Limitations on Friedel-Crafts Alkylation There are three major limitations on Friedel-Crafts...
-
Upload
prosper-robinson -
Category
Documents
-
view
217 -
download
2
Transcript of Limitations on Friedel-Crafts Alkylation There are three major limitations on Friedel-Crafts...

Limitations on Friedel-Crafts Alkylation
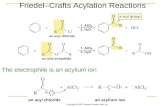
There are three major limitations on Friedel-Crafts alkylations.1. carbocation rearrangements are common.

+ CH3CH2CH2-Br, AlCl3
CHCH3H3C
AlCl3+
C CH3H3C
CH3
isopropylbenzene
tert-butylbenzeneCH3CHCH2-Br
CH3
Examples of Carbocation rearangementExamples of Carbocation rearangement

2. alkylation fails on benzene rings bearing one or more of these strongly electron-withdrawing groups.
Y
RXAlCl3
SO3H NO2 NR3+
CF3 CCl3
C N
CHO
CRO
COHO
CORO
CNH2
O
+ No reaction
When Y Equals Any of These Groups, the BenzeneRing Does Not Undergo Friedel-Crafts Alkylation

Examples:
Br
+ CH3CH2-Br, AlCl3
Br Br
CH2CH3
CH2CH3+
COOH
+ CH3-Br, AlCl3 NR
+ CH3-Br, AlCl3 NR
NO2

3. alkylation fails on benzene rings bearing amine groups.
Basic amino groups (–NH2,–NHR, & –NR2) react with lewis acids (AlCl3 )
Not Friedel-Crafts reactive

Arenes:
Hydrocarbons contain both aliphatic and aromatic parts.
1.Alkylbenzenes
2. Alkenylbenzenes
3. Alkynylbenzenes
CH=CH2 styrene
C CH Phenylacetylene

Alkylbenzene
syntheses:1. Friedel-Crafts alkylation
H3CH2C Br+
AlBr3
CH2
H3C

2-Acylation of Aromatic Rings then reduction of the carbonyl group by Zn(Hg)/HCl
Met
hod A
giv
es
100 %
10 al
kylb
enze
ne
Method B gives
Mixture of alkylbenzene
Note: method A: acylation then reduction method B: friedel crafts alkylation

Reactions of Alkylbenezene on Side chain
1- oxidation:
Alkyl side chains can be oxidized to CO2H by strong reagents such as KMnO4 and Na2Cr2O7 if they have a C-H next to the ringConverts an alkylbenzene into a benzoic acid, ArR ArCO2H

2- Free radical halogenation in side chain:
Br2 / light
Note: halogen atom adds to the benzyl carbon atom (the carbon atom directly bonded with benzene ring
Benzyl carbon

CH2CH3
benzyl free radical > 3o > 2o > 1o > CH3
CHCH3
CHCH3CHCH3 CHCH3.
.
.
.
X2 2 X.
+ X .
Mechanism:

Alkenylbenzenes
syntheses:1. modification of side chain
CHCH3
CHCH3
C
CH=CH2
CH
Br
OHstyrene
H+, heat
H2, Pd-C
KOH

+ CH2=CH-Br no reactionAlBr3
Note1: Friedel-Crafts alkylation not normally used for alkenylbenzene synthesis
Vinyl bromide

Reactions of Alkenylbenzenes
1- oxidation
CH=CH2
CH=CH2
CH=CH2
CHCH2
COOH
CH=O
OHOH
+ CO2
+ O=CH2
KMnO4
heat
1. O3
2. Zn, H2O
KMnO4

2 -reduction
CH=CHCH3 CH2CH2CH3
H2, Ni
CHCHCH3
CHCH2CH3
CHCH2CH3
Br
Br
Br
OSO3H
Br2, CCl4
HBr
H2SO4
CH=CHCH3
3 -Addition

CH=CH2 + Cl2, Fe CHCH2
Cl Cl
CH2CH3 CH2CH3CH=CH2
Cl
CHCH3Cl2, FeCl
ClCl
Cl2, hvKOH(alc)
p-chlorostyrene
Note 2:alkenes are more reactive than aromatic rings In syntheses of alkenylbenzenes, the carbon-carbon double bond must be synthesized after any EAS reactions
If is started by EAS in the presence of double bond the addition will take place in stead of halogenation

How we can prepare:
1. P- chlorostyrene
2. P-bromobenzoic acid
3. Allylbenzene

CH2CH3 CH2CH3
CH=CH2
Cl
CHCH3
CH3-CH2Cl
AlCl3
Cl2, FeCl3
Cl
ClCl
Cl2, hv
p-chlorostyrene
KOH
1. P- chlorostyrene
Answers:

+ CH2=CHCH2-Br, AlCl3 CH2CH=CH2
allylbenzene
2. P-bromobenzoic acid
3. Allylbenzene