Familial adenomatous polyposis Best Practice Guidelines.
-
Upload
angelina-jeffries -
Category
Documents
-
view
240 -
download
5
Transcript of Familial adenomatous polyposis Best Practice Guidelines.

Familial adenomatous polyposis
Best Practice Guidelines

Lynch: Cancer:100, No1,2004
Contribution of familial cancer syndromes to colorectal cancer

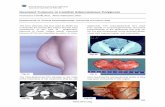
Familial adenomatous polyposis - FAP
Thousands of polyps in colon
Certain to become malignant by fourth decade
Prevention of cancer depends on regular sigmoidoscopy
Molecular testing can be used to indicate those requiring clinical screening

Extra colonic manifestations in FAP
Congenital hypertrophy of the retinal pigment epithelium
Desmoid tumour
Osteomas
Epidermoid cysts
Upper GI adenomas

APC gene associated with FAP

Nature of APC mutations
Hundreds identified Mutations are nonsense, small deletions
or insertions (~80%) Large deletions (7-12%) “All” mutations lead to a truncated
protein product and hence identifiable as pathogenic
Pick up rate for mutations is > 90%

Distribution of APC mutations
0
10
20
30
40
50
60
70
80
3 4 5 6 7 8 9 10 11 12 13 14
15.2
15.3
15.4
15.4
Exon
nu
mb
er

Specific mutations
5bp deletions at codons 1308 and 1061 account for ~5% of cases
Attenuated disease associated with 5’ and 3’ mutations and also with mutations in exon 9
p.Ile1307Lys mutation found in approximately 6% of the Ashkenazi Jewish population and has been associated with increased risk of colon cancer Predictive testing not recommended
p.Glu1317Gln – probably not now associated with colon cancer


FAP- summary Clinically identifiable condition Caused by mutations in one gene Effect of mutations is clear High pick up rate for mutations Effective clinical screening test for those at high
risk Some evidence of genotype-phenotype
correlations though of little value clinically

BP GuidelinesMutation analysis MLPA for deletions/duplications Either mutation scanning approach or
direct sequencing PTT for exon 15?
~66% of mutations Linked markers
Intragenic and flanking markers available When should these be used?
Cytogenetics?

BP Guidelines Reporting
Identification of a clearly pathogenic mutation Confirms a diagnosis of FAP Offer presymptomatic testing to relatives
At what age?

BP Guidelines Reporting
No mutation identified Need to describe sensitivity of testing
carried out Cannot completely rule out FAP Offer linkage if clinical diagnosis is
sufficiently sound

BP Guidelines Reporting
“(this patient) was found to carry the APC p.Ile1307Lys/p.Glu1317Gln variant. No other variants were detected. This variant was formerly considered a predisposition allele for colorectal cancer, however, morerecent papers [cite ref(s)] indicate that there is no statistically or clinically significant association between carrying the variant and increased risk of colorectal cancer. Detection of this variant does not confirm a diagnosis of FAP and predictive testing for this variant is not indicated in (this patient’s) relatives.”
• Identification of p.Ile1307Lys/p.Glu 1317Gln

BP Guidelines Reporting
Other missense variant or synonymous change Ensure that change does not affect splicing Report according to BP unclassified variant
guidelines Are these relevant at all as all known
mutations causing FAP are truncating?

BP Guidelines Reporting
Presymptomatic testing If mutation present patient is highly likely to
develop FAP APC mutations are “100%” penetrant
If absent won’t develop FAP and is a population risk of colon cancer

Prenatal testing
Is there a role for it? Prenatal PGD?