Lipids
description
Transcript of Lipids
-
1Lipids Chapter 2 Lipids Chapter 2
DR. LEONARDO C. MEDINA, JR.
-
2Chapter Outline1. Lipids: Hydrophobic Molecules
2. Classification of Lipids
3. Simple Lipids
4. Compound Lipids
5 Steroids
6 Hydrophobic Lipids and Biology
-
3Lipids: Hydrophobic Lipids: Hydrophobic MoleculesMolecules
-
4 Lipids are water insoluble, oily, greasy biochemical compounds that can be extracted from cells by nonpolar solvents such as ether, choroform, or benzene.
Unlike carbohydrates, lipids share no common chemical structure.
-
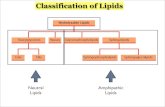
5Classification Classification of Lipidsof Lipids
-
6Lipid structures vary markedly. The following classification scheme recognizes important structural differences:
1. Simple lipids
2. Compound lipids
3. Steroids
4. Miscellaneous lipids
-
7Simple LipidsSimple Lipids
-
8Fatty Acids
Fatty acids, which form part of most lipids, are carboxylic acids with long, hydrophobic carbon chains.
All the fatty acids listed in Table are straight-chain compounds with an even number of carbon atoms.
-
9
-
10
Unsaturated Fatty Acids
Unsaturated fatty acids may be cis or trans isomers.
The trans isomer is almost a linear molecule, while the double bond in the cis isomer introduces a kink into the fatty acid structure.
-
11
-
12
Eicosanoids The biochemicals derived from the fatty
acid arachidonic acid are collectively termed eicosanoids.
Prostaglandins are the best known of the eicosanoid class, which also includes the leukotrienes, prostacyclins, and thromboxanes.
Cell membranes release arachidonic acid in response to a variety of circumstances, including infection and allergic reactions.
-
13Several examples of eicosanoids.
-
14
Drugs and Eicosanoids
Many drugs control one or more of the eicosanoids physiological effects.
Non-steroidal anti-inflammatory drugs (NSAIDs) block the oxidation of arachidonic acid to form prostaglandins and thromboxanes. e.g. ibuprofen, ketoprofen,
-
15
Fats and Oils Chemically, fats and oils are esters of
glycerol and the higher molar-mass fatty acids.
H C O
C OH
C OH
H
H
C R
O
C R'
O
C R"
Oglycerolportion
-
16
Fats may be considered to be triesters formed from the glycerol and three molecules of fatty acids.
Most of the fatty acids in these esters have 14-18 carbons. Because there are three ester groups per glycerol, the
molecules are called triacylglycerols, or triglycerides. The three R groups are usually different.
H C O
C OH
C OH
H
H
C R
O
C R'
O
C R"
Oglycerolportion
-
17
Waxes Waxes are esters of high-molar-mass fatty acids
and high-molar-mass alcohols. The alcohol (ROH) contributes up to about 30
carbons, and the fatty acid (RCOOH) also provides an equivalent number of carbons.
Waxes are very large molecules with almost no polar groups.
ROCR'
O
-
18
Compound LipidsCompound LipidsCompound Lipids
-
19
Phospholipids The phospholipids are a group of compounds that yield one
or more fatty acid molecules, a phosphate group, and usually a nitrogenous base upon hydrolysis.
A lipid with both hydrophobic and hydrophilic character is needed to make membranes. Phospholipids are one of the most important membrane components.
H2C O
HC O
H2C O
fatty acid
fatty acid
phosphate + nitrogen base
hydrophilic
hydrophobic
-
20
Phospholipids Phosphatidic acids are gylceryl esters of fatty acids
and phosphoric acid. The phosphatidic acids are important intermediates
in the synthesis of triacylglycerols and other phospholipids.
H2C O
HC O
H2C O
fatty acid
fatty acid
phosphate
hydrophilic
hydrophobic
-
21
Phospholipids Phosphatidyl cholines (lecithins) are glyceryl esters
of fatty acids, phosphoric acid, and choline. Phosphatidly cholines are synthesized in the liver
and are present in considerable amounts in nerve tissue and brain substance.
H2C O
HC O
H2C O
fatty acid
fatty acid
phosphate + choline
hydrophilic
hydrophobic
-
22
Phospholipids Phosphatidyl Ethanolamines (Cephalins) are
glyceryl esters of fatty acids, phosphoric acid, and ethanolamine (HOCH2 CH2 NH2 ).
H2C O
HC O
H2C O
fatty acid
fatty acid
phosphate + ethanolamine
hydrophilic
hydrophobic
-
23
Sphingolipids Sphingolipids are compounds that, when
hydrolyzed, yield a hydrophilic group (either phosphate and choline or a carbohydrate), a long- chain fatty acid (18-26 carbons), and sphingosine (an unsaturated amino alcohol).
HC CH=CH(CH2)12CH3
HC NH2
H2C OH
OH
-
24
Glycolipids Sphingolipids that contain a carbohydrate group
are also known as glycolipids. cerebrosides and gangliosides
HC CH=CH(CH2)12CH3
HCHN
H2C O-D-glucose or D-galactose
OH
C
O
R
-
25
SteroidsSteroids
-
26
Steroids Steroids are compounds that have the
steroid nucleus, which consists of four fused carbocyclic rings.
-
27
Structure of cholesterol and cholic acid.
-
28
Biosynthetic relationships between steroids derived from cholesterol.
-
29
Hydrophobic Lipids Hydrophobic Lipids and Biologyand Biology
-
30
The hydrophobic nature of lipids has many important biological consequences.
1. Lipid aggregation that causes atherosclerosis
2. Lipid aggregation that forms biological membranes
-
31
1. Lipid molecules will aggregate to minimize their contact with water.
2. Depending on the general shape of the lipid molecule, different-shaped aggregates form.
3. Smaller lipids like fatty acids will come together to make micelles.
-
32
CH3(CH2)nCOOH
hydrophobic hydrophilic
If the fatty acid is shown as depicted below, then a micelle can be visualized as.
-
33
Cutaway of a micelle. Note that the hydrophobic chains extend to the center of this aggregate.
-
34
More complex lipids such as phospholipids and sphinglolipids are shaped differently than fatty acids and will aggregate differently in water solutions. They form liposomes
hydrophobichydrophilic
-
35
A cutaway of a single liposome. Note the bilayer shell and the water solution core.
-
36
Atherosclerosis
Atherosclerosis is a metabolic disease that leads to deposits of cholesterol and other lipids on the inner walls of the arteries.
Improper transport of cholesterol through the blood contributes to atherosclerosis.
-
37
Atherosclerosis
Cholesterol (and other lipids) must be packaged for transport because lipid aggregates in the aqueous bloodstream.
The liver packages dietary lipid into aggregates known as very-low-density- lipoproteins (VLDL).
-
38
A cutaway of VLDL. Note the single-layer shell and the lipid core.
-
39
The lipid distribution system through the bloodstream.
-
40
Biological MembranesBiological Membranes
-
41
The fluid-mosaic model of a membrane. Note the hydrophilic exterior and the hydrophobic interior.
-
42
How proteins aid membrane transport: Molecules or ions (symbolized by Y or X) can move from high concentration to low concentration without energy (facilitated diffusion), but movement in the reverse direction requires energy (active transport).
-
43
Lipids Chapter 2 Chapter OutlineLipids: Hydrophobic MoleculesSlide Number 4Slide Number 5Slide Number 6Simple LipidsFatty AcidsSlide Number 9Unsaturated Fatty AcidsSlide Number 11EicosanoidsSlide Number 13Drugs and EicosanoidsFats and OilsSlide Number 16WaxesCompound LipidsPhospholipidsSlide Number 20Slide Number 21Slide Number 22Slide Number 23Slide Number 24SteroidsSteroidsSlide Number 27Slide Number 28Hydrophobic Lipids and BiologyThe hydrophobic nature of lipids has many important biological consequences.Slide Number 31Slide Number 32Slide Number 33Slide Number 34Slide Number 35AtherosclerosisSlide Number 37Slide Number 38Slide Number 39Slide Number 40Slide Number 41Slide Number 42Slide Number 43