How Water Quality in the Kabul River, Pakistan, Can be ... Water Quality in the Kabul River,...
Transcript of How Water Quality in the Kabul River, Pakistan, Can be ... Water Quality in the Kabul River,...

Advanced Studies in Biology, Vol. 8, 2016, no. 4, 151 - 171
HIKARI Ltd, www.m-hikari.com http://dx.doi.org/10.12988/asb.2016.6830
How Water Quality in the Kabul River,
Pakistan, Can be Determined with Algal
Bio-indication
Sophia Barinova1*, Izaz Khuram2, Asadullah2, Nadeem Ahmad2,
Samin Jan3 and Dong Hyun Shin4
1Institute of Evolution, University of Haifa, Mount Carmel
199 Abba Khoushi Ave., Haifa 3498838, Israel *Corresponding author
2Department of Botany, University of Peshawar, Khyber, Pakhtunkhwa, Pakistan
3Department of Botany, Islamia College Peshawar
Khyber, Pakhtunkhwa Pakistan
4School of Applied Biosciences, College of Agriculture and Life Sciences
Kyungpook National University, Daegu 702-701, Republic of Korea
Copyright © 2016 Sophia Barinova et al. This article is distributed under the Creative
Commons Attribution License, which permits unrestricted use, distribution, and reproduction in
any medium, provided the original work is properly cited.
Abstract
Altogether 209 species of algae and cyanobacteria has been revealed in
2014-2015 from four sites in the Kabul River that flow across the Peshawar
Valley. Green algae, diatoms, and Charophyta filamentous algae were diverse and
characterize regional feature in the river basin with high agricultural activity.
Species richness and algal abundance were increase down the river. Index
saprobity S varied between 1.55 and 1.59 (Class III of water quality) and reflects
decreasing of water quality down the river as a result of pollutants impact, which
brings mostly the Swat tributary. Bio-indication results show prevalence of
benthic or plankto-benthic species, temperate temperature indicators, slow
streaming middle oxygenated waters inhabitants, salinity-indifferents,
eurysaprobes, mesotrophes, and autotrophic algae with tolerance to middle concentration of Nitrates that reflects middle polluted waters III-IV Class of Water

152 Sophia Barinova et al.
Quality in the Kabul River. Our analysis shows that integral bio-indication in
water quality assessment with using of algal communities can give relevant results
of self-purification possibility, which can be employed in purpose of monitoring
the regional water quality as economy and resultative method.
Keywords: Periphyton, Phytoplankton, River, Water Quality, Bio-indication
1 Introduction
The ongoing climate change can be taken into account in addition to the
human factor. The global warming primarily influences the hydrological regime
of water ecosystems in arid regions [1] such as of Pakistan.
Algal diversity ecologically plays an important role in the understanding of
aquatic ecosystem, their productivity and water quality [2]. Habitat characteristics
play an important role to affect freshwater algal communities [3]. The study of
algae at their habitats is the prime object of algal ecology [4]. Rivers are complex
ecosystems consisting of biotic communities (biocoenoses) and diverse habitats
(biotopes). The characteristics of the habitats change from the head-waters to the
sites of eventual discharge [5].
It is known that the pollution of the river due to the number of people on its
banks and the intensity of development of the economy and have synergetic effect
with global warming. Because studies of the Kabul River in the early stages, there
is no published information on the anthropogenic load of the studied section of the
river, so we used only the impressions in the process of collecting.
The aquatic objects in Pakistan are represented mostly the rivers. Algal
diversity of the Pakistan rivers can help in ecological assessment of the water
quality and water using intensity for agriculture. The algal communities in
Pakistan rivers start to study in last two decades and is so far from exhaustive.
Algal flora of the Kunhar River, Pakistan, was ecologically studied for the first
time [6]. Algal Diversity of the Meenachil River, India was studied during the
premonsoon and monsoon period of 2014 [7]. Systematics and geographic
distribution of Chroococcus was studied in Pakistan [8]. Algal species diversity
distribution and its relationships to altitude of aquatic habitats were studied on the
Hindu Cush Mountains communities of the Swat River [9]. The importance of
water resources quality in Pakistan is now one of major problem. Therefore, the
state try to find some economy methods for dissolve this problem. One of the
methods for water resources quality assessment is the bio-indication. Algal
bio-indication approaches were employed in Pakistan earlier for the Swat River
algal communities [9]. The Swat River is the upper high mountain tributary of the
Kabul River. In the Kabul River were no more publications about algal diversity
than about freshwater green algal biofouling of boats [10].
The aim of present study was to reveal algal diversity in the Kabul River sites
before and after the Swat River input and employ the bio-indication methods for
the water quality assessment.

How water quality in the Kabul River, Pakistan… 153
Description of study site
Peshawar Valley is the most distinct region within the entire Khyber
Pakhtunkhwa, Pakistan. The Valley lies on the coordinates about 34°07’58’’ N
and 71°41’45’’E and has an altitude of about 335 m [11].
The climate of Peshawar Valley has been termed as a modified
Mediterranean type of climate. The summers are hot and the winters are cold. The
range of temperature varies between 50C° (highest) to -3C° (lowest). Four
seasons are recognized in Peshawar valley [11, 12].
The Kabul River is an itinerary between Afghanistan and Pakistan. It is 700
km long, of which 563.27 km is in Afghanistan and 136.79 km in Pakistan [13]. It
is rising in North Eastern Afghanistan at the base of the Wonay Pass in the
Paghman Range (Selsela-e-Kuh-e-Paghman) and ends in the Indus River near
Khairabad, North-West of Islamabad, Pakistan [12]. The depth and width of the
channel ratio is highly flexible in different seasons and vary in 2-3 m deep and
30-90 m width. The average discharge at Warsak Dam is 20.5×103m3sec−1. The
variations in river flow are the result of seasonal rains, glacial and snowmelt [14].
The major tributary is the Swat River, originating in Kalam travels in the hills
of Swat Valley and enters to the Kabul River near Jalabella. Two other tributaries
are the Bara River and the Jindai River, which flow into the Kabul River near
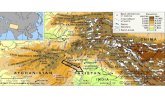
Khairabad [12]. Four research sites were selected in the Peshawar Valley (Figure
1) to assess the algal diversity, ecology, and water quality in the Kabul River:
Warsak (1), Sardaryab (2), Nowshera (3) and Khairabad (4).
Figure 1. Peshawar Valley with the Kabul River sampling sites
2 Materials and Methods
1. Field sampling
Sampling was carried out during the autumn and spring seasons in 2014-2015.
A total of 30 samples were collected from four sampling sites in a 100-meter
radius on each individual location from different habitats including phytoplankton
and periphyton (epiphyton, epilithon, and epipsammon). The samples were trans-

154 Sophia Barinova et al.
ported into the Phycology and Tissue Culture laboratory, Department of Botany,
University of Peshawar, in standard specimen bottles with icebox.
2. Preservation, Labeling and Storage
Temporary preservation was processed immediately after sampling with
neutral Lugol’s Iodine solution, 0.5 ml per 100 ml water sample, and stored for a
short time [15].
Permanent preservation of samples was in 3 % FAA (Formalin, acetic acid and
alcohol) and kept for a long time to avoid spoilage [15]. The permanently
preserved specimens were vouchered and submitted to the Department of Botany
herbarium, University of Peshawar.
3. Laboratory Processing
Microscopic morphology of the Non-Diatomaceous isolates was determined
by using the wet-mount staining method [15]. This was done by using a sterile
micromanipulator to pick up algal filaments from temporarily preserved samples
and placed onto a clean glass slide on which a drop of distilled water had been
added. A drop of lactophenol cotton blue stain was added, and the preparation was
covered with clean cover slips. The slides were subsequently viewed under 10×,
20×, 40× 60× and 100× Nikon Eclipse E200 microscope objectives. Images of the
taxa were taken with a BRESSER digital microscope. The Diatomaceous isolates
were cleaned by using peroxide (H2O2) technique [16] (Swift, 1967). The empty
frustules were then mounted and analyzed for their morphology. Standard
references [2,17-23] were usede for taxonomic identification.
Physicochemical properties of the water from the sampling sites were
measured in parallel with algal sampling. Temperature and water pH were
measured with HANNA HI98190 portable meter and Turbidity with HANNA
HI98703 meter in the laboratory. Electrical conductivity and Total Dissolved
Solids (TDS) of the water samples were measured with HANNA HI98192 meter
in the laboratory. Total Suspended Solids (TSS) values in the water samples were
measured with HACH TSS meter in the laboratory. Dissolved oxygen (DO) and
Biochemical Oxygen Demand (B.O.D.) were measured by using HANNA
HI98193 meter in the laboratory.
Alkalinity as CaCO3 was analyzed by titration against standard sulphuric acid.
Hardness as CaCO3 and Sulphates were analyzed by complexometric titrations
using EDTA (0.01M). Nitrites and Nitrates were analyzed by spectrophotometric
(Colorimetric) method. Ammonia and Phosphates (PO43-) were analyzed by
spectrophotometric method using Nessler’s reagent. Phosphates (PO43-) Chlorides
(Cl-) were determined by titrating against silver nitrate (0.1 N) using potassium
chromate as indicator.
Ecological preferences of revealed algal species were assessed according [24].
Bio-indication approaches were taken from these references and later [25].

How water quality in the Kabul River, Pakistan… 155
Indices saprobity S [26, 27] were calculated on the basis of identified species
for each community and quantitative investigations of phytoplankton as:
𝑆 = ∑ (𝑠𝑖 × 𝑎𝑖) ∑(𝑎𝑖)
𝑛
𝑖=1
⁄
𝑛
𝑖=1
(Eq. 1)
Where: S – Saprobity index of algal community; si – species–specific saprobity
index; ai – species abundance.
The integral index of aquatic ecosystem sustainability (WESI) was
constructed on results of our studies [24, 25]. Indices calculation is based on the
water quality ranks as determined by Sládeček’s saprobity indices and nitrate (or
phosphates) concentrations rank.
WESI = Rank S / Rank N-NO3 (Eq. 2)
where:
Rank S – the rank of water quality according to the range of the Sládeček’s
saprobity indices calculated for the sampling station.
Rank N-NO3 – water quality rank according to the range of nitric-nitrogen
concentrations scale.
At WESI ≥ 1, the photosynthetic level is positively correlated with the level of
nitrate concentration. At WESI < 1, photosynthesis is suppressed (presumably due
to a toxic disturbance).
2 Results and Discussion
1. Chemical variables
The water variables dynamic over sampling stations of the Kabul River are
represented in Figure 2. It shown that water temperature, pH, conductivity, TDS,
TSS, Salinity, Chlorides, and Alkalinity were increased down the river.
Concentration of Nitrates, Nitrites, and Ammonia were dramatically increased
immediately after the Swat River tributary input in site 2. In the same time the
Dissolved oxygen, Total Hardness, and Sulfates were decreased in site 2 whereas
the Phosphates here increased. That all let us to conclude that the Swat tributary
influence on the water quality of the Kabul River is high because its water was
slightly low enriched by major ions but bring more polluted waters to the Kabul
River. Hydrochemical variables of the Kabul River in autumn 2014 and spring
2015 are corresponded to the levels of middle polluted water, with increasing of
the organic pollution as BOD down the river. Index Saprobity S, calculated with
species abundance, is increased after station 2 in sites Nowshera and Khairabad
from 1.55 up to 1.59. Index reflects middle polluted water Class III of water quality
with developed community (Figure 3). It is known that temperature, phosphorus
and nitrogen are essential elements for controlling the development of algae
[28-31]. Therefore, our analysis reveals the trophic base for algal community
development, which increased down the river. This situation looks like similar to
it in the Songhua River from climatically similar region of China [32] where water variables of pollution also increased down the river with index saprobity S

156 Sophia Barinova et al.
in the same range 1.57-1.79 that reflect Class III of water quality.
Figure 2. Dynamic of major chemical variables over the Kabul River sampling
stations
2. Algal communities
Altogether 209 species of algae and cyanobacteria were found in four sites of
the Kabul River (Table 1). In Figure 3 are presented qualitative (No of Species)
and quantitative (Abundance Scores) data for four sites. Species richness and algal
abundance are increased similar down the river (Figure 4). Whereas major
abundance in communities was represents by charophytes (Figure 3), species
richness was mostly represented by charophyta also (Figure 4). The algal
community in the major left high mountain tributary of the Kabul River the Swat
River was not so reach in species and strongly regulated by water temperature [9].
Nevertheless, the community structure in the Swat River which flow below 1400
m was similar to it in the studied part of the Kabul River with prevalence of green
algae and diatoms. It is remarkable that Charophyta algae represent about a
quarter of the algal flora in the both rivers and the most diverse of all was
Spirogyra and other filamentous algae. It can be represent some regional feature
in the river basin with high agricultural activity. The prevalence of green algae in
the riverine community is relater also with the latitude of the river basin and
therefore with sunlight intensity such as in the rivers Ukraine [33], China [32],
and Israel [25, 34].
Figure 3. Dynamic of major biological variables over the Kabul River sampling
stations

How water quality in the Kabul River, Pakistan… 157
Table 1. Ecological preferences of algal species and its abundance over the sites in
the Kabul River
Taxa W S N K Hab T Oxy Sal Wat Sap Index
S Tro Nutr
Cyanobacteria
Aphanocapsa incerta (Lemmermann)
G.Cronberg & Komárek 5 5 5 5 P-B - - i - b 2.2 me -
Chroococcus minimus (Keissler)
Lemmermann 1 1 1 1 P-B - - hl - - - o-m -
Chroococcus turgidus (Kützing)
Nägeli 1 1 1 1 P-B,S - aer hl - x-b 0.8 - -
Gloeocapsa punctata Nägeli 5 5 5 5 Ep,S - aer hl - - - - -
Gloeocapsa rupestris Kützing 5 5 0 5 Ep,S - aer - - - - - -
Kamptonema formosum (Bory ex
Gomont) Strunecký, Komárek &
J.Smarda
0 0 3 3 P-B,S - st - - a 3.1 me -
Lyngbya aestuarii Liebman ex
Gomont 0 1 2 0 P-B,S - - mh - o 1.3 - -
Lyngbya majuscula Harvey ex
Gomont 0 2 2 2 - - - - - - - - -
Lyngbya martensiana Meneghini ex
Gomont 2 2 0 0 P-B,S warm st-str - - b-o 1.7 o-m -
Merismopedia convoluta Brébisson
ex Kützing 5 0 0 0 P-B - - - - b-a 2.5 o-m -
Merismopedia tenuissima
Lemmermann 5 5 5 5 P-B - - hl - b-a 2.4 e -
Merismopedia chondroidea Wittrock 0 5 5 5 - - - - - - - - -
Merismopedia trolleri Bachmann 1 2 2 2 - - - - - - - - -
Microcoleus autumnalis (Gomont)
Strunecky, Komárek & J.R.Johansen 0 0 0 5 B,S - st-str - - b 2.3 - -
Microcoleus setchellianus (Gomont)
Strunecky, Komárek & Johansen 3 3 3 3 - - - - - - - - -
Microcystis aeruginosa (Kützing)
Kützing 3 3 0 0 P - - hl - b 2.1 e -
Oscillatoria anguina Bory ex Gomont 5 5 5 5 B,S - - - - - - o-m -
Oscillatoria limosa C.Agardh ex
Gomont 0 0 0 1 P-B - st-str hl - a-o 2.6 e -
Oscillatoria major Vaucher ex Forti 0 0 1 2 B,Ep - - - - b 2.3 m -
Oscillatoria princeps Vaucher ex
Gomont 1 2 2 2 P-B,S - st-str - - a-o 2.8 o-m -
Oscillatoria tenuis C.Agardh ex
Gomont 0 1 0 1 P-B,S - st-str hl - a-o 2.6 me -
Oscillatoria jenensis G.Schmid 0 0 2 2 - - - - - - - - -
Phormidium ambiguum Gomont 3 3 3 3 B,S eterm st-str i - b 2.3 me -
Phormidium articulatum
(N.L.Gardner) Anagnostidis &
Komárek
3 3 3 3 - - - - - - - - -
Phormidium breve (Kützing ex
Gomont) Anagnostidis & Komárek 3 3 0 3 P-B,S - st,aer - - a 3.1 - -
Phormidium chalybeum (Mertens ex
Gomont) Anagnostidis & Komárek 3 3 3 3 P-B,S - st-str - - a 3.3 e -
Phormidium incrustatum Gomont ex
Gomont 3 3 3 3 B - st-str - - x 0.1 o -

158 Sophia Barinova et al.
Table 1. (Continued): Ecological preferences of algal species and its abundance
over the sites in the Kabul River
Phormidium irriguum (Kützing ex
Gomont) Anagnostidis & Komárek 3 3 3 3 B,Ep - aer - - - - me -
Phormidium lucidum Kützing ex
Gomont 0 3 3 3 P-B warm st-str hl - - - - -
Phormidium nigrum (Vaucher ex
Gomont) Anagnostidis & Komárek 1 0 1 0 P-B warm - - - b 2.2 m -
Phormidium retzii Kützing ex
Gomont 0 3 3 3 B,S - st-str - - o-b 1.4 o -
Phormidium stagninum Anagnostidis 1 1 2 1 B,Ep - - - - - - o-m -
Phormidium subfuscum Kützing ex
Gomont 3 3 3 3 B,S - st-str - - o-b 1.5 o -
Phormidium uncinatum Gomont ex
Gomont 0 0 3 3 P-B,S eterm st-str i - b 2.3 me -
Phormidium nigroviride (Thwaites ex
Gomont) Anagnostidis & Komárek 0 3 3 3 - - - - - - - - -
Spirulina major Kützing ex Gomont 0 0 1 1 P-B,S warm st hl - a 3.4 - -
Spirulina nordstedtii Gomont 0 1 1 1 B - - mh - - - - -
Bacillariophyta
Achnanthes elata
(Leuduger-Fortmorel) H.P.Gandhi 0 2 2 2 - - - - - - - - -
Aulacoseira granulata (Ehrenberg)
Simonsen 1 1 1 1 P-B temp st-str i es b 2.0 me ate
Cocconeis placentula Ehrenberg 3 3 3 0 P-B temp st-str i es o 1.35 me ate
Cyclotella meneghiniana Kützing 1 1 0 1 P-B temp st hl sp a-o 2.8 e hne
Cymatopleura solea (Brébisson) W.
Smith 2 0 0 0 B - st-str i - b 2.1 e ate
Cymbella affinis Kützing 3 3 3 3 B temp st-str i sx o 1.1 ot ats
Cymbella cistula (Ehrenberg)
O.Kirchner 0 0 0 3 B - st-str i sx o 1.2 e ats
Cymbella tumida (Brébisson) van
Heurck 2 2 1 2 B temp str i sx b 2.2 me ats
Diatoma tenuis C.Agardh 2 3 3 3 P-B - st-str hl sx o 1.3 e ate
Diatoma vulgaris Bory 3 3 0 3 P-B - st-str i sx b 2.2 me ate
Didymosphenia geminata (Lyngbye)
Mart.Schmidt 1 1 1 1 B - st-str i sx o-x 0.7 ot -
Fragilaria capucina Desmazières var.
capucina 0 2 2 0 P-B - - i es b-o 1.6 m -
Fragilaria capucina var. vaucheriae
(Kützing) Lange-Bertalot 2 3 3 1 P-B,Ep - st-str i sx o-a 1.95 e ate
Fragilaria crotonensis Kitton 0 0 2 1 P - st-str I es o-b 1.5 m ate
Fragilaria goulardii (Brébisson ex
Grunow) Lange-Bertalot 0 0 1 2 - - - - - - - - -
Frustulia vulgaris (Thwaites) De Toni 1 0 1 0 P-B - st i es o-a 1.8 me ate
Gomphonema acuminatum Ehrenberg 2 2 2 3 B - st i es o-b 1.4 o-m ats
Gomphonema augur Ehrenberg 0 2 3 2 B - str i es o-b 1.5 me ats
Gomphonema micropus Kützing 1 2 0 0 B - str i es o 1.3 ot ate
Gyrosigma acuminatum (Kützing)
Rabenhorst 1 1 1 1 B cool st-str i es o-a 1.95 me ate

How water quality in the Kabul River, Pakistan… 159
Table 1. (Continued): Ecological preferences of algal species and its abundance
over the sites in the Kabul River
Gyrosigma nodiferum (Grunow)
Reimer 0 0 1 2 B - - i es b-o 1.7 - -
Mastogloia elliptica (C.Agardh)
Cleve 0 0 2 1 B - - mh - - - - -
Melosira crenulata (Ehrenberg)
Kützing 0 1 2 0 P-B - - - - x-o 0.5 - -
Melosira varians C.Agardh 0 0 2 2 P-B temp st-str hl es b 2.1 me hne
Meridion circulare (Greville)
C.Agardh 2 0 0 0 B - str i es o 1.1 o-m ate
Navicula radiosa Kützing 2 2 1 3 P-B temp st-str i es o 1.3 me ate
Navicula tripunctata (O.F.Müller)
Bory 0 3 2 3 P-B - st-str i es b-o 1.7 e ate
Nitzschia amphibia Grunow 0 0 0 2 P-B,S temp st-str i sp b 2.1 e hne
Nitzschia intermedia Hantzsch 0 1 1 2 P-B - - i es b 2.0 e -
Nitzschia palea (Kützing) W.Smith 2 1 2 2 P-B temp - i sp a-o 2.8 he hce
Nitzschia paleacea Grunow 3 2 2 1 P-B - st-str i es b 2.2 e hce
Nitzschia solita Hustedt 1 1 2 1 B - st mh es a-b 3.6 e -
Nitzschia vitrea G.Norman 0 1 1 1 P-B - - mh - a-o 2.7 e -
Pinnularia viridis (Nitzsch)
Ehrenberg 2 1 2 2 P-B temp st-str i es x 0.3 o-e ate
Rhoicosphenia abbreviata
(C.Agardh) Lange-Bertalot 0 1 2 1 B - st-str i es o-a 1.9 me ate
Rhopalodia gibba (Ehrenberg) Otto
Müller 0 2 1 0 B temp - i es o-b 1.4 o-m -
Stenopterobia pelagica Hustedt 1 2 1 1 - - - - - - - - -
Stephanodiscus hantzschii Grunow 0 1 2 1 P temp st i es a-o 2.7 o-m hne
Surirella ovalis Brébisson 0 0 0 1 P-B - st-str I es a 3.0 me ate
Surirella robusta Ehrenberg 1 1 1 1 P-B - st-str I es x-o 0.5 ot -
Surirella splendida (Ehrenberg)
Kützing 1 1 1 1 P-B - st-str i - o-x 0.7 me -
Surirella striatula Turpin 0 1 1 0 P-B temp - hl - b 2.0 e -
Synedra cyclopum Brutschy 2 2 3 3 B - - i - o-b 1.5 ot -
Tryblionella compressa (J.W.Bailey)
M.Poulin 2 2 2 2 B eterm - mh - - - - -
Ulnaria ulna (Nitzsch) Compère 2 1 1 1 P-B temp st-str i es b-a 2.4 o-e ate
Xanthophyta
Tribonema affine (Kützing) G.S.West 5 5 5 5 B - - hb - x-b 0.8 - -
Tribonema microchloron Ettl 0 0 5 5 - - - - - - - - -
Tribonema minus (Wille) Hazen 0 0 5 5 B - - i - x-b 0.9 - -
Tribonema viride Pascher 5 5 5 5 P-B - - i - o-x 0.7 - -
Tribonema delicatissimum Ettl 0 0 0 5 - - - - - - - - -
Vaucheria fontinalis (Linnaeus)
T.A.Christensen 5 5 5 5 B - - - - o 1.2 - -
Vaucheria sessilis (Vaucher) De
Candolle 5 5 0 0 B - - - - o-b 1.5 - -

160 Sophia Barinova et al.
Table 1. (Continued): Ecological preferences of algal species and its abundance
over the sites in the Kabul River
Vaucheria undulata C.-C.Jao 5 0 5 0 - - - - - - - - -
Vaucheria longipes Collins 0 5 5 5 - - - - - - - - -
Euglenophyta
Euglena mutabilis F.Schmitz 0 2 0 1 B - st-str - - x-b 0.8 - -
Euglena pisciformis Klebs 1 1 1 1 P-B eterm st-str mh - a 3.0 - -
Euglena sanguinea Ehrenberg 2 1 1 1 P-B - st-str i - o 1.25 - -
Euglena viridis (O.F.Müller)
Ehrenberg 0 1 1 0 P-B,S eterm st-str mh - i 4.0 - -
Phacus acuminatus Stokes 1 1 1 1 P-B eterm st-str i - b-a 2.5 - -
Phacus caudatus Hübner 0 1 1 1 P-B eterm st-str i - b 2.3 - -
Chlorophyta
Chaetophora elegans (Roth)
C.Agardh 4 4 4 4 B - - - - o 1.2 - -
Chaetophora lobata Schrank 4 4 4 4 B - - - - o 1.1 - -
Chlamydomonas debaryana
Gorozhankin 0 1 2 1 P - - - - a 3.0 - -
Chlamydomonas ehrenbergii
Gorozhankin 0 2 0 2 P - - - - p-a 3.5 - -
Chlamydomonas globosa J.W.Snow 0 0 0 1 P,S - - - - o-a 1.9 - -
Chlorella minutissima Fott &
Nováková 0 2 3 0 - - - - - - - - -
Chlorella vulgaris Beyerinck 0 0 3 3 P-B,pb,S - - hl - a 3.1 - -
Chlorococcum infusionum (Schrank)
Meneghini 1 1 2 2 P,S - st - - a-o 2.7 - -
Cladophora fracta (O.F.Müller ex
Vahl) Kützing 4 4 4 4 P-B - st-str - - b 2.3 - -
Cladophora glomerata (Linnaeus)
Kützing var. glomerata 4 4 4 4 P-B - st-str i - o-a 1.9 - -
Cladophora glomerata var. crassior
(C.Agardh) Hoek 4 4 4 4 B - - - - o-a 1.9 - -
Cladophora rivularis (Linnaeus)
Hoek 4 4 4 4 - - - - - - - - -
Dactylococcus infusionum Nägeli 0 4 4 4 - - - - - - - - -
Desmodesmus abundans (Kirchner)
E.Hegewald 0 1 1 2 P-B - st-str - - o-a 1.9 - -
Desmodesmus communis
(E.Hegewald) E.Hegewald 0 0 1 0 P-B - st-str i - b 2.15 - -
Hydrodictyon reticulatum (Linnaeus)
Bory 4 4 0 4 P-B - st - - o-a 1.8 - -
Microspora amoena (Kützing)
Rabenhorst 0 5 5 5 B - - - - x-b 0.8 - -
Microspora pachyderma (Wille)
Lagerheim 5 5 5 5 B - - - - o 1.0 - -
Microspora stagnorum (Kützing)
Lagerheim 0 0 0 5 B - st - - b-o 1.6 - -
Microspora willeana Lagerheim 5 5 5 5 B - - - - o 1.2 - -
Microspora wittrockii (Wille)
Lagerheim 0 5 5 5 - - - - - - - - -

How water quality in the Kabul River, Pakistan… 161
Table 1. (Continued): Ecological preferences of algal species and its abundance
over the sites in the Kabul River Mychonastes homosphaera (Skuja)
Kalina & Puncochárová 0 1 1 0 P-B,S - st - - a 3.1 - -
Oedogonium capilliforme Kützing ex
Hirn 5 5 5 5 - - - - - - - - -
Oedogonium crassum Wittrock ex
Hirn 0 5 5 5 - - - - - - - - -
Oedogonium curvum Pringsheim ex
Hirn 0 5 5 5 - - - - - - - - -
Oedogonium echinospermum
A.Braun ex Hirn 0 5 5 5 B - - - - o 1.1 - -
Oedogonium gracilius Tiffany 5 5 5 5 - - - - - - - - -
Oedogonium itzigsohnii De Bary ex
Hirn 0 5 0 0 B - - - - o 1.1 - -
Oedogonium princeps Wittrock ex
Hirn 0 0 5 5 - - - - - - - - -
Pandorina morum (O.F.Müller) Bory 0 1 1 1 P - st i - b 2.3 - -
Rhizoclonium africanum Kützing 5 5 5 5 - - - - - - - - -
Rhizoclonium crassipellitum West &
G.S.West 5 5 5 5 - - - - - - - - -
Rhizoclonium hieroglyphicum
(C.Agardh) Kützing 5 5 5 5 B - st-str hl - o-a 1.9 - -
Rhizoclonium fontanum Kützing 5 5 5 5 - - - - - - - - -
Scenedesmus aculeotatus Reinsch 1 1 1 1 P-B - st-str - - b-o 1.6 - -
Scenedesmus armatus (Chodat)
Chodat 1 0 0 0 P-B - st-str - - b 2.2 - -
Stigeoclonium lubricum (Dillwyn)
Kützing 0 4 4 4 B - - - - b 2.3 - -
Stigeoclonium subsecundum
(Kützing) Kützing 4 4 4 4 B - - - - b 2.3 - -
Tetraëdron caudatum (Corda)
Hansgirg 1 1 2 1 P-B - st-str i - b 2.0 - -
Tetraëdron minimum (A.Braun)
Hansgirg 2 1 1 1 P-B - st-str i - b 2.1 - -
Ulothrix aequalis Kützing 5 5 5 5 B - - - - o 1.3 - -
Ulothrix tenerrima (Kützing) Kützing 0 0 5 0 B - st i - o-a 1.8 - -
Ulothrix tenuissima Kützing 5 0 5 5 B - - i - b-o 1.6 - -
Ulothrix zonata (F.Weber & Mohr)
Kützing 0 5 0 5 P-B - st-str i - o-a 1.8 - -
Charophyta
Chara braunii var. schweinitzii
(A.Braun) Zaneveld 4 4 4 4 B - - - - o 1.2 - -
Chara vulgaris Linnaeus 4 4 4 4 B - st-str - - o 1.1 - -
Closterium acerosum Ehrenberg ex
Ralfs 1 1 1 0 P-B - st-str i - a-o 2.6 e -
Closterium attenuatum Ralfs 0 0 1 1 B - - - - - - m -
Closterium ehrenbergii Meneghini ex
Ralfs 0 1 1 1 P-B - st-str hb - o-b 1.5 me -
Closterium incurvum Brébisson 0 1 1 1 B - - - - o-a 1.8 me -
Closterium moniliferum Ehrenberg ex
Ralfs 2 1 1 1 P-B - st-str i - b 2.1 me -

162 Sophia Barinova et al.
Table 1. (Continued): Ecological preferences of algal species and its abundance
over the sites in the Kabul River
Closterium parvulum Nägeli 0 1 0 0 P-B - - i - b 2.0 m -
Cosmarium angulosum Brébisson 1 1 1 1 B - - - - - - m -
Cosmarium bioculatum Brébisson ex
Ralfs 1 1 1 1 P-B - st-str hb - x-o 0.5 m -
Cosmarium crenatum Ralfs ex Ralfs 0 1 0 0 B,aer - aer - - - - m -
Cosmarium granatum Brébisson ex
Ralfs 1 0 0 1 B - st-str i - o 1.2 m -
Cosmarium impressulum Elfving 0 1 1 1 B,P-B - - hb - b-o 1.6 m -
Cosmarium laeve Rabenhorst 0 1 1 1 P-B - st-str hb - o-a 1.9 me -
Cosmarium punctatum Nordstedt 1 1 0 1 - - - - - - - - -
Cosmarium subcrenatum Hantzsch 2 1 1 1 B,aer - aer - - o 1.1 m -
Cosmarium subtumidum Nordstedt 0 0 2 2 B - - - - - - o-m -
Cosmarium turpinii Brébisson 1 1 1 2 P-B - - i - o-x 0.7 me -
Mougeotia americana Transeau 5 5 5 5 - - - - - - - - -
Mougeotia calcarea (Cleve) Wittrock 5 5 5 5 B - - - - o-x 0.7 - -
Mougeotia floridana Transeau 5 5 5 5 - - - - - - - - -
Mougeotia laevis (Kützing)
W.Archer 5 5 5 0 B - - - - o 1.0 - -
Mougeotia maltae Skuja 0 5 5 5 B - - - - o 1.0 - -
Mougeotia micropora Taft 0 0 0 5 - - - - - - - - -
Mougeotia nummuloides (Hassall) De
Toni 0 5 0 5 B - - - - o 1.0 - -
Mougeotia robusta (De Bary)
Wittrock 5 0 0 5 B - - - - o 1.0 - -
Mougeotia viridis (Kützing) Wittrock 5 5 5 0 B - - - - o 1.3 - -
Spirogyra australica Czurda 5 5 5 5 - - - - - - - - -
Spirogyra bellis (Hassall) P.Crouan &
H.Crouan 5 5 0 5 - - - - - - - - -
Spirogyra borgeana Transeau 5 5 5 5 - - - - - - - - -
Spirogyra californica Stancheva,
J.D.Hall, McCourt & Sheath 0 0 5 0 - - - - - - - - -
Spirogyra cataeniformis (Hassall)
Kützing 5 5 5 5 - - - - - - - - -
Spirogyra colligata Hodgetts 5 5 5 5 - - - - - - - - -
Spirogyra crassa (Kützing) Kützing 5 5 5 5 B - - - - o-b 1.5 - -
Spirogyra daedaleoides Czurda 5 5 5 5 - - - - - - - - -
Spirogyra elongata (Vaucher)
Dumortier 5 5 5 5 - - - - - - - - -
Spirogyra fluviatilis Hilse 5 5 5 5 P-B - - oh - x-b 0.8 - -
Spirogyra juliana Stancheva,
J.D.Hall, McCourt & Sheath 5 5 5 5 - - - - - - - - -
Spirogyra kuusamoensis Hirn 0 5 5 5 - - - - - - - - -
Spirogyra lutetiana Petit 5 0 0 0 - - - - - - - - -
Spirogyra majuscula Kützing 5 5 5 5 B - - - - o-a 1.8 - -
Spirogyra maxima (Hassall) Wittrock 5 5 5 5 B - - - - o 1.1 - -

How water quality in the Kabul River, Pakistan… 163
Table 1. (Continued): Ecological preferences of algal species and its abundance
over the sites in the Kabul River
Spirogyra micropunctata Transeau 5 5 5 5 - - - - - - - - -
Spirogyra notabilis Taft 5 5 5 5 - - - - - - - - -
Spirogyra parvula (Transeau) Czurda 5 5 5 5 B - st-str - - - - - -
Spirogyra pratensis f. minor (Liu)
C.-C.Jao 5 5 5 5 - - - - - - - - -
Spirogyra protecta H.C.Wood 5 5 5 5 - - - - - - - - -
Spirogyra scrobiculata (Stockmayer)
Czurda 5 5 5 0 - - - - - - - - -
Spirogyra setiformis (Roth) Martens
ex Meneghini 0 5 5 5 - - - - - - - - -
Spirogyra spreeiana Rabenhorst 0 0 0 5 - - - - - - - - -
Spirogyra teodorescui Transeau 0 5 0 5 - - - - - - - - -
Spirogyra varians (Hassall) Kützing 5 5 5 5 P-B - - oh - b 2.1 - -
Spirogyra weberi Kützing var. weberi 5 5 5 5 - - - - - - - - -
Spirogyra weberi var. grevilleana
(Hassal) O.Kirchner 5 5 5 5 B - st - - - - - -
Spirogyra aequinoctialis G.S.West 5 5 5 5 - - - - - - - - -
Temnogyra collinsii I.F.Lewis 5 5 5 5 - - - - - - - - -
Zygnema aplanosporum Stancheva,
J.D.Hall & Sheath 5 5 5 5 - - - - - - - - -
Zygnema argillosum Kadlubowska 0 0 5 5 - - - - - - - - -
Zygnema carinthiacum
Beck-Mannagetta 5 5 5 5 B - - - - o 1.0 - -
Zygnema cruciatum (Vaucher)
C.Agardh 0 5 5 5 B - - - - x-b 0.8 - -
Zygnema cylindricum Transeau 5 5 5 5 - - - - - - - - -
Zygnema giganteum Randhawa 0 0 0 5 - - - - - - - - -
Zygnema inconspicuum Czurda 5 5 5 5 B - - - - o 1.0 - -
Zygnema maius Czurda 0 5 5 5 - - - - - - - - -
Zygnema normanii Taft 5 5 5 5 - - - - - - - - -
Zygnema stellinum (O.F.Müller)
C.Agardh 5 5 5 5 B - - - - o-x 0.7 - -
Zygnema sterile Transeau 5 5 5 5 B - - - - o 1.0 - -
Zygnemopsis transeauana Randhawa 0 5 5 5 - - - - - - - - -
Table legend: Sites: W – 1. Warsak; S – 2. Sardaryab; N – 3. Nowshera; K – 4. Khairabad. Substrate preferences (Habitat): P – planktonic,
P-B – plankto-benthic, B – benthic, Ep – epiphyte, S - soil. Temperature preferences (Temp): cool – cool-water, temp – temperate, eterm –
eurythermic, warm – warm-water. Oxygenation and streaming (Oxy): st – standing water, str – streaming water, st-str – low streaming
water, aer – aerophiles. Halobity degree according Hustedt [35] (Sal): hb – oligohalobes-halophobes, i – oligohalobes-indifferent, mh –
mesohalobes, hl – halophiles. Saprobity groups according Watanabe et al. [36] (D): sp – saprophiles, es – eurysaprobes, sx – saproxenes.
Species-specific Index of Saprobity (S). Self-purification zone preferences (Sap): x – xenosaprob; x-o – xeno-oligosaprob; o-x –
oligo-xenosaprob; x-b – xeno-beta-mesosaprob; o – oligosaprob; o-b – oligo-beta-msosaprob; b-o – beta-oligosaprob; o-a –
oligo-alpha-mesosaprob; b – beta-mesosaprob; b-a – beta-alpha-mesosaprob; a – alpha-mesosaprob; a-p – alpha-polysaprob; a-b –
alpha-beta-mesosaprob; i – i-eusaprob. Trophic state (Tro) [37]: ot – oligotraphentic; o-m – oligo-mesotraphentic; m – mesotraphentic; me –
meso-eutraphentic; e – eutraphentic; he – hypereutraphentic; o-e – oligo- to eutraphentic (hypereutraphentic). Nitrogen uptake metabolism
(Aut-Het) [37]: ats – nitrogen-autotrophic taxa, tolerating very small concentrations of organically bound nitrogen; ate –
nitrogen-autotrophic taxa, tolerating elevated concentrations of organically bound nitrogen; hne – facultatively nitrogen-heterotrophic taxa,
needing periodically elevated concentrations of organically bound nitrogen; hce – obligately nitrogen-heterotrophic taxa, needing
continuously elevated concentrations of organically bound nitrogen.

164 Sophia Barinova et al.
3. Bio-indication of Water Quality
We constructed bio-indication plots on the base of ecological preferences of
the Kabul River species (Table 1) for revealed indicators distribution over major
environmental variables (Figures 4-6). Can be seen that number of species as well
as total number of each group indicators were sufficiently increase after the Swat
tributary input.
Figure 4. Bio-indicational plots in the Kabul River sampling stations of taxonomic
Division, Habitat preferences, temperature and oxygen.
Bio-indicational plots in Figure 4 show prevalence of benthic or
plankto-benthic species, temperate temperature indicators, and slow streaming
middle oxygenated waters.

How water quality in the Kabul River, Pakistan… 165
Figure 5. Bio-indicational plots in the Kabul River sampling stations by salinity,
organic pollution according Watanabe, and according Sládeček indices, and
Classes of Water Quality.
Bio-indicational plots in Figure 5 show prevalence of salinity-indifferents,
eurysaprobes, III-IV Class of Water Quality that reflects middle polluted waters in
the Kabul River. The role of salinity in the riverine and especially lacustrine
community was studied during last year’s [38-41]. Usually salinity play the
negative role in algal species richness but here, in the Kabul River, we can see
that algal community was not suppressed and even stimulated down the river
because the chlorides come together with nutrients.

166 Sophia Barinova et al.
Figure 6. Bio-indicational plots in the Kabul River sampling stations by trophic
state and type of nutrition.
Bio-indicational plots in Figure 6 show prevalence of mesotrophes,
autotrophic algae with tolerance to middle concentration of Nitrates. The
hydrological regime has both indirect and direct effect on aquatic community and
water quality through the changes of depth, nutrient concentrations, and water
volume. Generally, the symptoms of eutrophication of aquatic ecosystems are
more clearly manifested in periods of low water levels [42,43]. Here, in the Kabul
River, we can see that algal community was affected with water temperature and
nutrients, which increased down the river especially in the low-water periods of
autumn and spring. Therefore, studied part of the river can be characterized on the
base of bio-indication as middle polluted in the mesotrophic stage with tendency
of increasing of the eutrophication level. The same situation we can see in other
similar rivers which basins are in the Southern Eurasia regions such as Songhua,
Lower Jordan or Southern Bug [25,32-34,40]. Here integral bio-indicational
method for water quality and aquatic ecosystem sustainable assessment has been
represented relevant and stabile results, which can be employed in monitoring of
regional water quality.
3. Conclusion
Our results let us to conclude that firstly studied part of the Kabul River have
developed algal community with increasing of species richness and its abundance
in communities down the river. This process was stimulated not only the water
temperature increasing but also nutrients runoff from agricultural and domestic
activity in the Peshawar Valley and from the major tributary the Swat River. The
Swat River left high mountain tributary bring to the Kabul River its low enriched by major ions but more organically polluted waters. Algal community was increase in

How water quality in the Kabul River, Pakistan… 167
species richness below the tributary input [44] as we can see in the rivers in the
climatically similar regions of Eurasia [32]. We revealed prevalence of green
algae and diatoms in studied algal community. The Charophyta species such as
filamentous Spirogyra and other filamentous algae were diverse and can be
represent some regional feature in the river basin with high agricultural activity
such as in China [32].
Water temperature can be characterized as one of the major variable that
stimulated riverine algal community. In other hand, water salinity was impacted
variable, which increased together with BOD and can be results of organic
pollution impact that come not only from industrial and agricultural activity in the
Peshawar Valley but also from the Swat River tributary as can be seen in the
bio-indication plots. The nitrates concentration was also important variable, which
together with temperature were affected riverine diversity in the Kabul River.
Bio-indication reflect arising of eutrophication level down the river but
calculated indices of saprobity S in the sites of studied part of the Kabul River
show high self-purification capacity of algal community. Therefore, we can to
conclude that integral and economy method of bio-indication for water quality
assessment can be employed in purpose of monitoring the regional water quality.
Acknowledgements. This work has been partly supported by the Israeli Ministry
of Absorption.
References
[1] E. Jeppesen, S. Brucet, L. Naselli-Flores, E. Papastergiadou, K. Stefanidis,
T. Noges et al., Ecological impacts of global warming and water abstraction
on lakes and reservoirs due to changes in water level and related changes in
salinity, Hydrobiologia, 750 (2015), 201-227.
http://dx.doi.org/10.1007/s10750-014-2169-x
[2] J.D. Wehr, Freshwater Algae of North America: Ecology and Classification,
Academic Press, 2002.
[3] F.E. Round, Biology of the Algae, Arnold, 1973, 57-107.
[4] R.J. Stevenson, M.L. Bothwell, R.L. Lowe, J.H. Thorp, Algal Ecology:
Freshwater Benthic Ecosystem, Academic press, 1973.
[5] D.V. Chapman, Water Quality Assessments: a Guide to the Use of Biota,
Sediments and Water in Environmental Monitoring, Taylor and Francis,
1996. http://dx.doi.org/10.4324/noe0419216001

168 Sophia Barinova et al.
[6] M.K. Leghari, S.B. Waheed, M.K. Leghorn, Ecological study of algal flora
of Kunhar River of Pakistan, Pakistan Journal of Botany, 33 (2001),
629-636.
[7] S. Sebastian, Algal diversity of river Meenachil in Kerala, India, Indian
Journal of Applied Research, 6 (2016), no. 3, 203-204.
[8] M. Munir, R. Qureshi, M. Ilyas, M. Munazir, M.K. Leghari, Systematics of
Chroococcus from Pakistan, Pakistan Journal of Botany, 48 (2016), no. 1,
255-262.
[9] S. Barinova, N. Ali, Barkatullah, F.M. Sarim, Ecological adaptation to
altitude of algal communities in the Swat Valley (Hindu Cush Mountains,
Pakistan), Expert Opinion on Environmental Biology, 2 (2013), no. 2,
1-15. http://dx.doi.org/10.4172/2325-9655.1000104
[10] I. Khuram, N. Ahmad, S. Jan, S. Barinova, Freshwater green algal
biofouling of boats in the Kabul River, Pakistan, Oceanological and
Hydrobiological Studies, 43 (2014), no. 4, 329-336.
http://dx.doi.org/10.2478/s13545-014-0150-y
[11] K.M. Salim, M.H. Khan, The Diatomales: the Fresh Water Diatoms of
Peshawar Valley, Dept. Botany, Peshawar Univ. Press, Peshawar, Pakistan,
1960.
[12] Government of Pakistan, Population census organization statistics division
government of Pakistan, Islamabad Census Publication, 68 (1998), 01-25.
[13] R.K. Gresswell, A.J. Huxley, Standard Encyclopedia of the World's Rivers
and Lakes, G. P. Putnam's Sons, New York, NY, 1965.
[14] A.M. Yousafzai, A.R. Khan, A.R. Shakoori, Pollution of large, subtropical
rivers-river Kabul, Khyber-Pakhtun Khwa province, Pakistan:
physico-chemical indicators, Pakistan Journal of Zoology, 42 (2010), no. 6,
795-808.
[15] L. Edler, M. Elbrächter, The Utermöhl Method for Quantitative
Phytoplankton Analysis: Microscopic and Molecular Methods for
Quantitative Phytoplankton Analysis, Paris: UNESCO Publishing, 12-13,
2010.
[16] E. Swift, Cleaning diatom frustules with ultraviolet radiation and peroxide,
Phycologia, 6 (1967), no. 2, 161-163.
http://dx.doi.org/10.2216/i0031-8884-6-2-161.1

How water quality in the Kabul River, Pakistan… 169
[17] E.G. Bellinger, D.C. Sigee, Freshwater Algae: Identification, Enumeration
and Use as Bioindicators, John Wiley & Sons, Ltd., Chichester, West
Sussex, 2015. http://dx.doi.org/10.1002/9781118917152
[18] E.J. Cox, Identification of Freshwater Diatoms from Live Material,
Chapman & Hall, London, Weinheim, New York, Tokyo, Melbourne, and
Madras, 1996.
[19] G.W. Prescott, Algae of the Western Great Lakes Area, with an Illustrated
Key to the Genera of Desmids and Freshwater Diatoms, Dubuque, Iowa, W.
M. C. Brown Company, 1962. http://dx.doi.org/10.5962/bhl.title.4650
[20] T.V. Desikachary, Cyanophyta, New Dehli, India, Indian Council of
Agriculture Research, 1959.
[21] L.H. Tiffany, M.E. Britton, The Algae of Illinois, Chicago Univ. Press,
Chicago, U.S.A., 1952.
[22] E.N. Transeau, The Zygnemataceae, Columbus, U.S.A., Ohio State Univ.
Press, 1951.
[23] F.S. Collins, The Green Algae of North America, Tufts College Studies
(Science), 2, 1909, 79-480. http://dx.doi.org/10.5962/bhl.title.23727
[24] S.S. Barinova, L.A. Medvedeva, O.V. Anissimova, Diversity of Algal
Indicators in Environmental Assessment, Pilies Studio, Tel Aviv, 2006.
[25] S. Barinova, Algal Diversity Dynamics, Ecological Assessment, and
Monitoring in the River Ecosystems of the Eastern Mediterranean, Nova
Science Publishers, New York, 2011.
[26] V. Sládeček, System of water quality from the biological point of view,
Ergebnisse der Limnologie, 7 (1973), 1-128.
[27] V. Sládeček, Diatoms as indicators of organic pollution, Acta Hydrochimica
et Hydrobiologica, 14 (1986), 555-566.
http://dx.doi.org/10.1002/aheh.19860140519
[28] A.V. Goncharov, Comparison of Reservoirs in the Moskva–Vazuza Water
System in Terms of Phytoplankton Abundance and Eutrophication Degree,
Water Resources, 34 (2007), no. 1, 70-74.
http://dx.doi.org/10.1134/s0097807807010071
[29] H. Xu, G. Min, J. Choi, K.A.S. Al-Rasheid, X. Lin, M. Zhu, Temporal
dynamics of phytoplankton communities in a semi-enclosed mariculture

170 Sophia Barinova et al.
pond and their responses to environmental factors, Chinese Journal of
Oceanology and Limnology, 28 (2010), no. 2, 295-303.
http://dx.doi.org/10.1007/s00343-010-9257-1
[30] M. Tolotti, M. Manca, N. Angeli, G. Morabito, B. Thaler, E. Rott, E.
Stuchlik, Temperature modulated effects of nutrients on phytoplankton
changes in a mountain lake, Hydrobiologia, 698 (2012), 61-75.
http://dx.doi.org/10.1007/s10750-012-1146-5
[31] Y. Mingli, Z.H. Cuixia, J. Zengjie, G. Shujin, S. Jun, Seasonal variations in
phytoplankton community structure in the Sanggou, Ailian, and Lidao Bays,
J. Ocean Univ. China (Oceanic and Coastal Sea Research), 13 (2014), no.
6, 1012-1024. http://dx.doi.org/10.1007/s11802-014-2305-2
[32] S. Barinova, Na Liu, Jiyang Ding, Yonglei An, Xueming Qin, Chenxin Wu,
Ecological assessment of water quality of the Songhua River upper reaches
by algal communities, Acta Ecologica Sinica, 36 (2016), no. 3, 126-132.
http://dx.doi.org/10.1016/j.chnaes.2015.12.001
[33] O. Bilous, S. Barinova, P. Klochenko, Phytoplankton communities in
ecological assessment of the Southern Bug River upper reaches (Ukraine),
Ecohydrology and Hydrobiology, 12 (2012), no. 3, 211-230.
http://dx.doi.org/10.1016/s1642-3593(12)70205-7
[34] S. Barinova, V.A. Krassilov, Algal diversity and bio-indication of water
resources in Israel, International Journal of Environment and Resource, 1
(2012), no. 2, 62-72.
[35] F. Hustedt, Systematische und Ökologische Untersuchungen über die
Diatomeenflora von Java, Bali und Sumatra, Arch. Hydrobiol. Suppl., 15
(1938-1939), 131-177, 393-506, 638-790; 16, 1-155, 274-394.
[36] T. Watanabe, K. Asai, A. Houki, Numerical estimation to organic pollution
of flowing water by using the epilithic diatom assemblage – Diatom
Assemblage Index (DAIpo), Science Total Environment, 55 (1986),
209-218. http://dx.doi.org/10.1016/0048-9697(86)90180-4
[37] H. Van Dam, A. Martens, J. Sinkeldam, A coded checklist and ecological
indicator values of freshwater diatoms from the Netherlands, Netherlands J.
Aquatic Ecol., 28 (1994), no. 1, 117-133.
http://dx.doi.org/10.1007/bf02334251
[38] S.S. Barinova, P.M. Tsarenko, E. Nevo, Algae of experimental pools on the
Dead Sea coast, Israel, Israel Journal of Plant Science, 52 (2004), no. 3,
265-275. http://dx.doi.org/10.1560/v889-764e-mcdy-npdp

How water quality in the Kabul River, Pakistan… 171
[39] S.S. Barinova, T.M. Bragina, E. Nevo, Algal species diversity of arid region
lakes in Kazakhstan and Israel, Community Ecology, 10 (2009), no. 1, 7-16.
http://dx.doi.org/10.1556/comec.10.2009.1.2
[40] S. Barinova, M. Tavassi, H. Glassman, E. Nevo, Algal indication of
pollution in the Lower Jordan River, Israel, Applied Ecology and
Environmental Research, 8 (2010a), no. 1, 19-38.
http://dx.doi.org/10.15666/aeer/0801_019038
[41] V. Klymiuk, S. Barinova, Phytoplankton cell size in saline lakes, Research
Journal of Pharmaceutical, Biological and Chemical Sciences, 7 (2016), no.
1, 1077-1085.
[42] Y. ZhengJian, L. DeFu, J. DaoBin, X. ShangBin, Influence of the
impounding process of the Three Gorges Reservoir up to water level 172.5
m on water eutrophication in the Xiangxi Bay, Science China Technological
Sciences, 53 (2010), no. 4, 1114-1125.
http://dx.doi.org/10.1007/s11431-009-0387-7
[43] X. Chuai, X. Chen, L. Yang, J. Zeng, A. Miao, H. Zhao, Effects of climatic
changes and anthropogenic activities on lake eutrophication in different
ecoregions, Int. J. Environ. Sci. Technol., 9 (2012), 503-514.
http://dx.doi.org/10.1007/s13762-012-0066-2
[44] Z. Ullah, H. Khan, A. Waseem, Q. Mahmood, U. Farooq, Water quality
assessment of the River Kabul at Peshawar, Pakistan: Industrial and urban
wastewater impacts, Journal of Water Chemistry and Technology, 35
(2013), no. 4, 170-176. http://dx.doi.org/10.3103/s1063455x1304005x
Received: August 25, 2016; Published: October 11, 2016