically available through NCIA (phantom and anonymized clinical trial imaging and meta data)

MDACC MR Research
NCI RIDER Reports
• Clarke L, Croft B, Nordstrom R, Zhang H, Kelloff G. Editorial: Quantitative imaging for evaluation of response to cancer therapy
• Meyer CR, Armato III SG, Fenimore CP, McLennan G, Bidaut LM, Barboriak DP, Gavrielides MA, Jackson EF, McNitt-Gray MF, Kinahan PE, Petrick N, Zhao B. Quantitative imaging to assess tumor response to therapy: common themes of measurement, truth data and error sources
• McNitt-Gray MF, Bidaut LM, Armato III SG, Meyer CR, Gavrielides MA, McLennan G, Petrick N, Zhao B, Reeves AP, Beichel R, Kim H-JG, Kinnard L. CT assessment of response to therapy: tumor volume change measurement, truth data and error
• Kinahan PE, Doot RK, Wanner-Roybal M, Bidaut LM, Armato III SG, Meyer CR, McLennan G. PET/CT assessment of response to therapy: tumor change measurement, truth data and error
• Jackson EF, Barboriak DP, Bidaut LM, Meyer CR. MR assessment of response to therapy: tumor change measurement, truth data and error sources

MDACC MR Research
NCI RIDER DCE-MRI Phantom Data
Gel-filled compartments with varying T1 relaxation times
Eurospin TO5 – DiagnosticSonar, Ltd.

MDACC MR Research
RIDER T1 Measurements – Vendor 1
AMR7 Run 1 MultiFlip vs IR
y = 1.0907x - 15.548
R2 = 0.9981
0
500
1000
1500
2000
0 500 1000 1500 2000
IR T1 (ms)
Ave
Mu
ltif
lip
T1
(ms)
Week 0 Run 1 vs Run 2 (AMR7)
y = 0.9935x + 2.1855
R2 = 0.9999
0
500
1000
1500
2000
0 500 1000 1500 2000
Run 1 T1 (ms)
Ru
n 2
T1
(ms)
Week 0 vs Week 1 (AMR7)
y = 0.9838x + 2.5057
R2 = 0.9999
0
500
1000
1500
2000
0 500 1000 1500 2000
Week 0 T1 (ms)
Wee
k 1
T1
(ms)
Run 1 = baseline Run 2 = 2 hrs post baseline Week 1 = 1 week post baseline
Bosca & Jackson, AAPM 2009; Jackson et al., Trans Oncol, Dec 2009

MDACC MR Research
RIDER T1 Measurements – Vendor 2
Run 1 = baseline Run 2 = 2 hrs post baseline Week 1 = 1 week post baseline
Bosca & Jackson, AAPM 2009; Jackson et al., Trans Oncol, Dec 2009

MDACC MR Research
Imaging Response Assessment Teams
• IRAT: MR Committee / Phantom Development
– Current status:
• MR phantom based on the commercially available MagPhan/ADNI phantom (The Phantom Laboratory).
• Phantom distributed to IRAT MR sites to obtain multicenter, single vendor data.
• Similar to, but pre-dates, RSNA QIBA DCE-MRI project acquisition and data analysis strategy

MDACC MR Research
RSNA QI Biomarker Alliance
• RSNA QIBA: DCE-MRI Technical Committee
– Multiple subcommittees:
• Phantom development/selection
• Scan protocol / data analysis
• Synthetic DCE-MRI test data
– MR phantom based on the IRAT DCE-MRI phantom
– Phantoms to be distributed to multiple MR sites to obtain multicenter (N=6), multivendor (N=3) data
– Data analysis provided by core lab
http://qibawiki.rsna.org/index.php?title=DCE-MRI

MDACC MR Research
Phantom Scans• Phantom and “cuboid” scanned using torso phased array coil to mimic
typical Phase I clinical trial scans (liver mets / lung mets).
RSNA QI Biomarker Alliance

MDACC MR Research
• RSNA QIBA: DCE-MRI Technical Committee
– Phantom measurements:
• Phased array acquisition
• Body coil acquisition
• SNR acquisition
• VFA T1 measurement acquisition
• DCE acquisition
– Each of the above acquisitions repeated with phantom rotated by 90, 180, 270, and 360o
– All acquisitions repeated one week later
Ratio map correction for B1
sensitivity characteristics
RSNA QI Biomarker Alliance

RSNA QIBA DCE-MRI
Difference in T1 from each contrast sphere, week 1 minus week 0.
Difference in R1 from each contrast sphere, week 1 minus week 0.

RSNA QIBA DCE-MRI
Comparison of R1 vs Signal Intensity at Rotation “A”

MDACC MR Research
ISMRM Ad Hoc Committee
• ISMRM: Ad Hoc Committee for Standards for Quantitative MR
– Membership includes MR physicists, technologists, radiologists, NIST representative, NCI/CIP representatives, vendors, pharma. Expertise in research trials using quantitative MR.
– Current status:
• White paper on quantitative MR
• Design specifications & construction of a MR system phantom (collaboration with and funding by NIST)
• Initial multicenter/multivendor phantom pilot studies to begin in late 2009 / early 2010.
http://wiki.ismrm.org/twiki/bin/view/QuantitativeMR/

ISMRM SQMR System PhantomSpatial accuracy
Contrast response
Section thickness
High contrast resolution
0.7 0.8, 0.9, 1.0, 1.1 mm
All materials fully characterized by NIST.

ISMRM SQMR System PhantomT1 Compartments T2 Compartments
PD Compartments

MDACC MR Research
AAPM Working Group
• AAPM: Working Group for Standards for Quantitative MR Measures– Charge
• Development of quantitative MR phantoms and acquisition techniques / parameters specific to a given quantitative measure. Obtaining repeat measures on each phantom at multiple centers and using current high-field scanners from all major vendors.
• Develop signal response characteristic standards for relevant pulse sequences. These response characteristic standards will allow ongoing QC at individual centers on a given platform (across upgrades) and comparisons between vendor platforms (and hardware/software versions.
• Develop sets of annotated test data that can be publicly distributed (via the NCIA, for example) and used to validate image analysis software tools.

MDACC MR Research
RSNA QI Initiatives
RSNA
TQI
(Toward Quantitative Imaging)
Education / Clinical Practice
QIBA
(Quantitative Imaging Biomarker
Alliance)
Biomarker precision / instrumentation
Imaging Biomarker Roundtable
Communication amongst various
societies & groups
CTSA Imaging Working Group
Translational research
infrastructure
Based on information from Dan Sullivan, MD

MDACC MR Research
Quantitative Imaging Initiatives
Uniform Protocols for Imaging in Clinical Trials
(UPICT - CTSA)
Imaging Biomarker Quality Control / Phantom
Development Groups
(NIST, FDA, Inter-Society and Inter-Agency WGs)
NCI caBIG Imaging Workspace
(NCIA, LIDC, RIDER)
Imaging Response Assessment Teams / Quantitative Imaging
Network (NCI)
NCI / FDA / NIST
Scientific Societies
Imaging Scientists / Animal Imaging Cores
Imaging Equipment Vendors, Pharma, & CROs