IgA Nephropathy_New Updates
-
Upload
rahul-pabalkar -
Category
Documents
-
view
125 -
download
1
Transcript of IgA Nephropathy_New Updates

Current UpdatesCurrent Updates
Immunoglobulin A (IgA) Nephropathy
Dr. Om Kumar

IgA Nephropathy 2
Epidemiology
Most common cause of primary glomerulonephritis in most developed countries
Peak incidence in 2nd to 3rd decade of life
2:1 male to female ratio
Most common in Asians & Caucasians
Available evidence suggests an increasing incidence in India!
4.2% 1987
7 – 10% 1995
12-15% 2009
Chandrika BK. IgA nephropathy in Kerala, India: A retrospective study. Indian J Pathol Microbiol 2009;52:14-6
Tam
il Na
duKerala
Delhi

IgA Nephropathy 3
Clinical Features
40- 50% present with gross hematuria, usually following an upper respiratory infection
30- 40% present with microscopic hematuria and non-nephrotic proteinuria
<10% present with nephrotic syndrome or acute RPGN
Rarely presents with malignant HTN or AKI

IgA Nephropathy 4
Clinical Associations
Hepatic Cirrhosis
Gluten Enteropathy
HIV
Minimal Change
Membranous Nephropathy
Wegener’s
Dermatitis Herpetiformis
Seronegative Arthritis
Small Cell Carcinoma
Disseminated TB
Mycosis Fungoides

IgA Nephropathy 5
Diagnosis
Gold standard for diagnosis is Biopsy
IgA- fibronectin not used any longer
– Assays cannot reliably distinguish between free IgA and IgA-fibronectin complexes
Skin biopsy is not predictive of IgA nephropathy
Serum IgA levels are not reliable

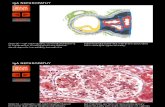
IgA Nephropathy 6
Pathology- Light Microscopy
Most common appearance is mesangial hypercellularity
Crescents and tubular sloughing are not uncommon with gross hematuria and renal insufficiency

IgA Nephropathy 7
Pathology- Immunofluorescence
Pathognomonic finding is prominent, globular deposits of IgA in the mesangium
Dominant form is polymeric IgA1
Minimal staining for C1q or its absence distinguishes IgAN from lupus nephritis

IgA Nephropathy 8
Pathology- Electron Microscopy
Electron dense deposits primarily in the mesangium
May have focal thinning of basement membrane

IgA Nephropathy 9
PathogenesisPathogenesis
Inciting event in the pathogenesis is mesangial deposition of IgA
Three key elements contribute to development of IgA nephopathy:
1. Persistence of characteristic pIgA1
2. “Reactivity” of glomerular mesangium
3. How the kidney responds

IgA Nephropathy 10

IgA Nephropathy 11
IgAIgA
Tetramer consisting of two identical light chains and two heavy chains
Two isotypes: IgA1 and IgA2
Two “compartments” of IgA: the mucosa associated lymphoepithelial tissue (MALT) and the bone marrow-plasma compartment
Circulating IgA contains 90% IgA1; 10% IgA2

IgA Nephropathy 12
IgA

IgA Nephropathy 13
IgA

IgA Nephropathy 14
Characteristics of IgA in IgAN
No single pathogenic antigen yet identified
Elevated IgA level by itself does not cause IgA nephropathy
Thought that abnormal glycosylation plays a role

IgA Nephropathy 15
IgA Production and Clearance in IgAN
Associated with upper respiratory tract infections, so thought that the mucosa was the site of IgA production
Studies have shown that abnormal pIgA1 comes from bone marrow rather than mucosal sites
In IgAN, have decreased hepatic clearance as well as decreased myeloid clearance, further leading to elevated pathogenic IgA levels

IgA Nephropathy 16
IgA and the Mesangium
In IgAN, rate of IgA deposition either exceeds mesangial clearance capacity or is somehow resistant to clearance
Not all IgA deposition is is associated with inflammation, and IgA deposition is not necessarily irreversible
Deposition may be enhanced by abnormal glycosolation

IgA Nephropathy 17
IgA and the Mesangium
May have abnormal clearance because have impaired binding of IgA to mesangial cell receptors, leading to accumulation
Once mesangial cells bind to IgA, triggers a proinflammatory and profibrotic responsehave increased expression of TGF-beta and components of renin-angiotensin sytem

IgA Nephropathy 18
Treatment - Nonimmunosuppressive Therapy
ANGIOTENSIN INHIBITION
– Reduce proteinuria by reducing intra- glomerular pressure and improving size-selective properties of the glomerular wall
– Used in patients with >500mg/day or if hypertensive
– No randomized controlled trial shows benefit of ACE-I and/or ARB, with most being underpowered

IgA Nephropathy 19
Treatment- Nonimmunosuppressive Therapy
FISH OIL
– Mechanism unclear, but likely due to anti-inflammatory properties
– Use limited by taste and patient compliance
– No consensus among studies on outcome with fish oil

IgA Nephropathy 20
Treatment- Immunosuppressive Therapy
Available studies have been small with limited follow-up, so most studies are inconclusive
Reserved for those with evidence of active inflammation

IgA Nephropathy 21
Treatment- Immunosuppressive Therapy
GLUCOCORTICOIDS
– SW Pediatric Nephrology Study Group randomized 96 patients to omega-3 fatty acids (4g/day); alternate day prednisone; or placebo
– Primary end point: reduction in GFR to below 60% of baseline
– At 3 years, primary outcome was seen more frequently in fish-oil group (19%) than in prednisone (9%) or placebo (9%)

IgA Nephropathy 22
Treatment- Immunosuppressive Therapy
COMBINATION THERAPY
– Lv et al randomized 63 patients to either ACE-I (Cilazapril) or ACE-I and prednisone (0.8mg/kg/day x 8 weeks, then tapered by 5-10mg every 2 weeks
– Primary end point: 50% increase in serum creatinine
– Secondary end point: 50% reduction in proteinuria

IgA Nephropathy 23
Treatment- Immunosuppressive Therapy

IgA Nephropathy 24
Treatment- Summary

IgA Nephropathy 25
Treatment- Other Interventions
TONSILLECTOMY
– No randomized trials showing a benefit
– One nonrandomized trial of 55 patients compared steroids alone vs steroids with tonsillectomy
– Those who received combination therapy had remission of proteinuria and hematuria at 2 years, as well as decreased mesangial proliferation and IgA deposition

IgA Nephropathy 26
Treatment- Other Interventions
IVIG
– Small studies show that may have reduction in proteinuria and stabilization of renal function
VITAMIN D
– Small, uncontrolled, short-term study decrease in proteinuria as measured by spot protein / creatinine

IgA Nephropathy 27
PrognosisPrognosis
<10% of patients have complete resolution of urinary abnormalities
1.5% of patients with IgA nephropathy reach ESRD per year
15-25% of any published cohort will require renal replacement therapy within 10 years of presentation; 20-30% at 20 years

IgA Nephropathy 28
Clinical Prognostic MarkersClinical Prognostic Markers
Poor Prognosis:
Severity of proteinuria
HTN
Renal impairment
Increasing age
Duration of preceding symptoms
Increased BMI
Good Prognosis:
Recurrent macroscopic hematuria
No Impact on Prognosis:
–Gender
–Ethnicity
–Serum IgA level

IgA Nephropathy 29
Histopathologic Prognostic Histopathologic Prognostic MarkersMarkers
Poor PrognosisLight Microscopy:
–Capsular adhesions & crescents
–Glomerular sclerosis–Tubule atrophy–Interstitial fibrosis–Vascular wall thickening
Immunofluorescence:–Capillary-loop IgA deposits
Ultrastructure:–Mesangiolysis–GMB abnormalities
Good Prognosis:
Minimal light microscopic abnormalities
No Impact on Prognosis:
Intensity of IgA deposits
Co-deposition of mesangial IgG, IgM, or C3

IgA Nephropathy 30
Transplantation
Frequency of histologic or clinically significant recurrence varies in the literature
Similar graft survival as those patients with non-IgA glomerular disease and non-glomerular disease
Large retrospective analysis from Australia showed an estimated 10-year incidence of graft loss due to recurrent disease of 9.7%
However, it is a very suitable alternative for patients

IgA Nephropathy 31
Transplantation
Likelihood of recurrence does not appear to be affected by the type of induction or maintenance immunosuppression used
Angiotensin inhibition may delay progression of recurrent disease
No clear evidence that graft survival is improved by using angiotensin inhibition once disease recurs
Case reports of fish oil have a favorable effect in recurrent IgA nephropathy, but no studies support this

IgA Challenges &
Opportunities!

Challenges

IgA Nephropathy 34
Opportunities available for new tests & treatments that may eventually lead to control of this chronic form of kidney disease
IgA Nephropathy poses many challenges in its Etiology, Pathogenesis, Prevention & Treatment
IgA Nephropathy is a significant contributor to the incidence of ESRD in many countries
Current Facts !
1
2
3

IgA Nephropathy 35
Diagnostic & Prognostic Challenges
Can IgA nephropathy be diagnosed without a renal biopsy?
Renal Biopsy remain the ‘Gold Standard’for diagnosis !
New, sensitive & reasonably noninvasive tests are emerging
– Test for Abnormally glycosylated IgA subclass I (IgA1)
– Such tests offer great promise for use in genetic & epidemiology studies, in which renal routine biopsy is impractical
1

IgA Nephropathy 36
Diagnostic & Prognostic Challenges
Can IgA nephropathy be diagnosed without a renal biopsy?
Renal Biopsy remain the ‘Gold Standard’ for diagnosis !
New, sensitive & reasonably noninvasive tests are emerging
– Test for Abnormally glycosylated IgA subclass I (IgA1)
– Such tests offer great promise for use in genetic & epidemiology studies, in which renal routine biopsy is impractical
1

IgA Nephropathy 37
Diagnostic & Prognostic Challenges
Signs & Symptoms are non-specific!
Most common clinical presentation – Macroscopic hematuria ± proteinuria
Similar presentation in thin basement membrane nephropathy, Alport syndrome and membranoproliferative glomerulonephritis
– IgA nephropathy can be reliably distinguished ONLY by renal biopsy and electron microscopy
2

IgA Nephropathy 38
Diagnostic & Prognostic Challenges
Can we better predict which patients with IgA nephropathy will develop renal failure?
Several factors, if present at the time of discovery or developing within a relatively short time thereafter (usually 6 months to 1 year), appear to predict a progressive course and, eventually ESRD
3

IgA Nephropathy 39
Diagnostic & Prognostic Challenges
Are clinical risk factors more useful than pathological risk factors in IgA nephropathy?
Clinical factors appear to have greater predictive power than pathologic factors for long-term outcome
If we can find new risk factors that can predict progressive disease earlier, the knowledge will help us in designing future clinical trials, which will be vital if progress is to be made towards controlling IgA nephropathy
4

IgA Nephropathy 40
Diagnostic & Prognostic Challenges
How can IgA nephropathy be diagnosed and treated before the ‘point of no return’?
IgAN progresses silently, and many patients do not receive the diagnosis until late in its course
The “point of no return” appears to be an estimated GFR of about 30 mL/min/1.73 m2 (stage 4 chronic kidney disease)
The need is for early diagnosis and treatment based on factors that can accurately predict an unfavorable outcome
The challenge is to translate these findings into rational, safe, and effective therapies applicable across a broad spectrum of disease
5

IgA Nephropathy 41
Diagnostic & Prognostic Challenges
How can IgA nephropathy be diagnosed and treated before the ‘point of no return’?
For patients at risk of developing ESRD, the two most critical goals are:– Control blood pressure rigorously, preferably
with ACE inhibitor, an ARB, or both &– Reduce proteinuria to less than 500 mg/ day
If these two goals can be met without undue side effects and patient compliance, many patients can avoid ESRD
5

Opportunities Genetics, Proteomics, New Tests &
Treatments

IgA Nephropathy 43
Opportunities - Genetics
Genetic studies may lead to novel treatments for IgA nephropathy!
At present, genetic testing based on genomic or transcriptosomic analysis does not have much diagnostic value
Most current genetic studies actually examine susceptibility to the clinical expression of disease rather than susceptibility to the mesangial IgA deposition that underlies the disease
The opportunity that lies ahead in genetic testing of IgA nephropathy (including haplotype analysis) appears to be primarily in the elucidation of potential pathogenetic pathways, in the refinement of prognosis and the definition of treatment responsiveness (pharmacogenomics)
If a gene (or group of genes) can be identified that is strongly and consistently associated with IgA nephropathy across diverse populations, then a new era in targeted therapy of IgA nephropathy will be unleashed!
1

IgA Nephropathy 44
Opportunities - Proteomics
Proteomics may prove useful in diagnosis and prognosis of IgA nephropathy!
– Preliminary studies have shown that this technique may provide a novel noninvasive means of diagnosing IgA nephropathy, and
– It may have additional value as a prognostic tool
2

IgA Nephropathy 45
Opportunities - IgA1 testing
IgA1 testing may help detect IgA nephropathy early in its course
– Abnormally undergalactosylated and oversialyted epitopes at the hinge region of the IgA1 molecule play a critical role in the pathogenesis of sporadic IgA nephropathy
– This discovery provides a great opportunity for profiling patients suspected of having IgA nephropathy on the basis of sensitive determination of the serum level of abnormal IgA1 molecules
3

IgA Nephropathy 46
Opportunities - IgA1 testing
IgA1 testing may help detect IgA nephropathy early in its course
– Abnormally undergalactosylated and oversialyted epitopes at the hinge region of the IgA1 molecule play a critical role in the pathogenesis of sporadic IgA nephropathy
– This discovery provides a great opportunity for profiling patients suspected of having IgA nephropathy on the basis of sensitive determination of the serum level of abnormal IgA1 molecules
3

IgA Nephropathy 47
Opportunities - IgA1 testing
IgA1 testing may help detect IgA nephropathy early in its course
3

IgA Nephropathy 48
Opportunities - Knowledge of secondary mediators
Knowledge of secondary mediators may also lead to new treatments for IgA nephropathy!
– Detailed knowledge of the participation of specific cell types and the “cytokine milieu” (eg, interleukin 4, interferon) in directing the abnormality toward defective glycosylation would also be very important in designing new approaches to diagnosis and therapy
– Lack of a suitable animal model of IgA nephropathy that mimics all aspects of the human condition
4

IgA Nephropathy 49
Opportunities - Knowledge of secondary mediators
Prognostic biopsy analysis may be improved in IgA nephropathy!
– Great opportunities lie in refining the value of renal biopsy in prognostication
– Much better clinico-pathological correlations, especially with respect to outcomes, among well-characterized patient with IgA nephropathy are greatly needed.
– New nonconventional markers of progression, such as “tubulitis,” deposition of fibroblastspecific proteins, and the proteome of the deposited immunoglobulins and complemen show much promise!
5

IgA Nephropathy 50
Opportunities – Addition of Immunosuppresors
Immunosuppressive therapy could be added to ACE inhibitors or ARBs in IgA nephropathy!
The clinical trials performed have a number of limitations:
– the numbers of patients were relatively small, follow-up was relatively short, and the findings may not apply to the IgA nephropathy population at large or to specific patients having features that diverge from those in the patients enrolled in the studies.
– The value of initial therapy with an ACE inhibitor, an ARB, or both in combination appears well established
– Many opportunities for combining Angiotensin II inhibition and immunosuppressive therapy are being explored!
6

Much Work Needs to be Much Work Needs to be done !done !

IgA Nephropathy 52
Summary
Much work needs to be done in the field of therapeutics in IgA nephropathy.
At present, the prospects for the development of a safe and effective novel therapy for IgA nephropathy (eg, approvable by the US Food and Drug Administration) appear great!
The nature of the disease mandates long-term observation, agents that are very safe (with low rates of ESRD, death, and transplantation), and dependency on surrogate markers of efficacy.
Well designed and executed studies are the need of the hour!

IgA Nephropathy 53

Additional Slides

IgA Nephropathy 55
Treatment- Nonimmunosuppressive Therapy
ANGIOTENSIN INHIBITION
– HKVIN trial randomized 109 Chinese patients to either valsartan or placebo
– Valsartan group had lower baseline proteinuria and achieved target BP
– Primary end point: doubling of serum creatinine or ESRD
– Fewer patients in valsartan group (1) reached primary endpoint than in placebo (4)

IgA Nephropathy 56
Treatment- Nonimmunosuppressive Therapy
ANGIOTENSIN INHIBITION
– IgACE trial randomized 65 patients to either benazepril or placebo
– Primary end point: >30% decrease in renal function
– Secondary composite end point: worsening or proteinuria or >30% decrease in renal function
– Fewer patients reached primary end point in benazepril group (1) than in the placebo group (5)

IgA Nephropathy 57
Treatment- Nonimmunosuppressive Therapy
ANGIOTENSIN INHIBITION
– Maschio et al randomized 44 patients to enalapril or other non-ACE/ARB antihypertensive
– Patients had >.5g/day proteinuria and Cr<1.6
– At six years, renal survival more likely in ACE-I group (92%) than in control (55%)
– Only enalapril group showed significant decrease in proteinuria

IgA Nephropathy 58
Treatment- Nonimmunosuppressive Therapy
FISH OIL
– Denadio et al in 1994 published in NEJM a study that randomized 104 patients to fish oil 12g daily vs placebo with olive oil
– Primary end point: 50% increase in serum creatinine
– At four years of follow-up, fish oil group had lower incidence of primary end point (6% vs 33%) and lower incidence of death or ESRD (10% vs 40%)
– Benefits continued at six years of follow up

IgA Nephropathy 59
Treatment- Nonimmunosuppressive Therapy
FISH OIL
– SW Pediatric Nephrology Study Group randomized 96 patients to omega-3 fatty acids (4g/day); alternate day prednisone; or placebo
– Primary end point: reduction in GFR to below 60% of baseline
– At 3 years, primary outcome was seen more frequently in fish-oil group (19%) than in prednisone (9%) or placebo (9%)

IgA Nephropathy 60
Treatment- Immunosuppressive Therapy
COMBINATION THERAPY
– Ballardie et al performed a single center study of 38 patients with IgAN with impaired renal function
– Randomized to no therapy or prednisone, cyclophosphamide, and azathioprine
– Those with combination therapy had significant reduction in proteinuria during the first 6 months (1.8g/day vs 4.4g/day) and higher renal survival at 2 years (82% vs 68%) and at 5 years (72% vs 6%)

IgA Nephropathy 61
References
Barratt J, Feehally J: IgA Nephropathy. J Am Soc Nephrol 16: 2088-2097, 2005
Donadio JV, Grande JP: A controlled trial of fish oil in IgA nephropathy. N Engl J Med 1994; 331:1194.
Li PK, Leung CB, Chow KM, et al: Hong Kong study using valsartan in IgA nephropathy (HKVIN): a double-blind, randomized, placebo-controlled study. Am J Kidney Dis 2006; 47:751.
Lv J, Zhang H, Chen Y, et al: Combination therapy of prednisone and ACE inhibitor vs. ACE inhibitor alone in patients with IgA nephropathy: a randomized controlled trial. Am J Kidney Dis 2009; 53:26.
Pozzi C, Bolasco PG, Fogazzi GB, et al: Corticosteroids in IgA nephropathy: A randomized controlled trial. Lancet 1999; 353:883.
Van der Boog PJM, van Kooten C, de Fitjer JW, Daha MW: Role of macromolecular IgA in IgA nephropathy. Kidney International 67: 813-21, 2005