II III I II. Factors Affecting Solvation (p. 471 – 477) Ch. 16 –Solutions.
-
Upload
jackson-ellis -
Category
Documents
-
view
215 -
download
2
Transcript of II III I II. Factors Affecting Solvation (p. 471 – 477) Ch. 16 –Solutions.

II
III
I II. Factors Affecting Solvation
(p. 471 – 477)
Ch. 16 –SolutionsCh. 16 –Solutions

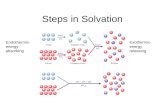
A. SolvationA. Solvation
Solvation – Solvation – the process of dissolving
solute particles are separated and pulled into solution
solute particles are surrounded by solvent particles

A. SolvationA. Solvation
StrongElectrolyte
Non-Electrolyte
solute exists asions only
- +
salt
- +
sugar
solute exists asmolecules
only
- +
acetic acid
WeakElectrolyte
solute exists asions and
molecules DISSOCIATION IONIZATION
View animation online.

B. Factors Affecting SolvationB. Factors Affecting Solvation
Molecules are constantly in motion according to…• Kinetic Theory
When particles collide, energy is transferred

B. Factors Affecting SolvationB. Factors Affecting Solvation
Solubility = amount of a substance that will dissolve in a liquid
Smaller pieces of a substance dissolve faster b/c of larger surface area
Stirring or shaking speeds dissolving b/c particles are moving faster and colliding more
Heating speeds dissolving (see above)Not all substances dissolve

C. SolubilityC. Solubility
Water is universal solvent b/c of its polarity
If something can dissolve in something else, it is said to be soluble
If it cannot dissolve, it is said to be insoluble
“Like dissolves like”

C. SolubilityC. Solubility
NONPOLAR
NONPOLAR
POLAR
POLAR
““Like Dissolves Like”Like Dissolves Like”““Like Dissolves Like”Like Dissolves Like”

C. SolubilityC. Solubility
SATURATED SOLUTION
no more solute dissolves
UNSATURATED SOLUTIONmore solute dissolves
SUPERSATURATED SOLUTION
becomes unstable, crystals form
concentration

C. SolubilityC. Solubility
Solubility CurvesSolubility Curves• maximum grams of solute that will
dissolve in 100 g of solvent at a given temperature
• varies with temp• based on a saturated soln

C. SolubilityC. Solubility
Solubility CurveSolubility Curve• shows the
dependence of solubility on temperature

C. SolubilityC. Solubility
Solids are more soluble at...Solids are more soluble at...• high temperatures.
Gases are more soluble at...Gases are more soluble at...• low temperatures &• high pressures (Henry’s Law).• EX: nitrogen narcosis, the “bends,” soda












![JSP 471 Defence Nuclear Emergency Response Part 1: Directive · ^}(^[ W }o] Ç^ u v ii JSP 471 Pt 1 (V2.1 Dec 18) Preface How to use this JSP 1. JSP 471 sets Ministry of Defence (MOD)](https://static.fdocuments.us/doc/165x107/5e733d23467d6302710782e7/jsp-471-defence-nuclear-emergency-response-part-1-directive-w-o-u-v.jpg)