Heart Transplantation Ventricular Assist Device€¦ · Heart Transplantation & Ventricular Assist...
Transcript of Heart Transplantation Ventricular Assist Device€¦ · Heart Transplantation & Ventricular Assist...

Heart Transplantation
&
Ventricular Assist Device
Dr Timmy WK Au
Cardiothoracic Surgery,
University of Hong Kong,
Queen Mary Hospital,
2013 Hospital Authority Convention

Relevant Financial Relationship
Disclosure Statement
I will not discuss off label use and/or investigational use of drugs/devices
The following relevant financial relationships exist related to my role in this
session: No relationships to disclose
Heart Transplantation & Ventricular Assist Device
Timmy WK Au FRCS FHKAM

Management of
“End-Stage Heart Failure”
Drugs including inotropes
Implantable devices like Automatic
Implantable Cardiac Defibrillators (AICD)
and/or CRT
Intra-Aortic Balloon Pump
ECMO
Ventricular Assist Device
Cardiac Transplantation

Surgical Therapy - What Evidence
Surgical Therapy Current recommendation Level of Evidence
Heart Transplantation Class I B
CABG for
Ischaemic Cardiomyopathy
Class I / II B
LVAD Class IIA B
Mitral Surgery Class IIB C
Surgical Ventricular
Restoration Class IIB C
Prosthetic Restrain
Device Class III C
ACC / AHA CHF guidelines Mx 2005

That was how it started
…………..in 1967

Heart transplantation
Historical background
1905 Carrel - heart transplantation in dog
1944 Medawar - concepts of organ rejection
1964 Shumway - technique of heart transplant
1967 Barnard - world first human heart transplant
1980 Stanford University - Cyclosporin A
1996 USA - 3500 heart transplant / year

NUMBER OF HEART TRANSPLANTS
REPORTED BY YEAR
0
500
1000
1500
2000
2500
3000
3500
4000
4500
5000
1982
1983
1984
1985
1986
1987
1988
1989
1990
1991
1992
1993
1994
1995
1996
1997
1998
1999
2000
2001
2002
2003
2004
2005
2006
2007
2008
2009
Nu
mb
er
of
Tra
ns
pla
nts
Other
EuropeNorth America
NOTE: This figure includes only the heart transplants that are
reported to the ISHLT Transplant Registry. As such, the
presented data may not mirror the changes in the number of
heart transplants performed worldwide
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

AVERAGE CENTER VOLUME Heart Transplants: January 1, 2003 – June 30, 2010
45
103
30
13 246
62
0
10
20
30
40
50
60
70
80
90
100
110
1-4 5-9 10-19 20-29 30-39 40-49 50-74 75+
Average number of heart transplants per year
Nu
mb
er
of
ce
nte
rs
0
5
10
15
20
25
30
35
40
45
50
55
Number of centers Percentage of transplants
Pe
rce
nta
ge
of
tra
ns
pla
nts
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

Queen Mary Hospital
Heart Transplant in HK

HEART TRANSPLANTATION Kaplan-Meier Survival (1/1982-6/2005)
0
20
40
60
80
100
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25
Years
Su
rviv
al (%
)
Half-life = 11.0 years
Conditional Half-life = 14.0 years
N=89,006
N at risk at 25 years = 98
HEART TRANSPLANTATION Kaplan-Meier Survival (1/1982-6/2009)
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

Actuarial survival of Heart
Transplant in HK
1 year survival 87%
5 years survival 82%
10 years survival 58%

ADULT HEART TRANSPLANT RECIPIENTS:
Relative Incidence of Leading Causes of Death (Deaths: January 1992 - June 2010)
0
10
20
30
40
50
0-30 Days
(N=4,094)
31 Days –
1 Year
(N=4,028)
>1 Year – 3
Years
(N=3,166)
>3 Years –
5 Years
(N=2,674)
>5 Years –
10 Years
(N=6,273)
>10 – 15
Years
(N=3,616)
>15 Years
(N=1,753)
CAV Acute RejectionMalignancy (non-Lymph/PTLD) Infection (non-CMV)Graft Failure Multiple Organ FailureRenal Failure
Pe
rce
nta
ge
of
De
ath
s
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANT RECIPIENTS:
Cumulative Incidence of Leading Causes of Death (Transplants: January 1992 - June 2009)
0%
2%
4%
6%
8%
10%
12%
14%
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15
Time (years)
CAV Acute Rejection
Malignancy (non-Lymph/PTLD) Graft Failure
CMV Infection (non-CMV)
Inc
ide
nc
e o
f C
au
se
-Sp
ec
ific
De
ath
s
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

Post-transplant complications
Infections - bacterial, viral, fungal
Rejection
Hypertension - Cyclosporin / steroid
Hyperlipidaemia - Cyclosporin / steroid
Allograft coronary disease - 50% in 5 yrs
Renal dysfunction - Cyclosporin
Malignancy - skin, lymphoma, lung cancer
GI - ulcers, jaundice, perforation
Osteoporosis - steroid / age > 60
Psychological - anxiety, depression

Recent trend in Heart Tx
• Marginal donor heart management
• Changing trend in immunosuppression Rx
• Statins and angiotensin receptor blockers
• Reoperation for heart transplant patients
• Sophisticated LV assist device
• Xenotransplantation ???

Marginal donor heart management
Impaired heart contractility secondary to brain death triggered autonomic and cytokine storm
Optimize
• Haemodynamic, metabolic and respiratory
• Swan Ganz catheter
• Hormonal resuscitation – steroid, thyroxin, vasopressin & insulin
• Post THx ECMO support

Today’s Status of Heart Tx
Survival is excellent
Incidence of Acute Rejection is low
Long term complications are still
worrying
No growth is past 10 years
Growing demand for Mechanical
Circulatory and VAD support

289
completed HTx work-up
206 suitable
For HTx 83 delisted
120 heart
transplanted
61 died
while waiting
25
waiting list
In average, 30 % of eligible Tx patients died while
waiting for suitable donor !
Potential Heart Transplant Candidates Referred to
the Grantham Hospital for Assessment 1992-2012

ADULT HEART TRANSPLANTATION % OF PATIENTS BRIDGED WITH MECHANICAL
CIRCULATORY SUPPORT* (Transplants: 1/2000 – 12/2009)
0
10
20
30
40
50
2000 2001 2002 2003 2004 2005 2006 2007 2008 2009
Year
% o
f p
ati
en
ts
* LVAD, RVAD, TAH ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

The man with the vision ….

Pagani FD ISHLT 2013

The ‘Ideal’ Blood Pump
Simple to use – able to set up & prime quickly involving minimal equipment & personnel
Minimal priming volume and connections
Gentle, atraumatic blood handling capabilities, comprised of biocompatible materials
Minimal heat generation
Minimal anticoagulant requirements
Easy to visualize during support
Easy to transport
Able to fully support cardiac output (days/weeks)
Cost effective

Pulsatile VAD - Excor

Different types of design of VADs

The Evolution of VAD Devices
Paracorporeal
Pneumatic
Pulsatile
Uni- or
Biventricular
Implantable
Electric
Pulsatile
Large
Multiple moving
parts
Implantable
Electric
Continuous flow
Axial design
Smaller
Single moving part
Implantable
Electric
Continuous flow
Centrifugal design
Smaller
Bearingless
Implantable
Electric
Continuous flow
Axial design
Smaller
Partial support

The REMATCH Trial
Randomised Evaluation of Mechanical Assistance in the Treatment of Congestive Heart Failure [REMATCH]
129 patient with end-
stage CHF randomised
to permanent HeartMate
I vs optimal medical
therapy – 20 centers in
US Rose et al. REMATCH study group. N Engl J Med 2001

Survival of patients in the randomized evaluation of
mechanical assistance therapy as an alternative in CHF study.
Rose EA et al. N Engl J Med 2001

HeartMate II Initial BTT Results (n=133)
NEJM 2007;357:885-96.

Survival Following VAD Implantation
ISHLT 2009 Report

ISHLT 2013 Quarterly Report

Summary of BTT Outcomes Reference Study Enrollment
period
n Survival to Tx,
Recovery, or
Ongoing Device
Support at 180
Days
Miller, Pagani, Russell et al
NEJM 357:885-896, 2007
HM II Pivotal
Trial 3/05- 5/06 133 79%
Pagani, Miller, Russell et al
JACC 54:312-321, 2009
HM II Pivotal
Trial 3/05- 3/07 281 84%
Starling, Naka, Boyle et al
JACC 57(19): 1890-9, 2011.
Post Approval
Study
4/08 – 8/08 169 90%
John, Ann Thorac Surg 19:
1406-13. 2011
Post-trial
Study
4/08-9/10 1496 89%

Surgical Therapy – Current Status US
Surgical Therapy Current recommendation Level of Evidence
Heart Transplantation Class I B
CABG for
Ischaemic Cardiomyopathy
Class I / II B
LVAD Class IIA B
Mitral Surgery Class IIB C
Surgical Ventricular
Restoration Class IIB C
Prosthetic Restrain
Device Class III C
ACC / AHA CHF guidelines Mx 2005

Current Status - Europe
I
IIA

INTERMACS Profiles & Timing of VAD Implant
Description Time Frame
1 Patient with life-threatening hypotension despite rapidly escalating inotropic
support, critical organ hypoperfusion with increasing lactate levels and/or systemic
acidosis. “Crash and burn”
within hours
2 Patient with declining function despite IV inotropic support, may be manifest
by worsening renal function, nutritional depletion, inability to restore volume
balance. “Sliding on inotropes”
few days
3 Patient with stable blood pressure, organ function, nutrition, and symptoms on IV
inotropic support, but cannot wean off inotropes. “Dependent stability”
Elective over a
few weeks
4 Patient can be stabilized close to normal volume status but with relapses of
CHF, & fluid retention. Intensive management strategies . “Frequent flyer”
Elective over
weeks to months
5 Patient is comfortable at rest without congestive symptoms, but may have underlying refractory
elevated volume status, often with renal dysfunction. “Housebound”
Variable, depends
on nutrition, organ
function & activity
6 Patient without evidence of fluid overload is comfortable at rest and with activities, but fatigues
after the first minutes of any meaningful activity. “Walking wounded”
Variable, depends
on nutrition, organ
function,& activity
7 A placeholder for future specification, patients without recent unstable fluid balance, living
comfortably with meaningful activity limited to mild exertion. Not indicated


Bridge to
Recovery

Bridge to Recovery
Post cardiopulmonary bypass, and cannot be
weaned from the heart lung machine
Continue to deteriorate despite multiple
inotropes (at increasing dosages) and intraaortic
balloon support (IABP)
Acute decompensated heart failure secondary to
acute myocarditis / cardiomyopathy
Acute STEMI in combination with cardiogenic
shock

Bridge to
Decision

Bridge to Decision
Significant cardiac event and have questionable
neurologic status
Intervention has started prior to making a full
determination of recovery and outcome
Intervention has started prior to speaking with
the family

Bridge to Decision

Bridge to
Bridge

Bridge to a Bridge
A less expensive, or shorter termed device, is
initiated, and then switch to a long term
device once the patients condition has
stabilized (Cost considerations)
Peri-corporeal based systems are less
expensive, quicker, and require less
equipment
Opportunity to stabilize the patient -> before
better planning or transferral

CentriMag BiVentricular Support 1. Left and Right Ventricules Support
2. CentriMag cost 10% of HeartMate II
3. Proven means for Bridge to LVAD, Bridge
to transplant, Bridge to Recovery
4. Superior to ECMO

0
2
4
6
8
10
12
14
16
18
2007 2008 2009 2010 2011 2012 (up to 1/10/12)
Nu
mb
er
of
Case
ECMO operation during the year from 2007 to Aug. 2012
ECMO and CentriMag program for
Severe Cardiogenic Shock in QMH


Update of CentriMag Program
Queen Mary Hospital
9 Patients received CentriMag BiVAD since
July 2011
•3 patients successfully had heart transplantation – 29 days, 21
days, 85 days
•1 patient successfully bridge to HeartMate II implantation
•4 patients died – fungal sepsis, pulmonary HT & multi-organs
failure
•1 patient is still waiting in ICU for Heart Tx

Bridge to
Transplant

Bridge to Transplant

Bridge to Candidacy

Implantable VAD Program in QMH
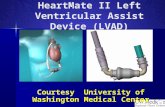
HeartMate II
[Thoratec]

Update of QMH LVAD Program [BTT]
Since August 2010
9 End stage heart failure patients received LVAD
7 patients discharged home
5 patients are back to full or part time work
2 died - massive cerebral heamorrhage 6 mth ;
- multi-organ bleeding and thromboembolic
stroke 2 mth .

Contraindications for Heart Tx
Relative contraindications
• Age ≥ 65 years
• Acute myocarditis, PVD, COPD
Contraindications
• Severe pulmonary HT (fixed PVR>5 Wu, PAP>60 mm Hg)
• Psychosocial problem (alcoholism, drug addiction, severe psychotic disease etc)
• Systemic illness (amyloidosis, connective tissue diseases, neurological and muscular disorders, morbid obesity, severe cachlexia)




Destination Therapy
Refractory NYHA Class IV Heart Failure
Non-candidate for heart Tx
LVEF<25%
Failed optimal medical management for 60 of past 90 days
VO2 < 12mL/Kg/Min or need for inotropes – Hypotension,
renal dysfunction, pulmonary congestion
BSA > 1.5 m2
Absence of co-morbidities limiting survival

Destination Therapy Outcomes
Reference Study Enrollment
period
n One-Year
Survival
Two-Year
Survival
Slaughter, Rogers,
Milano et al NEJM
2009;361:2241-51
HM II Pivotal
Trial
Primary Data
Cohort
3/05- 5/07 134 68% 58%
Park, Milano, Tatooles
et al Circ HF 2012;
5:241-248
HM II Pivotal
Trial
Continued
Access
Protocol (CAP)
5/07- 3/09 281 73% 63%



Outpatient Meds Costing Transplant
Medication Dose Wholesale Monthly Cost ($)
Tacrolimus 0.5 mg BID 52.1
Tacrolimus 5 mg BID 515.96
MMF 1500 mg q12 54.42
Prednisone 15 mg q12 4.24
Acyclovir 400 mg q12 33.93
Valcyte 450 mg q12 1181.18
Mepron 1500 mg daily 555.57
Bactrim SS 1 tab daily 2.99
Nystatin susp 5 cc 4 times daily 12.24
Pantoprazole 40 mg daily 2.97
Pravastatin 20 mg HS 2.13
Lasix 20 mg daily 1.68
Lantus 1 box 227.5
Aspart 1 box 250
Norvasc 10 mg daily 2.1
Asa 81 mg daily 1.25
Colace 100 mg tid 3.25
MVT 1 tab daily 1
Yearly Wholesale Cost:
$ 34,854
LVAD
Medication Dose Wholesale Monthly Cost ($)
Amiodarone 200 mg daily 5.25
Lasix 40 mg daily 0.5
Hydraiazine 50 mg tid 15.25
Coumadin 2.5 mg daily 3
Aspirin 325 mg daily 1.25
Toprol XL 50 mg daily 21.5
Famotidine 20 mg bid 3.5
Revatio 20 mg tid 100
Lisinopril 2.5 mg daily 0.75
KCI 20 mEq bid 24.5
Colace 100 mg tid 3.25
MVT 1 tab daily 1
Lantus 1 box 227.5
Aspart 1 box 250
Yearly Wholesale Cost:
$ 7,887

Contraindications for LVAD Implant
age > ?? years
Psychological illness
Irreversible major end-organ failure
Uncertain neurological status
Severe haemodynamic instability
Major bleeding tendency
Sepsis/ prolonged mechanical ventilation
Right heart failure


Limitation of VADs
Availability and Cost
Constant changing of Models / Size / Shape
Blood component damage
Out-Patient and Long Term management
Hypertension / Stroke – Axial pump
Bleeding & Thromboembolism
Durability & mechanical failure
Infection
Peter Houghton
implanted with Jarvik
LVAD . 2000–2008.

Complications of VADs
Right ventricular failure
Bleeding
Infection
Thrombo-embolism
Haemolysis
Device failure
Intra-abdominal cx
(sensitisation with HLA antibodies)


The Future
Small and finite number of heart Tx
LVAD as initial therapy for AHF
LVAD implants will grow rapidly globally
LVAD use in less ill NYHA III patients
LVADs will evolve and improve
• Totally implanted with power supply
• Smaller and lighter
• Reduced risk of stroke and infection
• Biventricular support
Tx will be used as salvage for complications and for
those not eligible for MCS devices


Pagani FD ISHLT 2013


Summary
End Stage Heart Failure is an increasing
burden in developed countries
Heart Tx is No longer the ONLY Gold
Standard therapy for End Stage CHF
Advances in VAD design can replace Heart
Tx in certain patient groups
Cost & effectiveness of VAD support/therapy
may surpass Heart Tx within 10 years

Prof. CK Mok Dr. SW Chiu

Thank You

Visionary that Beyond the Sound Bite


Device Manufacturer Type Approval Status as of July 2009
Novacor World Heart Pulsatile. Was approved for use in North America, European Union
and Japan. Now defunct and no longer supported by
the manufacturer.
HeartMate VXE Thoratec Pulsatile. FDA approval for BTT in 2001 and DT in 2003. CE Mark
Authorized. REMATCH Trial chosen PUMP.
Rarely used anymore due to reliability concerns.
HeatMate II Thoratec Rotor driven continuous
axial flow, ball and
cup bearings.
Approved for use in North America and EU. CE Mark
Authorized. FDA warning 2008 for Lead Failure.
Recently approved by FDA in the US for Destination
Therapy (as at January 2010).
HeartMate III Thoratec Continuous flow driven
by a magnetically
suspended axial
flow rotor.
Clinical trials yet to start, uncertain future.
Incor Berlin Heart Continuous flow driven
by a magnetically
suspended axial
flow rotor.
Approved for use in European Union. Used on
humanitarian approvals on case by case basis in the
US. Entered clinical trials in the US in 2009.
Jarvik 2000 Jarvik Heart Continuous flow, axial
rotor supported by
ceramic bearings.
Currently used in the United States as a bridge to heart
transplant under an FDA-approved clinical investigation.
In Europe, the Jarvik 2000 has earned CE Mark
certification for both bridge-to-transplant and lifetime
use. Child version currently being developed.
MicroMed Debekay
VAD
MicroMed Continuous flow driven
by axial rotor
supported by
ceramic bearings.
Approved for use in the European Union. The child version
is approved by the FDA for use in children in USA.
Undergoing clinical trials in USA for FDA approval.
VentriAssist VentraCor Continuous flow driven
by a
hydrodynamically
suspended
centrifugal rotor.
Approved for use in European Union and Australia.
Company declared bankruptcy while clinical
trials for FDA approval were underway in 2009.

2007 2008 2009 2010 2011 2012 (up to 1/10/12)
Adult 0 1 2 5 3 16
Paed 3 0 1 6 7 7
0
2
4
6
8
10
12
14
16
18
Nu
mb
er
of
Case
ECMO operation during the year from 2007 to Aug. 2012

NUMBER OF HEART TRANSPLANTS
REPORTED BY YEAR
32
2 67
0 1,2
54
2,3
49 2,9
89
3,5
04
3,7
96 4,4
56
4,6
69
4,6
39
4,8
41
4,7
47
4,7
41
4,6
14
4,5
16
4,2
58
3,9
92
3,8
55
3,8
30
3,7
25
3,6
50
3,6
08
3,7
08
3,7
64
3,7
65
3,7
20
3,7
42
18
7
0
500
1000
1500
2000
2500
3000
3500
4000
4500
5000
1982
1983
1984
1985
1986
1987
1988
1989
1990
1991
1992
1993
1994
1995
1996
1997
1998
1999
2000
2001
2002
2003
2004
2005
2006
2007
2008
2009
Nu
mb
er
of
Tra
ns
pla
nts
NOTE: This figure includes only the heart transplants that are
reported to the ISHLT Transplant Registry. As such, the
presented data may not mirror the changes in the number of
heart transplants performed worldwide
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTATION % OF PATIENTS BRIDGED WITH RVAD*
(Transplants: 1/2005 – 12/2009)
0
1
2
3
4
5
6
7
8
9
10
2005 2006 2007 2008 2009
Year
% o
f p
ati
en
ts
* RVAD, RVAD+LVAD ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTATION Kaplan-Meier Survival by Era
(Transplants: 1/1982 – 6/2009)
0
20
40
60
80
100
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15
Years
1982-1991 (N=20,504)
1992-2001 (N=36,879)
2002-6/2009 (N=22,477)
1982-1991 vs. 1992-2001: p = 0.8460
1982-1991 vs. 2002-6/2009: p < 0.0001
1992-2001 vs. 2002-6/2009: p < 0.0001
HALF-LIFE 1982-1991: 10.2 years; 1992-2001: 10.7 years; 2002-6/2009: NA
Su
rviv
al
(%)
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTATION Conditional Kaplan-Meier Survival by Era
(Transplants: 1/1982 – 6/2009)
0
20
40
60
80
100
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15
Years
1982-1991 (N=16,059)
1992-2001 (N=29,287)
2002-6/2009 (N=16,686)
1982-1991 vs. 1992-2001: p = 0.0002
1982-1991 vs. 2002-6/2009: p = 0.0013
1992-2001 vs. 2002-6/2009: p = 0.2327
HALF-LIFE 1982-1991: 13.9 years; 1992-2001: 13.2 years; 2002-6/2009: NA
Su
rviv
al (%
)
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

Heart Tx Alternative
“It’s not an artificial heart, you still got to
have a heart and it's still got to be working."

ADULT HEART TRANSPLANTATION Kaplan-Meier Survival by Age Group
(Transplants: 1/2002-6/2009)
0
20
40
60
80
100
0 1 2 3 4 5 6 7
Years
Su
rviv
al (%
)
18-29 (N=1,860) 30-39 (N=2,223)
40-49 (N=4,214) 50-59 (N=7,902)60-69 (N=6,010) 70+ (N=268)
All pair-wise comparisons are statistically significant at p < 0.05
except 18-29 vs. 60-69; 18-29 vs. 70+, 30-39 vs. 40-49; 30-39 vs. 50-
59 and 60-69 vs. 70+
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANT RECIPIENTS:
Relative Incidence of Leading Causes of Death (Deaths: January 2000 - June 2010)
0
10
20
30
40
50
0-30 Days
(N=1,852)
31 Days –
1 Year
(N=1,814)
>1 Year – 3
Years
(N=1,402)
>3 Years –
5 Years
(N=1,285)
>5 Years –
10 Years
(N=3,802)
>10 – 15
Years
(N=3,138)
>15 Years
(N=1,752)
CAV Acute RejectionMalignancy (non-Lymph/PTLD) Infection (non-CMV)Graft Failure Multiple Organ FailureRenal Failure
Pe
rce
nta
ge
of
De
ath
s
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

Limitation of Cardiac Tx
Donor availability
Immune incompatibility (sensitized)
Conditional half life
Post transplant complications


Device Manufacturer Type Approval Status as of July 2009
Novacor World Heart Pulsatile. Was approved for use in North America, European Union
and Japan. Now defunct and no longer supported by
the manufacturer.
HeartMate VXE Thoratec Pulsatile. FDA approval for BTT in 2001 and DT in 2003. CE Mark
Authorized. REMATCH Trial chosen PUMP.
Rarely used anymore due to reliability concerns.
HeatMate II Thoratec Rotor driven continuous
axial flow, ball and
cup bearings.
Approved for use in North America and EU. CE Mark
Authorized. FDA warning 2008 for Lead Failure.
Recently approved by FDA in the US for Destination
Therapy (as at January 2010).
HeartMate III Thoratec Continuous flow driven
by a magnetically
suspended axial
flow rotor.
Clinical trials yet to start, uncertain future.
Incor Berlin Heart Continuous flow driven
by a magnetically
suspended axial
flow rotor.
Approved for use in European Union. Used on
humanitarian approvals on case by case basis in
the US. Entered clinical trials in the US in 2009.
Jarvik 2000 Jarvik Heart Continuous flow, axial
rotor supported by
ceramic bearings.
Currently used in the United States as a bridge to heart
transplant under an FDA-approved clinical
investigation. In Europe, the Jarvik 2000 has earned
CE Mark certification for both bridge-to-transplant and
lifetime use. Child version currently being developed.
MicroMed
Debekay VAD
MicroMed Continuous flow driven
by axial rotor
supported by
ceramic bearings.
Approved for use in the European Union. The child version
is approved by the FDA for use in children in USA.
Undergoing clinical trials in USA for FDA approval.
VentriAssist VentraCor Continuous flow driven
by a
hydrodynamically
suspended
centrifugal rotor.
Approved for use in European Union and Australia.
Company declared bankruptcy while clinical
trials for FDA approval were underway in 2009.



Transplant Vs VAD:
Evolving & Future
Perspectives
Dr AU Wing Kuk, Timmy Consultant & Chief of Service
Cardiothoracic Surgery Department
Queen Mary Hospital

Bridge to Transplant
Severe reduction in cardiac output such that survival &
success of transplant is unlikely without MCS
Impending cardiogenic shock (despite inotropes + IABP)
resulting in acute renal / liver dysfunction
Pulmonary hypertension (PA>60) that would respond to
prolonged normalization of left atrial pressure
CentriMag versus implantable pump
Fully implantable devices allow for patient discharge
while awaiting donor organ (HeartMate II)

ADULT HEART TRANSPLANTATION Kaplan-Meier Survival by Age Group
(Transplants: 1/1982-6/2009)
0
20
40
60
80
100
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
Years
Su
rviv
al (%
)
18-29 (N=5,580) 30-39 (N=7,898)
40-49 (N=17,714) 50-59 (N=30,967)
60-69 (N=17,187) 70+ (N=514)
HALF-LIFE 18-29: 12.3 years; 30-39: 13.2 years; 40-49: 11.7 years; 50-59: 10.5 years;
60-69: 9.5 years; 70+: 7.5 years
All pair-wise comparisons are
statistically significant at p < 0.02
except 18-29 vs. 40-49 (p=0.2807)
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANT RECIPIENTS:
Cumulative Incidence of Leading Causes of Death (Transplants: January 2000 - June 2009)
0%
2%
4%
6%
8%
10%
0 1 2 3 4 5
Time (years)
CAV Acute Rejection
Malignancy (non-Lymph/PTLD) Graft Failure
CMV Infection (non-CMV)
Inc
ide
nc
e o
f C
au
se
-Sp
ec
ific
De
ath
s
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTS (1/2004-6/2009)
Risk Factors for 1 Year Mortality
Continuous Factors (see figures)
Recipient age
Recipient height
Recipient weight
Donor age
Donor weight
Transplant center volume
Ischemia time
Bilirubin
Serum creatinine
PRA
Pulmonary capillary wedge pressure
Recipient PVR
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTS (1/2004-6/2009)
Factors Not Significant for 1 Year Mortality
Recipient Factors: Prior malignancy, hospitalized, prior
pregnancy, balloon pump, diabetes, gender
Donor Factors: Clinical infection, history of diabetes,
gender, history of hypertension, cause of death, history
of malignancy
Transplant Factors: HLA mismatch, CMV mismatch,
prior transplant
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTS (1/2004-6/2009) Relative Risk of 1 Year Mortality with 95% Confidence Limits
Recipient Age
0
0.5
1
1.5
2
20 25 30 35 40 45 50 55 60 65 70
Recipient Age
p < 0.0001
Re
lati
ve
Ris
k o
f 1
Ye
ar
Mo
rta
lity
(N=10,271) ISHLT 2011 ISHLT
J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTS (1/2004-6/2009) Relative Risk of 1 Year Mortality with 95% Confidence Limits
Donor Age
0
0.5
1
1.5
2
2.5
15 20 25 30 35 40 45 50 55
Donor Age
p < 0.0001
Re
lati
ve
Ris
k o
f 1
Ye
ar
Mo
rta
lity
(N=10,271) ISHLT 2011 ISHLT
J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTS (1/2004-6/2009) Relative Risk of 1 Year Mortality with 95% Confidence Limits
Ischemia Time
0
0.5
1
1.5
2
30 60 90 120 150 180 210 240 270 300 330 360
Ischemia Time (minutes)
p < 0.0001Re
lati
ve
Ris
k o
f 1
Ye
ar
Mo
rta
lity
(N=10,271) ISHLT 2011 ISHLT
J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

AGE DISTRIBUTION OF HEART TRANSPLANT RECIPIENTS BY ERA
0
5
10
15
20
25
30
35
40
0-9 10-19 20-29 30-39 40-49 50-59 60-69 70+
Recipient Age
% o
f tr
an
sp
lan
ts
1982-1991 (N = 23,929)
1992-2001 (N =43,912)
2002-6/2010 (N = 31,398)
p < 0.0001
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

ADULT HEART TRANSPLANTATION Kaplan-Meier Survival by Age Group
(Transplants: 1/1982-6/2009)
0
20
40
60
80
100
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
Years
Su
rviv
al (%
)
18-29 (N=5,580) 30-39 (N=7,898)
40-49 (N=17,714) 50-59 (N=30,967)
60-69 (N=17,187) 70+ (N=514)
HALF-LIFE 18-29: 12.3 years; 30-39: 13.2 years; 40-49: 11.7 years; 50-59: 10.5 years;
60-69: 9.5 years; 70+: 7.5 years
All pair-wise comparisons are
statistically significant at p < 0.02
except 18-29 vs. 40-49 (p=0.2807)
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

AGE DISTRIBUTION OF HEART TRANSPLANT RECIPIENTS BY ERA
0
5
10
15
20
25
30
35
40
0-9 10-19 20-29 30-39 40-49 50-59 60-69 70+
Recipient Age
% o
f tr
an
sp
lan
ts
1982-1991 (N = 23,929)
1992-2001 (N =43,912)
2002-6/2010 (N = 31,398)
p < 0.0001
ISHLT 2011 ISHLT J Heart Lung Transplant. 2011 Oct; 30 (10): 1071-1132

The LVAD Current Status

Device Manufacturer Type Approval Status as of July 2009
Novacor World Heart Pulsatile. Was approved for use in North America, European Union
and Japan. Now defunct and no longer supported by
the manufacturer.
HeartMate VXE Thoratec Pulsatile. FDA approval for BTT in 2001 and DT in 2003. CE Mark
Authorized. REMATCH Trial chosen PUMP.
Rarely used anymore due to reliability concerns.
HeatMate II Thoratec Rotor driven continuous
axial flow, ball and
cup bearings.
Approved for use in North America and EU. CE Mark
Authorized. FDA warning 2008 for Lead Failure.
Recently approved by FDA in the US for Destination
Therapy (as at January 2010).
HeartMate III Thoratec Continuous flow driven
by a magnetically
suspended axial
flow rotor.
Clinical trials yet to start, uncertain future.
Incor Berlin Heart Continuous flow driven
by a magnetically
suspended axial
flow rotor.
Approved for use in European Union. Used on
humanitarian approvals on case by case basis in
the US. Entered clinical trials in the US in 2009.
Jarvik 2000 Jarvik Heart Continuous flow, axial
rotor supported by
ceramic bearings.
Currently used in the United States as a bridge to heart
transplant under an FDA-approved clinical
investigation. In Europe, the Jarvik 2000 has earned
CE Mark certification for both bridge-to-transplant and
lifetime use. Child version currently being developed.
MicroMed
Debekay VAD
MicroMed Continuous flow driven
by axial rotor
supported by
ceramic bearings.
Approved for use in the European Union. The child version
is approved by the FDA for use in children in USA.
Undergoing clinical trials in USA for FDA approval.
VentriAssist VentraCor Continuous flow driven
by a
hydrodynamically
suspended
centrifugal rotor.
Approved for use in European Union and Australia.
Company declared bankruptcy while clinical
trials for FDA approval were underway in 2009.

The Importance of RV Function

Evaluating RV Dysfunction

LVAD Limitations
Percutaneous power supply
Risk of Stroke
Risk of Infection
Risk of Bleeding
Requirement for full sternotomy ???
Risk of mechanical failure
• Pump thrombosis
• Lead Fracture
• Controller malfunction

Summary of DT Outcomes
Reference Study Enrollment
period
n One-Year Survival
Slaughter, Rogers, Milano et al N
NEJM 2009;361:2241-51
HM II Pivotal Trial 3/05- 5/07 133 68%
Slaughter MS, presented at ISHLT
2010
HM II Pivotal Trial 5/07- 3/09 252 74%

INTERMACS Profiles & Timing of VAD Implant
Description Time Frame
1 Patient with life-threatening hypotension despite rapidly escalating inotropic
support, critical organ hypoperfusion with increasing lactate levels and/or systemic
acidosis. “Crash and burn”
within hours
2 Patient with declining function despite IV inotropic support, may be manifest
by worsening renal function, nutritional depletion, inability to restore volume
balance. “Sliding on inotropes”
few days
3 Patient with stable blood pressure, organ function, nutrition, and symptoms on IV
inotropic support, but cannot wean off inotropes. “Dependent stability”
Elective over a
few weeks
4 Patient can be stabilized close to normal volume status but with relapses of CHF, &
fluid retention. Intensive management strategies . “Frequent flyer”
Elective over
weeks to months
5
Patient is comfortable at rest without congestive symptoms, but may have
underlying refractory elevated volume status, often with renal dysfunction.
“Housebound”
Variable, depends
upon nutrition,
organ function,
and activity
6 Patient without evidence of fluid overload is comfortable at rest and with activities,
but fatigues after the first minutes of any meaningful activity. “Walking wounded”
Variable, depends
upon nutrition,
organ function,
and activity
7 A placeholder for future specification, patients without recent unstable fluid balance,
living comfortably with meaningful activity limited to mild exertion. Not indicated