Learning Keys , Lehninger Chapter # 10 LIPIDS
-
Upload
tauqeer-ahmad -
Category
Education
-
view
315 -
download
7
Transcript of Learning Keys , Lehninger Chapter # 10 LIPIDS

Lehninger Principles of BiochemistryFourth Edition
David L. Nelson and Michael M. Cox
Fourth Edition
Chapter 10:
Lipids
Copyright © 2004 by W. H. Freeman & Company

Biological Importance
1. Fats & oils�storage lipids2. Phospholipids & sterols�structural lipids3. Others
i. Cofactorsi. Cofactorsii. Electron carriersiii. Light-absorbing pigmentsiv. Hydrophobic anchors for proteinsv. “chaperone” to help membrane protein foldvi. Emulifying agents in digestive tractvii. Hormonesviii. Intracellular messenger

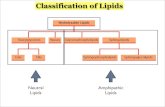
1. STORAGE LIPIDS
• Fats & Oils�derivatives of fatty acids (FA)
– FA�hydrocarbon
triacylglycerols
– FA�hydrocarbon derviatives like fossil fuel
– Burn them �exergonic reaction
– Two example: triacylglycerols andwaxes

Fatty Acids• FA=Carboxylic acid + hydrocarbon chain(HC)
– HC�between 4-36 carbon long (C4 to C36)
– Saturated HC�no double bond
– Unsaturated HC�one or >one
triacylglycerols
– Unsaturated HC�one or >one– A few has OH, methyl-group branch, rings

Nomenclature of FA
• Palmitic Acid wth 16C, saturated: 16:0
• Oleic Acid wth 18C, one double: 18:1
• To show the position of double bonds
triacylglycerols
• To show the position of double bonds– ∆ (delta) used: How? ∆no of Catom
• Example: # of C=20; # of DB=2, where?
between C-9&C-10
C-12&C-13
SO
20:2 (∆∆∆∆9,12)

triacylglycerols

Common
• most monounsaturated FA� ∆9
• Most polyunsaturated FA� ∆9,12 or ∆9,15
triacylglycerols

cis-configuration � highly commonTrans-configuration: produced by microp of rumen of ruminantsAnd during the hydrogenation of fish and vegetable oils
triacylglycerols
Trans FA���� LDL (bad cholesterol)

Physical properties
Depending on # of Carbon& # of double b.The more hydrocarbon�the more hydrophobic
The fewer double bond�the more hydrophobic
triacylglycerols
The fewer double bond�the more hydrophobic
The more hydrocarbon�the more melting point
The higher double bond�the lower melting point

tria
cylg
lyce
rols

triacylglycerols

triacylglycerols

Melting
• No double bond– Atoms close each other
– Possibility of van der Waals
triacylglycerols
– Possibility of van der Waals
• Size– Carboxyl side is polar; polar/hydrocarbon
• Double bond number– Not enough, cis configuration (kink)

Triacylglycerols
triacylglycerols

Types of triacylglycerols
• Simple triacylglycerols: only one kind of fatty acids (example: 16:0�tristearin)
• Mixed triacylglycerol (mostly): 2 or more
triacylglycerols
• Mixed triacylglycerol (mostly): 2 or more different fatty acids.
• Triacylglycerols�nonpolar
• Lipids�lower speicifc gravity than water
2 layerswater
oil

Roles of Triacylglycerols
• Store energy • Insulation
• Stored in cell as oily droplets serving as depots of
triacylglycerols
• Stored in cell as oily droplets serving as depots of metabolic fuel – adipocytes for vertebrates�fat droplets– As oil in seeds of plants
• Lipases: enzyme to hydrolyse TA to fatty acids to be transported anywhere as an energy battery

triacylglycerols

triacylglycerols

Why body uses TA to store energy
• More reduced than sugar�twice energy when hydrolysed
• Hydrophobic�unhydrated�less space but
triacylglycerols
• Hydrophobic�unhydrated�less space but higher energy (2 g water/1 g polysaccharide to hydrate it)– 15 kg TA � months
– Full glycogen depots supply less than one day’s energy

TA for insulation
• TA under skin� low temperature– Seals, walruses, penguins etc
triacylglycerols
– Seals, walruses, penguins etc (warm-blooded polar animals) �full of TA
– Hibernating animals (bear) both energy and insulation

triacylglycerols

Simple and mixed TA in food.
So different FA in sizeand saturation degree
triacylglycerols

Waxes
Waxes

Waxes

• Function of waxes depends on• Waxes are water repellent
• Firm consistency
• Waxes for hair� pliable (easy to bend), lubricated, waterproof
Waxes
lubricated, waterproof
• Birds’ feather
• Tropical plants’ leaves for water loss
• Industeial usage • Ointments, lotions and polishes from waxes

Structural lipids in Membrane
• A barrier• A double layer of lipids• Amphipatic (charged and hydrophobic)


I II III IV V
Called Phospholipid Called glycolipidCalled Phospholipid
Why?
Polar group to hydrophobicmoeity by
Phosphodiester bond
Called glycolipid
Why
Sugar at polar head

1. Glycerophospholipid
Glycerophospholipid

Glycerophospholipid

Glycerophospholipid

Fatty Acids
• Glycerophospholipid Fatty Acids
• Differences in different organism, tissues, even cells
Glycerophospholipid
• Generally– C-1 to C16 or C18
– C-2 to C18 or C20

Phospholipids with ether linked fatty acids: Ether lipids
Ester •Vertebrate heart tissue (plasmalogen�%50 of phospholipids)phospholipids)
•Halophilic (salt lovers) bacteria
•Cilliated protists
•Certain invertebrates


Galactolipids : plants (most abundant)
Localized in thylokoid membranein chloroplast
Make up 70-80% of total membrane lipid
No phosphate intake
Plants also have sulfolipids (sulfonated glucose to a diacylglycerol in glycosidic linkage)
-1


Archeabacteria’s membrane lipids
(Hydrocarbon)
Glycerol dialkyl glycerol tetraethers (GDGTs)

Sphingolipids
•4th biggest group•Polar head + 2 nonpolat tail•No glycerol•Majority � no phosphate group•Extra sphingosine•Its strucutre: 1 sphingosine + 1 FA + head


• Three subclass of sphingolipids1. Sphingomyelins
2. Glycosphingolipids
3. Gangliosides

• Three subclass of sphingolipids1. Sphingomyelins
– Phosphocholine (polar head group –X)– So called glycerophospholipid (due to phosphate group)
– Animal cells– Prominent in myelin sheat around axons
2. Glycosphingolipids2. Glycosphingolipids
3. Gangliosides

• Three subclass of sphingolipids1. Sphingomyelins
2. Glycosphingolipids– Generally outer face of plasma membrane
– Sugar (1 or 2) to ceramidine wthout phosphate g.
– Cerebroside: single sugar– Glucose����nonneural tissue
– Galactose����neural tissue
– Globosides: neutral – 2 or more sugar
– D-glucose; D-galactose; N-acetyl-D-galactosamine
3. Gangliosides

• Three subclass of sphingolipids1. Sphingomyelins
2. Glycosphingolipids
3. Gangliosides– most complex one
– Oligosaccharide + N-acetylneuraminic acid (sialic – Oligosaccharide + N-acetylneuraminic acid (sialic acid)– Negative charge due to sialic a.
– Accumulated in outer surface of plasma membrane
– Kinds and amount change during development



Degraded in lysosomes

Sterols
• With steroid nucleus ( 4 fused rings; 3X6+1X5) – Planar– Rigid– No rotation– No rotation– Most eukaryotics’ membrane– Plant can synthesize – Animals can do– But bacteria can not (a few exception)– Funciton: some in signaling, in the structure of bile
acids (cholestrol)


Lipids with active role passive role
1. Potent signals (hormones)
2. Enzyme cofactor (in cellular respiration and photosynthesis and in transfer of sugar moeties)
• Vitamines –A, D, E ,K
3. Pigment molecules (to absorb light to see (eye) or to produce chemical energy (leaf), colorization (orange of carrot)
4. volatile lipids (communication of plants)

Phosphatidylinositol: Regulate metabolism
•Phosphatidylinositol and its phosphorylated derivatives: phosphatidylinositol 4,5-bisphosphate �inositol 1,4,5-trisphosphate (IP3)
•some regulatory proteins binds to phosphatidylinositol 3,4,5-bisphosphate
•source of intracellular messengers.

Eicosanoids
• Paracrine hormone (near point)
• involved in– Reproduction– Reproduction
– inflammation
– Pain
– Fever
– Formation of blot clots
– Regulation of blood pressure
– Gastric acid segration
– Etc



• Prostaglandins: prostate gland– Two known groups
– PGE ether-soluble (subgroups PGE1 ...)
– PGF water soluble
– Regulation of cAMP• Regulation of celular and tissue functions

• Thromboxanes– By thrombocytes
• In blood clotting

• Leukotrienes– in leukocytes
– Powerful signals
– leukotriene D4, � contraction of the muscle lining the airways to the lung.
– Overproduction� asthmatic attacks
– strong contraction of smooth m. of lung (anaphylactic shock) � potentially fatal allergic reaction in individuals hypersensitive to bee stings, penicillin, or other agents

Steroid hormones
• derivatives of sterols– lack alkyl chain attached to D ring of cholesterol
• SH�produced�blood stream�enter cell�binds to protein�protein expression
• [low]�enough because of its ↑affinity
• major Steroid hormones:– sex hormones,
– hormones from adrenal cortex: • cortisol, aldosterone

male sex hormoneorgan: testis
female sex hovary and placenta
regulation ofglucosemetabolism
regulation of saltextraction
both from adreanl cortex
synthetic drug for inflammation
potent growthregulator invascular plants

Volatile Signals
• lipophilic compound for plant to…..– attrack pollinators and helpful organisms
against enemiesagainst enemies
– repel herbivors
– communicate with friends
– exmple: josmante

Vitamins
• vitamins �essential (mostly not produced by animals, most in diet)
• grouped into two• grouped into two– soluble in polar s. (water-soluble vitamins)
– soluble in nonpolar solvent(fat-soluble v)• A,D,E,K: isoprenoid compounds by condansation of
poly-isoprene units

Vitamin D3
• cholecalciferol – from skin from 7-dehydrocholesterol
• ultiately converted to 1,25-d,hydroxyvitamin D33
– hormone which regulates ……..• calcium uptake by intestine
• calcium level in kidney and bones
– interact directly with its target protein in nucleus
• deficincy �defective bone formation and rickets
• vitamin D2�by yeast and similar effect like D3
– added into milk

Vitamin A• retinol
• various forms functioning as…..– hormone such as retinoic acid
• receptor of cell nucleus to regulate gene in epithelial • receptor of cell nucleus to regulate gene in epithelial cell development
– visual pigment as retinal• initiate response of rop and cone to light
• sources: fish oil, liver,egg,milk, butter
• beta-carotene: pigments that gives carrots, sweet potatoes, yellow veetables
• Syptoms:dryness in skin,eyes and mocous, night blidness


•Tocopherols•Aromatic ring + along isoprenoid side chain
•Hydrophobic so associate with•Cell membrane•Lipid droplets•Lipoproteins
•Antioxidant because of its aromatic ring•Sources: egg, vegetable oil, white germ•Deficiency����rare; symp: RBC fragility

•A cycle of oxidation & reduction during the formation of •A cycle of oxidation & reduction during the formation of active protrombin
•active protrombin: an enzyme converting fibrinogen to fibrin
• fibrin: insoluble protein holding bloot clot together•Deficiency�rare; only in new borns•Source: green plants, K2 intestinal bacteria






Working with Lipids
• First degradation– Alkali
– Acidic– Acidic
– Hydrolytic enzymes• Glycosidase
• phospholipase

Working with Lipids
• Extraction– Neutral lidips (TG, waxes)by the
solvents• Ethyl ether, chloroform, benzen
(hydrophobic)
– Membrane Lidips by more polar solvents
• Ethanol, methanol
– Example: chloroform, ethanol, water (1:2:0.8)
– Then add water
» Two layers:lipids in chloroform

Seperation by..•Adsorption chromotography
•Silica gel: polar but insoluble•Polar lipids����adsorb to silica gel
•Neutral Lipids����eluted
•To remove polar lipids����polar solvents:
•For polar, uncharged:acetone•Charged: methanol
•TLC:•Very similar to Ad. Ch.•Very similar to Ad. Ch.•Plate with silica•Neutral lipids run faster•Trick in staining lipids
•Spray(a dye: RHODAMINE )•Đodine fumes: yellow-brown
•Đnteract with double bond•Other commercials