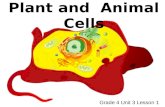
Plant Cells: Comparing Plant Cells with Animal Cells By: Brianna Shields October 12, 2004.
Immobilization of plant cells
-
Upload
aachal-jain -
Category
Science
-
view
31 -
download
0
Transcript of Immobilization of plant cells

IMMOBILIZATION OF PLANT CELLS
Aachal D. JainMsc. IIPaper 1

INTRODUCTION Plant cell culture has been for sometime
considered as an alternative method for the production of flavors, colors and pharmaceuticals to their extraction from plants.
One of the major limiting factors in the development of a commercial production system using plant cell culture has been the production cost of phytopharmaceuticals.
Immobilization of plant cells would be one method of increasing productivity and hence reducing the costs.

Immobilization is the newest culture technology of plant cell, and considered as to be the most “natural”.
It has been defined as a technique, which confines the cells to a defined region in a space while retaining their catalytic activity and prevents its entry into the mobile phase, which carries the substrate and product.
Immobilization of plant cells, protoplast or embroyos is achieved by binding these materials onto or within a solid support.

NEED FOR IMMOBILIZATION Protection from degradation and deactivation. Retention of enzyme, enzyme-free products. Retention of enzyme, enzyme-free products. Cost efficiency. Enhanced stability. Use as controlled release agents. The ability to stop the reaction rapidly by
removing the enzyme from the reaction Solution (or vice-versa).
Allows development of multi-enzyme reaction system.

MATRIX/SUPPORT USED IN IMMOBILIZATION TECHNIQUE
The matrix/support used should be cheap and easily available.
Their reaction with medium and cells should be as minimum as possible.
The matrix/supports are grouped into three major categories:
1. Natural Polymers: Alginate, chitosan and chitin, starch, cellulose, collagen.
2. Synthetic Polymers: DEAE cellulose, PVC, PEG.3. Inorganic materials: silica, glass, charcoal,
activated carbon.

METHODS OF IMMOBILIZATION

ADSORPTION It is the oldest and simplest method of cell
immobilization. Involves the physical binding of biocatalyst on
the surface of carrier matrix. Carrier may be organic or inorganic. Types of adsorbents: Cellulose, Polystyrene
resin, Glass, Alumina, Silica gel. The process of adsorption involves the weak
interactions like Vander Waal, ionic interactions or hydrogen bonds.

Methods of immobilization by Adsorption
1) Static Process: This is most efficient technique but requires maximum time. In this technique, enzyme is immobilized by allowing it to be in contact with the carrier without agitation.
2) Dynamic Process: This process typically involves the admixing of enzyme with the carrier under constant agitation using mechanical shaker.
3) Reactor loading: This process is employed for the commercial production of immobilized enzymes. The carrier is placed into the reactor and enzyme solution is transferred to the reactor with agitation of the whole content in the reactor.4) Electro-Deposition: In this technique, carrier is placed in
the vicinity of one of the electrode in an enzyme bath and electric current is applied leading to migration of enzyme towards the carrier. This results in deposition of enzyme on the surface of the carrier.

Advantages Disadvantages Little or no
conformation change of the enzyme.
Simple and cheap. No reagents are
required. Can be Recycled,
Regenerated & Reused.
Efficiency is less. Slow method. Desorption of the
enzyme protein resulting from changes in temperature, pH, and ionic strength.

CROSS LINKING This method is based on the formation of
covalent bonds between the enzyme molecules, by means of multifunctional reagents, leading to three dimensional cross linked aggregates.
It is used mostly as a means of stabilizing adsorbed enzymes and also for preventing
leakage from polyacrylamide gels. The most common reagent used for cross-linking is glutaraldehyde. No matrix or support is involved.

Advantages Disadvantages
Very little desorption(enzyme strongly bound).
Best used in conjunction with other methods.
Cross linking may cause significant changes in the active site.
Not cost effective.

COVALENT BONDING Involves the formation of covalent bond
between enzyme and support that forms a stable complex
Most widely used method for immobilization. The most common technique is to activate a
cellulose-based support with cyanogen bromide, which is then mixed with the enzyme.
The protein functional groups which could be utilized in covalent coupling are: amino, carboxylic, phenol ring, and indole group. Carriers/support used: cellulose, agarose etc.

Methods of covalent bonding1. Diazoation: bonding between amino
group of support and thyrosil or histidyl group of enzyme.
2. Peptide bond: between amino/carboxyl groups of support and enzyme.
3. Poly functional reagents: Use of bi-functional or multi-functional reagent (gluteraldehyde) which forms the bonding between amino groups of support and enzymes.

Advantages Disadvantages
The strength of binding is very strong, so, leakage of enzyme from the support is absent or very little.
This is a simple, mild and often successful method of wide applicability
Enzymes are chemically modified and so many are denatured during immobilization.
Only small amounts of enzymes may be immobilized (about 0.02 grams per gram of matrix).

ENTRAPMENT The entrapment method of immobilization is
based on localization of an enzyme within the lattice of polymer matrix, gels or capsule.
Retains enzyme and allows the penetration of substrate.
Polymers like polyacrylamide, collagen, cellulose acetate, calcium alginate or carrageenan are used as the matrices.
It can be classified into lattice and micro capsule type.
Polymers like polyacrylamide, collagen, cellulose acetate, calcium alginate or carrageenan etc are used as the matrices.

Methods of Entrapment
1. Inclusion in gels: Enzymes trapped in gel. Eg: Poly acrylamide gel, Poly vinyl alcohol gels.
2. Inclusion in fibers: Enzymes supported on fibre format. Eg: Cellulose and Poly -acryl amide gels.
3. Inclusion in micro capsules: Enzymes trapped on microcapsules made by monomer mixtures of polyamine, polybasic acid chloride monomers.

Advantages Disadvantages Easy to perform Loss of enzyme
activity upon immobilization is minimized.
The enzyme can leak into the surrounding medium.
Another problem is the mass transfer resistance to substrates and products.
Substrate cannot diffuse deep into the gel matrix.

ECAPSULATION Enclosing cells in a semi permeable
membrane capsule. Capsule is made up of nitro cellulose or
nylon. Effectiveness depends upon stability of
enzymes.

Advantages Disadvantages
Cheap and simple method
Large number of cells can be immobilized by encapsulation.
Pore size limitation.
Only small substrate molecules are allowed to cross the membrane.

APPLICATIONS OF PLANT CELL IMMOBILIZATION
Enhanced production of secondary metabolites: Eg: capsaicin, L- DOPA, methyl xanthines etc.Biotransformation: Bioconversion of β methyl digitoxin has been achieved using Digitalis lanata immobilized cell cultures upto 70 days.Other uses:1. It is used in synthetic seed technology.2. It can be used for transport of protoplasts.3. Immobilized plant cells can be cultured as
single cells for prolonged period.

THANK YOU