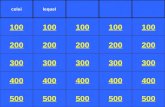
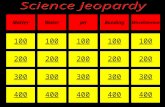
Hosted by Mrs. Sowa 100 200 400 300 400 Density Equation Graphs Matter Models Measuring m and V 300...
-
Upload
derrick-carr -
Category
Documents
-
view
215 -
download
0
Transcript of Hosted by Mrs. Sowa 100 200 400 300 400 Density Equation Graphs Matter Models Measuring m and V 300...
100 100
200 200
400 400
300
400
Density Equation
Graphs Matter Models
Measuring
m and V
300 300 300
200
400
200
100
500 500 500 500
100
Row 1, Col 1
What is the density of a substancethat has a mass of
125 g and a volume of 25 cm3?
5 g/cm3
1,2
When you graph data on the mass and volume of a substance,
which variable goes on the y-axis?
mass
1,3
Less, because water is less dense
than lead.
A cylinder of water that is equal in volume to a cylinder of lead
will have more, the same, or less mass than the lead?
1,4
How would you find the volume of a large block of wood?
Measure the length, width and height
and multiply them together.
2,2
How does a graph of mass vs. volume
tell you that density is
a characteristic property?
Since the mass divided by the volume for all amounts of the substance is the same, the points fall in a line.
3,1
An object with a density of 2.0 g/cm3 displaces 5 mL of water. What is its mass?
2.0 g/cm3 x 5 cm3 = 10 g
3,2
On a graph of mass vs. volume,
where would the line for water be
compared with the
line for vegetable oil?
The line for water would be above the line forvegetable oil b/c water is more dense.
3,3
Which is more dense and why?
They have the same density. Although B has more volume, it also has more matter. Also, the particles
are spaced the same for both A and B.
FIGURE 1 A B
A
BFigure 1
4,1
A piece of wax (0.9 g/cm3) and a piece of copper (9.0 g/cm3)
have the same volume.
How do their masses compare? Be specific!
The mass of the copper is 10 times greater than themass of the wax.
4,2
Consider a graph of m vs. V. Substances with lines that are steeper than the line for water
would _____when placed in water.
Sink
4,3
A cube of gold (19.3 g/cm3) and a cube of nylon (1.2 g/cm3) both have sides
that are 2 cm in length.
Which is more dense?
The cube of gold. (Size doesn’t matter)
5,1
A large nylon bolt has a density of 1.2 g/cm3 and mass of 24 g.
What is it’s volume?
24 g 1.2 g/cm3 = 20 cm3
5,2
When graphing mass and volume data for the Density and Sample Size Lab,
how could you determine that the cylinders were made from
different substances?
The data fell into two distinct lines that had different slopes (different densities)
5,3
Which substance has a greater mass?
B, because it has more matter particles.
FIGURE 1 A B
A
BFigure 1