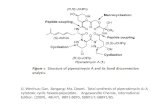
Hydrogenolysis of Sorbitol over Ni and Pt loaded on NaY M.Banu 4 th Annual Day 01.08.2010.
-
Upload
ruth-matthews -
Category
Documents
-
view
232 -
download
0
Transcript of Hydrogenolysis of Sorbitol over Ni and Pt loaded on NaY M.Banu 4 th Annual Day 01.08.2010.

Hydrogenolysis of Sorbitol over Ni and Pt loaded on NaY
M.Banu4thAnnual Day
01.08.2010

Cellulosic biomass is abundant on earth, and is an ideal feedstock for
the production of bio-based chemicals. In a simple processing scheme,
cellulose is hydrolyzed into simple sugars, then hydrogenated to the
sugar alcohols that are further transformed into valuable chemicals.
Introduction
CelluloseCellulose
glucoseglucose
SorbitolSorbitol
PG, EG Antifreeze
PG, EG Antifreeze
Glycerol Plastics
Glycerol Plastics
Lactic acidBiodegradable
plastics
Lactic acidBiodegradable
plastics
L-SorboseVitamins
L-SorboseVitamins
BioethanolBioethanol

• Hydrogenolysis of sorbitol (obtained from glucose) can be
used to produce valuable chemicals like ethylene glycol,
which are generally produced by multi-step
transformations from non-renewable petroleum based
feed-stocks.
• The transformation of carbohydrates from renewable
sources into value added chemicals has assumed
significance at present owing to the decreasing petroleum
reserves and increasing carbon accumulation in the
atmosphere.
• We report our studies on the use of Ni and Pt loaded on
NaY as catalysts for the hydrogenolysis of sorbitol.
Continued...

ExperimentalPreparation of catalysts
Wet impregnation Ion exchange
CharacterizationX-ray diffractionTextural analysis
Results and discussions
Effect of Ni(2, 4, 6 wt%) loading on NaYEffect of Ni(2, 4, 6 wt%) and Pt (1 wt%) loading on NaYEffect of catalyst amountEffect of TemperatureGlycerol hydrogenolysis over Ni and Pt-NaYEffect of Ca(OH)2
Conclusions
Contents of the presentation

Experimental

Catalysts prepared by wet impregnation method
2 wt%Ni/NaY4 wt%Ni/NaY6 wt%Ni/NaY
Catalysts prepared by ion exchange method
8 wt%Ni/NaY1 wt%Pt/NaY
Catalyst Preparation

1wt% of Pt2+(0.025M Pt(NH3)4Cl2+1.98g NaY Zeolite+25 ml water
Refluxed at 80 °C/12h.
2, 4, 6 % Ni from Ni(NO)3.6H2O
Filtered and washed with water
Dried at 110 °C/12 h
Ni(2, 4, 6 wt%)- Pt(1 wt%)/NaY Zeolite Catalyst
Calcined at 400 °C / 4h
Dried at 110 °C/12 h
Catalyst Preparation
Calcined at 400 °C / 4h
Reduced at 400 °C with H2 Flow/
3h

8 wt% of Ni2+(0.05M of Ni(NO)3.6H2O)+
5g NaY Zeolite+100 ml water
Refluxed at 80°C/ 12h.
Reduced at 400 °C with H2
Flow/ 3h
Filtered and washed with water
Dried at 110 °C/12 h
8 wt%Ni/NaY Zeolite Catalyst
Catalyst Preparation ( Ion Exchange)
Calcined at 400 °C / 4h

X-ray diffraction patterns of NaY and Ni-NaY
Characterization

X-ray diffraction patterns of NaY and Ni-Pt-NaY
Characterization

Sample BET Surface area (m2/g)
NaY 548
6%Ni1%Pt/NaY 502
6%Ni/NaY 536
2%Ni1%Pt/NaY 482
2%Ni/NaY 536
Surface areas of samples
Characterization

Analytical procedures
Products analysed by GC and Sorbitol analysed by making Trimethylsilyl derivative
5 ml of 15wt% Sorbitol + 1.5 ml DMF(As internal Standard)
TMS Mixture(1ml Hexamethyldisilazane + 0.1ml trifluoroacetic acid)
TMS derivative of sorbitol
0.5 ml of Internal standard + 0.5 ml of TMS Mixture
Preparation of Trimethylsilyl Sorbitol Derivative

Results and Discussions

OH
OH
OH
OH
OH
HOOH
OH
HO
HO
OH
OH
OH
OH
OH
OH
HOOH
EG
OH
Methanol
OH
Ethanol
CH4
Methane
H2
Sorbitol
Xylitol
Glycerol 1,2-PD
HO OH
1,3-PD
(I)
(II) (III)
,
,,
(IV) (V)
(VI)
HO
OH
OH
OH
Erythritol
,
Important Products from Sorbitol Hydrogenolysis

Conditions: 15 % sorbitol in water; temperature: 220 °C; catalyst amount: 0.5g; pressure: 60 bar; time: 6 h; stirring speed: 300 rpm.
Effect of Ni loading on Ni-NaY catalysts

Effect of Ni loading on Pt(1 wt%)-NaY
Conditions: 15 % sorbitol in water; temperature: 220 °C; pressure: 60 bar; time: 6 h; catalyst amount: 0.5g; stirring speed: 300 rpm.

#
Catalyst
[amount,
g]
Sorbitol
conversi
on (%)
Product selectivity (%)
Glycer
ol
Dihydric
alcohols Monohydr
ic
alcoholsa
Polyol
sb
Others
c
1,2-PD EG
16%Ni-NaY
[0.2]24 12 53 10 5 16 4
26%Ni-NaY
[0.5]66 14 62 7 7 10 -
36%Ni-NaY
[1.0]68 15 60 7 7 11 -
15 % sorbitol in water; temperature: 220 °C; pressure: 60 bar; time: 6 h; stirring speed: 300 rpm; a: methanol and ethanol; b: Trihydric (except glycerol) and higher alcohols; c: light products (some unidentified).
Effect of catalyst amount

Conditions: 15 % sorbitol in water; catalyst: 6%Ni/NaY; catalyst amount: 0.5g; pressure: 60 bar; time: 6 h; stirring speed: 300 rpm.
Effect of Temperature

# Catalyst[amount, g]
Sorbitol conversion (%)
Selectivity (%)
Glycerol
Dihydric alcohols
Monohydric alcoholsa
Polyolsb
Othersc
1,2-PD
EG
1 NaY [0.5] 15 17 8 1 4 41 29
28%Ni-NaY (IE) [0.5]
54 8 20 9 14 33 16
36%Ni-NaY
[0.5]66 14 62 7 7 10 -
15 % sorbitol in water; temperature: 220 °C; pressure: 60 bar; time: 6 h; stirring speed: 300 rpm; a: methanol and ethanol; b: Trihydric (except glycerol) and higher alcohols; c: light products (some unidentified).
Effect of catalyst preparation method

X-ray diffraction
Powder XRD patterns of (a) NaY, (b) reduced 8 wt% Ni-NaY (ion-exchanged) and (c) reduced 6 wt% Ni-NaY (impregnated).

Temperature programmed reduction profile
H2 consumption of a) 6 wt% Ni/NaY 1.018 mmol/g ; b) 8 wt% Ni/NaY 0.254 mmol/g .Note: baseline correction was done to calculate H2 consumption for ‘b’

# Catalyst[amount, g]
Glycerol conversion (%)
Selectivity (%)
1,2-PD EG Methanol Ethanol
Others
31%Pt-NaY
[0.5]1 - - 100 - -
26%Ni-NaY
[0.5] 32 1 1 56 4 38
15 % sorbitol in water; temperature: 220 °C; pressure: 60 bar; time: 6 h; stirring speed: 300 rpm
Glycerol hydrogenolysis over Ni & Pt-NaY

NO Catalyst[amount, g]
Sorbitol conversion (%)
Selectivity (%)
Glycerol
Dihydric alchols
Monohydric alcholsa
Polyolsb
Othersc
1,2-PD
EG
16%Ni-NaY
[0.5]66 14 62 7 7 10 -
2
6%Ni-NaY [0.5]
+Ca(OH)2 [0.5]
75 4 69 7 5 8 7
31%Pt-NaY
[0.5]43 62 6 3 24 1 4
4
1%Pt-NaY [0.5] +Ca(OH)2
[0.5]
60 70 6 3 11 2 8
5Ca(OH)2
[0.5]24 3 9 2 23 45 18
15 % sorbitol in water; temperature: 220 °C; pressure: 60 bar; time: 6 h; stirring speed: 300 rpm; a: methanol and ethanol; b: Trihydric (except glycerol) and higher alcohols; c: light products (some unidentified).
Effect of Ca(OH)2

Conclusions

Conclusions
The hydrogenolysis of an aqueous solution of sorbitol takes place in the presence of H2 over Pt and Ni loaded on NaY.
1,2-PD is the major product; over the Ni-NaY, while glycerol is the major product over the Pt-NaY.
OHOH
OH
OH
OH
HO
OH
OH
HO
OH
OH
H2
Sorbitol
Glycerol
1,2-PD
Ni-NaYH2
Pt-NaY

The addition of Pt to the Ni catalysts does not influence their activity or selectivity significantly.
Ca(OH)2 promotes the performance of both Ni and Pt catalysts without affecting product selectivity.
The catalyst 6%Ni/NaY (with Ca(OH)2) exhibited high conversion (75%) and good selectivity towards 1,2-PD (69%) at 220 °C.
Continued…