IB Chemistry on Lewis Structure, Ionic and Covalent Bonding
-
Upload
lawrence-kok -
Category
Education
-
view
2.229 -
download
7
description
Transcript of IB Chemistry on Lewis Structure, Ionic and Covalent Bonding

http://lawrencekok.blogspot.com
Prepared by Lawrence Kok
Tutorial on Lewis Structure, Ionic and Covalent bonding.

brown liquid
Ionic Bonding Group 1 and 17 Gp 1 Metal2.1
2.8.1
2.8.8.1
2.8.8.18.1
Na
Li
K
Rb
yellow gas
greenish gas
violet solid
Ionic bonding between metal and non metal
Metal •Low EN value•Lose electron (cation)•Electropositive ions
Non metal •High EN value•Gain electron (anion)•Electronegative ions
Gp 17 Non metal

brown liquid
Ionic Bonding Group 1 and 17 Gp 1 Metal2.1
2.8.1
2.8.8.1
2.8.8.18.1
Na
Li
K
Rb
yellow gas
greenish gas
violet solid
Ionic bonding between metal and non metal
Metal •Low EN value•Lose electron (cation)•Electropositive ions
Non metal •High EN value•Gain electron (anion)•Electronegative ions
2.8.1 2.8.7
Gp 17 Non metal
transfer electron
2.8 2.8.8
achieve stable octet structure
+ -
Na+ CI-Electrostatic force attraction bet opposite charge ions

brown liquid
Ionic Bonding Group 1 and 17 Gp 1 Metal2.1
2.8.1
2.8.8.1
2.8.8.18.1
Na
Li
K
Rb
Click here video 0n NaCI
yellow gas
greenish gas
violet solid
Ionic bonding between metal and non metal
Metal •Low EN value•Lose electron (cation)•Electropositive ions
Non metal •High EN value•Gain electron (anion)•Electronegative ions
2.8.1 2.8.7
Gp 17 Non metal
transfer electron
2.8 2.8.8
achieve stable octet structure
+ -
Na+ CI-Electrostatic force attraction bet opposite charge ions
Crystal lattice structure•+/- ion attract/ arranged in orderly manner•arrangement in 3D shape•strong electrostatic forces attraction bet opposite charged ions
Reaction bet sodium with chlorine
Crystal lattice structure

brown liquid
Gp 2 Metal2.2
2.8.2
2.8.8.2
2.8.8.18.2
Mg
Be
Ca
Sr
yellow gas
greenish gas
violet solid
Ionic bonding between metal and non metal
Metal •Low EN value•Lose electron (cation)•Electropositive ions
Non metal •High EN value•Gain electron (anion)•Electronegative ions
Gp 17 Non metal

brown liquid
Gp 2 Metal2.2
2.8.2
2.8.8.2
2.8.8.18.2
Mg
Be
Ca
Sr
yellow gas
greenish gas
violet solid
Ionic bonding between metal and non metal
Metal •Low EN value•Lose electron (cation)•Electropositive ions
Non metal •High EN value•Gain electron (anion)•Electronegative ions
2.8.2 2.7
Gp 17 Non metal
transfer electron
2.8
achieve stable octet structure
Mg2+ F-
Electrostatic force attraction bet opposite charge ions
2.8. 82.8
F-

brown liquid
Gp 2 Metal2.2
2.8.2
2.8.8.2
2.8.8.18.2
Mg
Be
Ca
Sr
Click here simulation ionic bonding
yellow gas
greenish gas
violet solid
Ionic bonding between metal and non metal
Metal •Low EN value•Lose electron (cation)•Electropositive ions
Non metal •High EN value•Gain electron (anion)•Electronegative ions
2.8.2 2.7
Gp 17 Non metal
transfer electron
2.8
achieve stable octet structure
Mg2+ F-
Electrostatic force attraction bet opposite charge ions
Crystal lattice structure
Crystal lattice structure•+/- ion attract/arrange orderly manner•arrangement in 3D shape•strong electrostatic forces attraction bet opposite charged ions
2.8. 82.8
F-

Cations/Metals/+ve ions
Gp1 Gp 2 Gp3 Transition metals ions ( variable oxidation states)
Oxidation state+1
Oxidation
state+2
Oxidation
state+3
Sc+3
Ti+2+3
V+2+3
Cr+2+3+6
Mn+2+3+6+7
Fe+2+3
Co+2+3
Ni+2
Cu+1+2
Zn+2
Li 1+ Be2+ Sc 3+ Ti 2+
Ti 3+
V 2+
V 3+
Cr 2+
C r3+
Cr6+
Mn 2+
Mn 3+
Mn 6+
Mn 7+
Fe 2+
Fe 3+
Co2+ Ni2+ Cu1+
Cu2+
Zn2+
Na 1+ Mg2+ Al 3+
K 1+ Ca2+
Anion/Non metal
Gp 5 Gp 6 Gp 7
Oxidation
state
Oxidation
state
Oxidation
state
-3 -2 -1
N3- O2- F-1
P3- S2- CI-1
Br-1
I-1
Metal/Cations/+ve ion
Non Metal/Anions/-ve ion
Writing Chemical Formula Ionic Compounds

Cations/Metals/+ve ions
Gp1 Gp 2 Gp3 Transition metals ions ( variable oxidation states)
Oxidation state+1
Oxidation
state+2
Oxidation
state+3
Sc+3
Ti+2+3
V+2+3
Cr+2+3+6
Mn+2+3+6+7
Fe+2+3
Co+2+3
Ni+2
Cu+1+2
Zn+2
Li 1+ Be2+ Sc 3+ Ti 2+
Ti 3+
V 2+
V 3+
Cr 2+
C r3+
Cr6+
Mn 2+
Mn 3+
Mn 6+
Mn 7+
Fe 2+
Fe 3+
Co2+ Ni2+ Cu1+
Cu2+
Zn2+
Na 1+ Mg2+ Al 3+
K 1+ Ca2+
Anion/Non metal
Gp 5 Gp 6 Gp 7
Oxidation
state
Oxidation
state
Oxidation
state
-3 -2 -1
N3- O2- F-1
P3- S2- CI-1
Br-1
I-1
Metal/Cations/+ve ion
Non Metal/Anions/-ve ion
Ionic Compound
Li2O MgCI2 Al2O3 FeO NiO CuO
Li3N Mg3N2 AlN Fe3N2 Ni3N2 Cu3N2
Writing Chemical Formula Ionic Compounds

Cations/Metals/+ve ions
Gp1 Gp 2 Gp3 Transition metals ions ( variable oxidation states)
Oxidation state+1
Oxidation
state+2
Oxidation
state+3
Sc+3
Ti+2+3
V+2+3
Cr+2+3+6
Mn+2+3+6+7
Fe+2+3
Co+2+3
Ni+2
Cu+1+2
Zn+2
Li 1+ Be2+ Sc 3+ Ti 2+
Ti 3+
V 2+
V 3+
Cr 2+
C r3+
Cr6+
Mn 2+
Mn 3+
Mn 6+
Mn 7+
Fe 2+
Fe 3+
Co2+ Ni2+ Cu1+
Cu2+
Zn2+
Na 1+ Mg2+ Al 3+
K 1+ Ca2+
Anion/Non metal
Gp 5 Gp 6 Gp 7
Oxidation
state
Oxidation
state
Oxidation
state
-3 -2 -1
N3- O2- F-1
P3- S2- CI-1
Br-1
I-1
Metal/Cations/+ve ion
Non Metal/Anions/-ve ion
Ionic Compound
Li2O MgCI2 Al2O3 FeO NiO CuO
Li3N Mg3N2 AlN Fe3N2 Ni3N2 Cu3N2
Oxidation state/Charge ion→ Li1+ O2-
Formula compound Li2
O1
Video on polyatomic ions
Writing Chemical Formula Ionic Compounds
Step 1 : Write Oxidation state/charge
Step 2 : Balance it, (electrically neutral) by cross multiply – as subscript

Polyatomic ions
Group of non-metals bonded together
Oxidation state
Oxidation state
Oxidation state
-1/+1 -2 -3
(OH)-1
Hydroxide(SO4)
2-
Sulphate(PO4)
3-
Phosphate
(CN)-1
Cyanide(SO3)
2-
Sulphite
(SCN)-1
Thiocyanate
(CO3)2-
Carbonate
(NO3)-1
Nitrate(S2O3)
2-
Thiosulphate
(NO2)-1
Nitrite(Cr2O7)
2-
Dichromate
(NH4)+1
Ammonium
Polyatomic ionsMetal/Cations/+ve ions
Cations/Metals/+ve ions
Gp1 Gp 2 Gp3 Transition metals ions ( variable oxidation states)
Oxidation state+1
Oxidation
state+2
Oxidation
state+3
Sc+3
Ti+2+3
V+2+3
Cr+2+3+6
Mn+2+3+6+7
Fe+2+3
Co+2+3
Ni+2
Cu+1+2
Li 1+ Be2+ Sc 3+ Ti 2+
Ti 3+
V 2+
V 3+
Cr 2+
C r3+
Cr6+
Mn 2+
Mn 3+
Mn 6+
Mn 7+
Fe 2+
Fe 3+
Co2+ Ni2+ Cu1+
Cu2+
Na 1+ Mg2+ Al 3+
K 1+ Ca2+
Writing Chemical Formula Ionic Compounds

Polyatomic ions
Group of non-metals bonded together
Oxidation state
Oxidation state
Oxidation state
-1/+1 -2 -3
(OH)-1
Hydroxide(SO4)
2-
Sulphate(PO4)
3-
Phosphate
(CN)-1
Cyanide(SO3)
2-
Sulphite
(SCN)-1
Thiocyanate
(CO3)2-
Carbonate
(NO3)-1
Nitrate(S2O3)
2-
Thiosulphate
(NO2)-1
Nitrite(Cr2O7)
2-
Dichromate
(NH4)+1
Ammonium
Polyatomic ions
Li2(CO3) Mg(CO3) Al2(CO3)3 Fe(CO3) Ni(CO3) Cu(CO3)
Li(OH) Mg(OH)2 Al(OH)3 Fe(OH)2 Ni(OH)2 Cu(OH)2
Li2(SO4) Mg(SO4) Al2(SO4)3 FeSO4 Ni(SO4) Cu(SO4)
Ionic Compound
Metal/Cations/+ve ions
Cations/Metals/+ve ions
Gp1 Gp 2 Gp3 Transition metals ions ( variable oxidation states)
Oxidation state+1
Oxidation
state+2
Oxidation
state+3
Sc+3
Ti+2+3
V+2+3
Cr+2+3+6
Mn+2+3+6+7
Fe+2+3
Co+2+3
Ni+2
Cu+1+2
Li 1+ Be2+ Sc 3+ Ti 2+
Ti 3+
V 2+
V 3+
Cr 2+
C r3+
Cr6+
Mn 2+
Mn 3+
Mn 6+
Mn 7+
Fe 2+
Fe 3+
Co2+ Ni2+ Cu1+
Cu2+
Na 1+ Mg2+ Al 3+
K 1+ Ca2+
Writing Chemical Formula Ionic Compounds

Polyatomic ions
Group of non-metals bonded together
Oxidation state
Oxidation state
Oxidation state
-1/+1 -2 -3
(OH)-1
Hydroxide(SO4)
2-
Sulphate(PO4)
3-
Phosphate
(CN)-1
Cyanide(SO3)
2-
Sulphite
(SCN)-1
Thiocyanate
(CO3)2-
Carbonate
(NO3)-1
Nitrate(S2O3)
2-
Thiosulphate
(NO2)-1
Nitrite(Cr2O7)
2-
Dichromate
(NH4)+1
Ammonium
Polyatomic ions
Li2(CO3) Mg(CO3) Al2(CO3)3 Fe(CO3) Ni(CO3) Cu(CO3)
Li(OH) Mg(OH)2 Al(OH)3 Fe(OH)2 Ni(OH)2 Cu(OH)2
Li2(SO4) Mg(SO4) Al2(SO4)3 FeSO4 Ni(SO4) Cu(SO4) Video on polyatomic ions
Ionic Compound
Metal/Cations/+ve ions
Cations/Metals/+ve ions
Gp1 Gp 2 Gp3 Transition metals ions ( variable oxidation states)
Oxidation state+1
Oxidation
state+2
Oxidation
state+3
Sc+3
Ti+2+3
V+2+3
Cr+2+3+6
Mn+2+3+6+7
Fe+2+3
Co+2+3
Ni+2
Cu+1+2
Li 1+ Be2+ Sc 3+ Ti 2+
Ti 3+
V 2+
V 3+
Cr 2+
C r3+
Cr6+
Mn 2+
Mn 3+
Mn 6+
Mn 7+
Fe 2+
Fe 3+
Co2+ Ni2+ Cu1+
Cu2+
Na 1+ Mg2+ Al 3+
K 1+ Ca2+
Oxidation state/Charge ion → Li1+ (CO3)
2-
Formula compound Li2
(CO3)1
Step 1 : Write Oxidation state/charge ion
Step 2 : Balance it, (electrically neutral) by cross multiply – as subscript
Writing Chemical Formula Ionic Compounds

Acids AlkaliMetal Hydroxide
Metal oxides Salts Gas
HCIHydrochloric acid
KOHPotassium hydroxide
CuOCopper(II) oxide
CaCO3
Calcium carbonate
COCarbon monoxide
HNO3
Nitric acidNaOH
Sodium hydroxideMgO
Magnesium oxideNa2CO3
Sodium carbonate
CO2
Carbon dioxide
H2SO3
Sulphurous acidCa(OH)2
Calcium HydroxideZnO
Zinc oxideNaHCO3
Sodium bicarbonate
SO2
Sulphur dioxide
HCOOHMethanoic acid
NH3
AmmoniaNa2O
Sodium oxideKNO3
Potassium nitrateSO3
Sulphur trioxide
CH3COOHEthanoic acid
Mg(OH)2
Magnesium hydroxide
Al2O3
Aluminium oxidePb(NO3)2
Lead (II) NitrateNO2
Nitrogen dioxide
H3PO4
Phosphoric acidCu(OH)2
Copper (II) hydroxide
Fe2O3
Iron(III) oxideNaNO3
Sodium nitrateCH4
Methane
H2CO3
Carbonic acidAl(OH)3
Aluminium hydroxide
K2SPotassium sulphide
PbI2
Lead (II) nitrateH2S
Hydrogen sulphide
HNO2
Nitrous acidFe(OH)2
Iron (II) hydroxidePbS
Lead(II) sulphideAgCI
Silver chlorideO2
Oxygen
HFHydrofluoric acid
Fe(OH)3
Iron (III) hydroxideZnS
Zinc sulphideMgSO4
Magnesium sulphate
N2
Nitrogen
HCIOHypochlorous acid
Zn(OH)2
Zinc hydroxideAI2S3
Aluminium sulphide
Na2S2O3
Sodium thiosulphate
CI2
Chlorine
Chemical Formula for common chemicals
Naming chemical compound
Writing chemical formula
Writing chemical formula
VIDEO TUTORIALS

brown liquid
yellow gas
greenish gas
violet solid
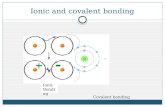
Covalent bonding between non metals
Gp 17 Non metalGp 17 Non metal
Non metal •High EN value•Gain electron (anion)•Electronegative ions
Covalent Bond Group 17
Non metal •High EN value•Gain electron (anion)•Electronegative ions

brown liquid
yellow gas
greenish gas
violet solid
Covalent bonding between non metals
2.8.7
Gp 17 Non metal
achieve stable octet structure
CI shared pair electron
Covalent BondingElectrostatic forces attraction between nucleus with shared pair electron
2.8.8
2.8.7 Sharing electron
Gp 17 Non metal
2.8.8
CI
Non metal •High EN value•Gain electron (anion)•Electronegative ions
Covalent Bond Group 17
CICI
Non metal •High EN value•Gain electron (anion)•Electronegative ions

brown liquid
Click here simulation on covalent bond
yellow gas
greenish gas
violet solid
Covalent bonding between non metals
2.8.7
Gp 17 Non metal
achieve stable octet structure
CI shared pair electron
Covalent BondingElectrostatic forces attraction between nucleus with shared pair electron
2.8.8
2.8.7 Sharing electron
Gp 17 Non metal
2.8.8
CI
Non metal •High EN value•Gain electron (anion)•Electronegative ions
Covalent Bond Group 17
CICI
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx:
:
:.
xx
X
xx
xx CI CI
:: xx
xx
CI CI
Non metal •High EN value•Gain electron (anion)•Electronegative ions
Single covalent bond – shared pair electron

Covalent bonding between non metals
Covalent BondingSingle covalent bond ONE pair shared e
2.8.8 2.8.8
Covalent Bond
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:.
xx
X
xx
xx CI CI
:: xx
xx
CI CI
Sharing electron
Bond Strength

Covalent bonding between non metals
Covalent BondingSingle covalent bond ONE pair shared e
2.8.8 2.8.8
Covalent Bond
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:.
xx
X
xx
xx
O
CI CI:
: xxxx
CI CI
Sharing electron
Bond Strength
2.8 2.8
Sharing electron
:O O: : x
x
xx
xx
O:
: xx
xx
O O
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Covalent BondingDouble covalent bond TWO pair shared e

Covalent bonding between non metals
Covalent BondingSingle covalent bond ONE pair shared e
2.8.8 2.8.8
Covalent Bond
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:
:.
xx
X
X
xx
xx
xx
O
CI CI:
: xxxx
xx
CI CI
Sharing electron
Bond Strength
2.8 2.8
Sharing electron
:O O: : x
x
xx
xx
O:
: xx
xx
N
O O
N:
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
C
: :. N N: N N
:
Covalent BondingDouble covalent bond TWO pair shared e
Covalent BondingTriple covalent bondTHREE pair shared e
O:
: xx: CO:
:
C :O:
: O
:
O O
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Covalent BondingDouble covalent bondTWO pair shared e

Bond length and Bond strength
Bond length = 0.199nm
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:
:.
xx
X
X
xx
xx
O
CI CI:
: xxxx
xx
CI CI
:O O: : x
x
xx
xx
O:
: xx
xx
N
O O
N:
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
: :.N N: N N
Bond length = 0.121nm
Bond length = 0.110nm

Bond Bond order
Bond strength
Bond length/pm
C - C 1 347 154
C = C 2 612 134
C Ξ C 3 820 120
N - N 1 159 145
N = N 2 418 123
N Ξ N 3 914 110
Bond length and Bond strength
Bond length = 0.199nm
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:
:.
xx
X
X
xx
xx
O
CI CI:
: xxxx
xx
CI CI
:O O: : x
x
xx
xx
O:
: xx
xx
N
O O
N:
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
: :.N N: N N
Triple bond > Double bond > Single bond
Bond length decrease
Bond strength Increase
(Double/Triple bond)
Bond length = 0.121nm
Bond length = 0.110nm
Bond order up – Bond strength up – Bond length down

Bond Bond order
Bond strength
Bond length/pm
C - C 1 347 154
C = C 2 612 134
C Ξ C 3 820 120
N - N 1 159 145
N = N 2 418 123
N Ξ N 3 914 110
Bond length and Bond strength
Bond length = 0.199nm
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:
:.
xx
X
X
xx
xx
O
CI CI:
: xxxx
xx
CI CI
:O O: : x
x
xx
xx
O:
: xx
xx
N
O O
N:
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
O
: :.N N: N N
:Triple bond > Double bond > Single
bond
Bonding pair e -involve in
bonding
Bond length decrease
Bond strength Increase
(Double/Triple bond)
Bond length = 0.121nm
Bond length = 0.110nm
Bond order up – Bond strength up – Bond length down
O
:
Non bonding pair (Lone pair electron)
Bonding pair electron
C O: :
Bonding pair electron
Dative bond (electron pair of oxy)
Types of bonding
Lone pair e –not involve in bonding
Dative/coordinate bond
- pair e come from an atom
: :

:
.CI
..x
Bond Bond order
Bond strength
Bond length/pm
C - C 1 347 154
C = C 2 612 134
C Ξ C 3 820 120
N - N 1 159 145
N = N 2 418 123
N Ξ N 3 914 110
Bond length and Bond strength
Bond length = 0.199nm
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
CI CI: xx
::
:
:.
xx
X
X
xx
xx
O
CI CI:
: xxxx
xx
CI CI
:O O: : x
x
xx
xx
O:
: xx
xx
N
O O
N:
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
Lewis structure/diagram . Electron cross dot . Valence/bonding pair electron
O
: :.N N: N N
:Triple bond > Double bond > Single
bond
Bonding pair e -involve in
bonding
Bond length decrease
Bond strength Increase
(Double/Triple bond)
Bond length = 0.121nm
Bond length = 0.110nm
Bond order up – Bond strength up – Bond length down
O
:
Non bonding pair (Lone pair electron)
Bonding pair electron
C O: :
Bonding pair electron
Dative bond (electron pair of oxy)
Types of bonding
Lone pair e –not involve in bonding
Dative/coordinate bond
- pair e come from an atom
Exception to octet rule
All period 2 element - observe octet rule except Be and B Electron deficientLess than 8 valence e
Expanded octetMore than 8 valence e
All period 3 element - observe octet rule except P and S
: BeCI CIx. :
::
::
x
x.
Be - 4 valence e
BCI CI: :::
:: x
: :B - 6 valence e
P S
CI
CI
CI
CI
CI
CI
CICI
CICICI
P - 10 valence e
S – 12 valence e
: :

O:
ONO C
N
C BeH N X x:Linear
O C H H NO N O
C OO CH N HH Be C N N O O:xx
:::
X x
X x
XX X X
X
X
X x
:
X x
:
:
+
+: :
:
:.
Electron deficient Be
Dative bond from N
NCxx :
Electron deficient N(7 valence electron)
CO2 HCN BeH2
CN - NO2 + NO
::

O:
ONO C
N
C BeH N
:
B
:
X x:
:
:
C SSO
Linear
O C H H NO N O
Trigonal
planar
C OO CH N HH Be C N N O O:xx
:::
X x
X x
XX X X
X
X
X x
:
X x
:
:
+
+: :
:
:.
F
F
F
Electron deficient Be
Dative bond from N
NCxx :
Electron deficient N(7 valence electron)
H
H O O
O O
O O
CO O
O
O N
O
O
BXF
X
X F
X
FC
H
H
X
x x
O
:
S
x
x
O OS
O:
xx
xx
O:::
: :
x x
O:
CO:
: :
X
O:::
X
x x
O :
: N
2- -
2-
O:
: :
X
x x
xx
O
:
: :
O:
:
-
Electron deficient B
O
x x
S
O
: : O: : ::
::
Dative bond from S
:
:
ON
O
O
:
:
: :
: :
-
O
x
x
x x
: :
::
||||
|| ||
Sulphur expanded octet (12 valence electron) ✓
BF3 CH2 O SO3 CO3 2- NO3
-
CO2 HCN BeH2
CN - NO2 + NO
::

:
O
HH
H2O
H H
O: :
O
F2O
F F
F F
O
S
CICI
SCI2 SH2
S
H H
S
CI CI
S
H
: : :: : :
NH2 -
N
HH
HH
N: :
-
-. .
.. . .. . . .
.. . . ... .
.. . . . . .H: : : : :
: : : :: :
All molecule – octet rule
Bend

:
O
O O
O
O O
||
::
:
:
::
: ::
N
:
:
:
O O
||
O
NO
:: :
:
:
: ::
SO O
||
OOS :::
:
SO O
||||
OOS ::
:: :::
:
:: ::
N
O O
||
N ::
OO
::
:::
:
-
-
O ON
::
:
::
:
:
O O:
::N
::
::
:
-
Dative bond from Nelectron deficient N(7 valence electron)
SO O
::
:
:
:
:::
Sulphur expanded octet (10 valence electron)
or
or
Dative bond from S
NO2- NO2 SO2 O3
O
HH
H2O
H H
O: :
O
F2O
F F
F F
O
S
CICI
SCI2 SH2
S
H H
S
CI CI
S
H
: : :: : :
NH2 -
N
HH
HH
N: :
-
-. .
.. . .. . . .
.. . . ... .
.. . . . . .H: : : : :
: : : :: :
All molecule – octet rule
Bend
Bend
:O::
O
:
::
O
:
:Dative bond from O

||
:CI
C
HH
CH4 NH4+
N
BH4- PCI4
+ POCI3
P
CI-+
Tetrahedral
H
H
H
HH
H
B
H
HHH CI
CICI
+
P
O
CICICI
C
H
H
HH
H
N
H
H
H
+
H
B H
H
H
-CI
PCI
CI
CI
+
:
:
:
:
:
:::
: :
:
:
O
::
P
CI
CI ::
: ::
:
::
: :

||
:
S
CI
Sulphur expanded octet (12 valence electron)
C
HH
CH4 NH4+
N
BH4- PCI4
+ POCI3
P
CI-+
Tetrahedral
H
H
H
HH
H
B
H
HHH CI
CICI
+
P
O
CICICI
C
H
H
HH
H
N
H
H
H
+
H
B H
H
H
-CI
PCI
CI
CI
+
:
:
:
:
:
:::
: :
:
:
O
::
P
CI
CI ::
: ::
:
::
: :
OSO4
2-
O
|| ||
OO
O
S
O
::
O
::
||
O
H2SO4 H3PO4
CIO4 -
||O
S ||O
OHHO
S
O
O
::
::
O
O:
O||
POH
OHHO
O
::O
P
OO
||O
CIO
||
OO
O
CI
::
:: O
::
O
O
: :
:
:
: :
:
:
:
::
:
::
: H : :
H
: :
:
: :
H HH
:
:::::
-
-
Phosphorus expanded octet (10 valence electron)
:: :
:
:
:
Chlorine expanded octet (14 valence electron)
Tetrahedral
2-
2-

:
O
HH
H2O
H H
O: :
O
F2O
F F
F F
O
S
CICI
SCI2 SH2
S
H H
S
CI CI
S
H
: : :: : :
NH2 -
N
HH
HH
N: :
-
-. .
.. . .. . . .
.. . . ... .
.. . . . . .H: : : : :
: : : :: :
Two lone pair central atom
Bend
Two lone pair

N
H
N
:
:
O
SO32- PH3 NH3
O
HH
H2O
H H
O: :
O
F2O
F F
F F
O
S
CICI
SCI2 SH2
S
H H
S
CI CI
S
H
: : :: : :
NH2 -
N
HH
HH
N: :
-
-. .
.. . .. . . .
.. . . ... .
.. . . . . .H: : : : :
: : : :: :
Two lone pair central atom
Bend
CIO3-
HH
PH
HH
H. .
HH
PH
H. .
H
:
O O
S||
O
SO
:
::
O
2-
2-
CIO
OO
CI
:
OOO
-
:
:
:
: : : :
: :
:
:
: : :
:
::
-
SOCI2
SCI
CIO||
S:::
OCI
CI
:
: :
::
::
:
One lone pair central atom
One lone pair
Pyrimidal
Two lone pair

N
H
N
:
:
O
SO32- PH3 NH3
O
HH
H2O
H H
O: :
O
F2O
F F
F F
O
S
CICI
SCI2 SH2
S
H H
S
CI CI
S
H
: : :: : :
NH2 -
N
HH
HH
N: :
-
-. .
.. . .. . . .
.. . . ... .
.. . . . . .H: : : : :
: : : :: :
Two lone pair central atom
Bend
CIO3-
HH
PH
HH
H. .
HH
PH
H. .
H
:
O O
S||
O
SO
:
::
O
2-
2-
CIO
OO
CI
:
OOO
-
:
:
:
: : : :
: :
:
:
: : :
:
::
-
SOCI2
SCI
CIO||
S:::
OCI
CI
:
: :
::
::
:
One lone pair central atom
One lone pair
Pyrimidal
Two lone pair
CH4
C
H
NH4+ BH4
- PCI4+ POCI3
HH
H
H
CH
HH
N
H
H HH
H
NH
HH
H
BHH H
H
BH
HH
CI
CI CI CI
P
CI
CICI
P
CI
O-
-
+
+
+
+
P
CI CICI
||
CI CICI
O
P
::
Tetrahedral
NO lone pair
:
::
:
::
::
:
:
:
::
:
::
::: :
::
:
NO lone pair central atom

P CICI
CICI
CI
P
CI
CI
CI
CI
CI
NO lone pair
NO lone pair central atom
PCI5
Trigonal bipyrimid
al
Central atom P- expanded octet - 10 valence electron

P CICI
CICI
CI
:
P
CI
CI
CI
CI
Seesawstructur
e
S
F
F
F
F
S
F
F
FF
Te
CI
CI
CI
CI
Te
CI
CI
CICI
:
I
FF
F
F
F
I
F
F
F
Xe
F
F
O
O
Xe
F
F
O ::
O
:
+
+
:
CI
:::::::
:
:
::
::
:::
:
::
::
:: :
::
:::
:
::
:
:
: :
:
:
:
::
:
::
::
:
:
NO lone pair
NO lone pair central atom
ONE lone pair
:ONE lone pair central atom
PCI5
SF4 TeCI4 (IF4)+ XeO2F2
Trigonal bipyrimid
al
Central atom S, Te, I and Xe - expanded octet - 10 valence electron
Central atom P- expanded octet - 10 valence electron

P CICI
CICI
CI
:
P
CI
CI
CI
CI
Seesawstructur
e
Tstructur
e
S
F
F
F
F
S
F
F
FF
Te
CI
CI
CI
CI
Te
CI
CI
CICI
:
CI
F
F
F
CI
F
F
F
I
CI
CI
CI
CI
I
CI CI
I
FF
F
F
F
I
F
F
F
Xe
F
F
O
O
Xe
F
F
O ::
O
:
+
+
:
CI
:::::::
:
:
::
::
:::
:
::
::
:: :
::
:::
:
::
:
:
: :
:
:
:
::
:
::
::
:
:
::
:
::
:
::
::
:
::
:
::
:
:
::
:
:
NO lone pair
NO lone pair central atom
ONE lone pair
:ONE lone pair central atom
TWO lone pair
::
TWO lone pair central atom
BrF
F
F
Br
F
F
F
: :
:
: :
:
::
:
:
:
PCI5
SF4 TeCI4 (IF4)+ XeO2F2
CIF3
ICI3 BrF3
Trigonal bipyrimid
al
Central atom S, Te, I and Xe - expanded octet - 10 valence electron
Central atom P- expanded octet - 10 valence electron
Central atom CI, I and Br - expanded octet - 10 valence electron

P CICI
CICI
CI
:
P
CI
CI
CI
CI
Seesawstructur
e
Tstructur
e
S
F
F
F
F
S
F
F
FF
Te
CI
CI
CI
CI
Te
CI
CI
CICI
:
CI
F
F
F
CI
F
F
F
I
CI
CI
CI
CI
I
CI CI
I
FF
F
F
F
I
F
F
F
Xe
F
F
O
O
Xe
F
F
O ::
O
:
+
+
:
CI
Linearstructur
e
I
I
I
Xe
CI
CI
I
F
F
:::::::
:
:
::
::
:::
:
::
::
:: :
::
:::
:
::
:
:
: :
:
:
:
::
:
::
::
:
:
::
:
::
:
::
::
:
::
:
::
:
::
::
::
::
::
::
::
::
::
:
::
:
:
: : :: : :: : :
NO lone pair
NO lone pair central atom
ONE lone pair
:ONE lone pair central atom
TWO lone pair
::
TWO lone pair central atom
THREE lone pair
THREE lone pair central atom
:::
BrF
F
F
Br
F
F
F
: :
:
: :
:
::
:
:
:
-
PCI5
SF4 TeCI4 (IF4)+ XeO2F2
CIF3
ICI3 BrF3
(I3)- (ICI2)- XeF2
Trigonal bipyrimid
al
Central atom S, Te, I and Xe - expanded octet - 10 valence electron
Central atom P- expanded octet - 10 valence electron
Central atom CI, I and Br - expanded octet - 10 valence electron
Central atom I and Xe - expanded octet - 10 valence electron
-

Octahedral
F
S
SF6
F
FF
F
F
PCI6-
P
CICI
CI
CI
CI
CI
IF5O
I
O||
F
F
F
F
F
F
SF
FF
F
FCI
PCI
CI
CI
CI
CII
F F
F
F
F ::
O
NO lone pair
:
:
:::
:
::: :
::
:
::
:
::
::
::
::
:
:
:
::
:
:
:
:::
: ::::
:
:
:
::
:
::
:
::
::
NO lone pair central atom
Central atom S, P and I- expanded octet - 12 valence electron
-
-

Octahedral
F
S
SF6
F
FF
F
F
PCI6-
P
CICI
CI
CI
CI
CI
IF5O
I
O||
F
F
F
F
F
F
SF
FF
F
FCI
PCI
CI
CI
CI
CII
F F
F
F
F ::
O
NO lone pair
Square pyrimidal
CI
Sb
Sb
CI
CI
CICI
CI
CI
CICI
CI
(SbCI5)2- BrF5
F
BrF
FF
F
F
Br
F
F
O
F
F
Xe||
F
FF
F
O
::
Xe
F
F
F
F
F
Te F
FF
F
F
Te F
F
F
F
XeOF4 (TeF5)-
-
-: : : :
:
ONE lone pair
:
:
:::
:
::: :
::
:
::
:
::
::
::
:
:
:
:
:
:
:
::
:
:
:
:::
: ::::
:
:
:
::
:
::
:
::
::
:
::
::
::
::
:
:
:
::::
:
::
:
:
::
::
::
::
:
:
::
:
::
::
:::
:
:
:
:
:
:
::
::
:
::
::
:
2-
2-
NO lone pair central atom
Central atom S, P and I- expanded octet - 12 valence electron
ONE lone pair central atom
Central atom Sb, Br, Xe, Te- expanded octet - 12 valence electron
-
-

Octahedral
F
S
SF6
F
FF
F
F
PCI6-
P
CICI
CI
CI
CI
CI
IF5O
I
O||
F
F
F
F
F
F
SF
FF
F
FCI
PCI
CI
CI
CI
CII
F F
F
F
F ::
O
NO lone pair
Square pyrimidal
CI
Sb
Sb
CI
CI
CICI
CI
CI
CICI
CI
(SbCI5)2- BrF5
XeF
FF
F
Xe
F
XeF4
F
F
F
F
BrF
FF
F
F
Br
F
F
O
F
F
Xe||
F
FF
F
O
::
Xe
F
F
F
F
F
Te F
FF
F
F
Te F
F
F
F
XeOF4 (TeF5)-
-
-
CICII
CICI
(ICI4)--
ICICI
CI
CI
-Square planar
: : : :
::
:
ONE lone pair
TWO lone pair
:
:
:::
:
::: :
::
:
::
:
::
::
::
:
:
:
:
:
:
:
::
:
:
:
:::
: ::::
:
:
:
::
:
::
:
::
::
:
::
::
::
::
:
:
:
::::
:
::
:
:
::
::
::
::
:
:
::
:
::
::
:::
:
:
:
:
:
:
::
::
:
::
::
:
2-
2-
::
::
::
::
:
:
:
:
::
::
::
::
::
::
::
::
NO lone pair central atom
Central atom S, P and I- expanded octet - 12 valence electron
ONE lone pair central atom
Central atom Sb, Br, Xe, Te- expanded octet - 12 valence electron
TWO lone pair central atom
Central atom Xe and I - expanded octet - 12 valence electron
-
-

Chemical Bonds
Achieve stable electron arrangement of noble gases
Ionic Bonds
Transfer of electron from metal to non metal
Metal donate e Non Metal accept e
Positive ion (cation)
Negative ion (anion)
Ionic compound
Solubility
Covalent Bonds
Sharing of electron bet non metal atoms
Covalent compound/molecules
Non Metal share electrons
Difference in physical property
m/p + b/pconductivityVolatility
+ -electrostatic forces attraction
Volatility
Solubility Conductivity
M/p B/p
Ionic compoun
d
Low Soluble in aqueoussolvent
High (In aqueous)
High
Covalent compoun
d
High Soluble in organicsolvent
Low Low
0 0.4 4
Difference in electronegativity
difference < 0.4covalent compound
difference > 2ionic compound
2
CI-Na+
EN - 0.9 EN - 3.0
Diff = 3 – 0.9 = 2.1
C2.5
H2.1
EN – 2.5 EN – 2.1
Diff = 2.5 – 2.1 = 0.4

Acknowledgements
Thanks to source of pictures and video used in this presentation
Thanks to Creative Commons for excellent contribution on licenseshttp://creativecommons.org/licenses/
Prepared by Lawrence Kok
Check out more video tutorials from my site and hope you enjoy this tutorialhttp://lawrencekok.blogspot.com