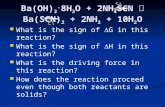
Equilibrium Follow-up. Equilibrium Problems The Haber process proceeds as follows: 2NH 3 (g) + 92KJ...
-
Upload
chandler-burriss -
Category
Documents
-
view
219 -
download
1
Transcript of Equilibrium Follow-up. Equilibrium Problems The Haber process proceeds as follows: 2NH 3 (g) + 92KJ...

Equilibrium Follow-up

Equilibrium Problems• The Haber process proceeds as follows:
2NH3 (g) + 92KJ ↔ N2 (g) + 3H2 (g)
If the equilibrium concentrations are:[NH3] = 3.1x10-2M
[N2] = 8.5x10-1M
[H2] = 3.1x10-3M
Then what is the value for K?Is the forward reaction favored?How can we increase the amount of product?

Answer• K = [H2]3[N2]
[NH3]2
K = (3.1x10-3)3 (8.5x10-1)(3.1x10-2)2
K = 2.6x10-5
-No it is not favored, the K value demonstrates that the reactant is more likely to be present at equilibrium than the products.-Product can be increased by increasing the temperature, decreasing the pressure, increasing the amount of NH3, or removing H2 or N2.

Kp And Kc
• In the reaction at 25oC :2NO(g) + Cl2(g) ↔ 2NOCl (g)
The equilibrium pressures were found to be:PNOCl = 1.2atm
PNO = 5.0x10-2atm
PCl2 = 3.0x10-1atm
• What is the Kp and Kc for this reaction?

Answer• Kp = PNOCl
2
(PNO)2(PCl2)
Kp = 1.22
(5.0x10-2)2(3.0x10-1)Kp = 1.9x103
Kp = Kc (RT)∆n ∆n = 2-3 = -1
So KP = Kc / RT or Kc = Kp (RT)
Kc = 1.9x103 (0.0821)(298)
= 4.6x104

The Reaction Quotient• It is possible to measure the concentrations of the
reactants and products in a reaction vessel at any time.
• They may not necessarily be at equilibrium yet.• When plugging those values into the Law of Mass
Action, you will still get a value, but it might not be K.
• We call this value the reaction quotient, and comparing this value to K, we can see which direction the reaction is favored to move.

You try!• Haber again!• N2 (g) + 3H2 (g) ↔ 2NH3 (g) • Give the concentrations:• [NH3] = 1.0x10-3M
• [N2] = 1.0x10-5M
• [H2] = 2.0x10-3M• Calculate the value for Q and predict which
direction the equilibrium will shift if the K is 6.0x10-2.

Answer• Q = (1.0x10-3)2
(1.0x10-5)(2.0x10-3)3
• Q = 1.3x107
Since K = 6.0x10-2 and Q is significantly higher, then the concentrations of the products needs to go down, and reactants up meaning the reaction will shift left. N2 (g) + 3H2 (g) ← 2NH3 (g)

Calculating Equilibrium Pressures• Lets start easy:• In the reaction N2O4 ↔ 2NO2 has a Kp value of 0.133.
If the pressure of N2O4 at equilibrium is 2.71atm, then what is the Equilibrium pressure of NO2?
• Tougher:• A 1.00L flask initially contained 0.298mol of PCl3 and
8.70x10-3 mol of PCl5. After the system reached equilibrium, 2.00x10-3 mol of Cl2 was found. Calculate K(c) and the Equilibrium concentrations.
• PCl5 ↔ PCl3 + Cl2

Answers
• Kp = PNO22/PN2O4
• 0.133 = PNO22/ 2.71
• PNO22 = 0.360
• PNO2 = 0.600

Answer Part 2• K = [Cl2][PCl3]/[PCl5]• Concentrations must be found first.• [Cl2]o = 0
• [PCl3]o = 0.298mol/1L = 0.298M
• [PCl5]o = 8.70x10-3M• Next we find the change to reach equilibrium.• ∆[Cl2] = +2.00x10-3M• Since they all have coefficients of 1, then• ∆[PCl3] = +2.00x10-3M
• ∆[PCl5] = -2.00x10-3M

Part 2 finish
• Final concentrations of gases:• [Cl2] = 2x10-3M
• [PCl3] = 0.300M
• [PCl5] = 6.70x10-3M• K = (2x10-3)(0.300)/(6.70x10-3)• K = 8.96x10-2

ICE• By knowing starting concentrations and K values,
it is possible to determine the concentrations once equilibrium is reached.
• It is based on the stoichiometry of the problem.• “I” stands for initial concentration• “C” stands for change in concentration• “E” stands for equilibrium concentration

ICE problem• For the reaction:• CO + H2O ↔ CO2 + H2O• The K value is 5.10. Calculate the equilibrium
concentrations if the initial concentrations of all components is 1M.

Answer[CO] [H2O] [CO2] [H2]
Initial 1M 1M 1M 1M
Change -x -x +x +x
Equilibrium 1M-x 1M-x 1M+x 1M+x
5.10 = (1+X)(1+X)/((1-X)(1-X))5.10 = (1+X)2/(1-X)2
2.26 = (1+X)/(1-X)X = 0.387M
[CO],[H2O] = 0.613M [CO2],[H2] = 1.387M

You try!
• H2 + F2 ↔ 2HF• If the equilibrium Constant is 1.15x102 then if
3.00 moles of each substance were added to a 1.5L flask, what will the equilibrium concentrations be?

answer• Must find Q first to know which way reaction
will shift.• Q = [HF]2/[H2][F2]• All are 2M gas solutions so • Q = 22/(2)(2) = 1• Since Q is less then K reaction shifts right.
[H2] [F2] [HF]
I 2M 2M 2M
C -x -x +2x
E 2M-x 2M-x 2M + 2x

Answer finish
• X = 1.528 so:• [H2] = [F2] = 0.472M• [HF] = 5.056M

Small K values
• If K is very small, and concentrations of substances are large, then the change is insignificant.
• X can be dropped at that point from the change of the reactants which makes the problem easier to solve.

You Try
• 2NOCl(g) ↔ 2NO(g) + Cl2(g)
• If the K is 1.6x10-5, what will the equilibrium concentrations be if 1.0mol of NOCl is added to an empty 2.0L flask?

Answer• Initial concentration of NOCl is 1mol/2L or 0.5M.
2NOCl ↔ 2NO + Cl2
K = [NO]2[Cl2]/[NOCl]2
1.6x10-5= (2x)2(x)/(0.5-2x)2
Since K is to the -5 and the starting concentration is 4 powers of ten greater, then we can ignore the change in the reactant concentration.
[NOCl] [NO] [Cl2]
I 0.5 0 0
C -2x +2x +x
E 0.5-2x +2x +x

Answer Cont.• So the equations becomes:1.6x10-5 = 4x3/(0.5)2
X = 1x10-2
As long as the change in concentration is smaller than 5%, it is valid to ignore the change.Plugging the change into the law of mass action and demonstrating that the K hasn’t changed is also a valid way to check.So the final concentrations are [NOCl] = 0.5M, [NO] = 2.0x10-2M and [Cl2] = 1.0x10-2M

One More time with Haber!• N2 (g) + 3H2 (g) ↔ 2NH3 (g)
• If the equilibrium concentrations of N2, H2, and NH3 were .399M, 1.197M, and 0.202M respectively, then what would the new equilibrium expression be under the same conditions if enough nitrogen gas was added to the system in order to raise the concentration of nitrogen to 1.399M?

Answer• First use the given equilibrium concentrations to
find the value for K.• K = 5.96x10-2
• Clearly since N2 was added, then the reaction will shift right.
• 5.96x10-2 = (0.202-2x)2/((1.399-x)(1.197-3x)3)
[N2] [H2] [NH3]
I 1.399 1.197 0.202
C -x -3x +2x
E 1.399-x 1.197-3x 0.202+2x
![Preparation and Characterization of Cyano Complexes of ...downloads.hindawi.com/journals/jchem/2010/378561.pdf · Preparation of (Ph 3P) 2NH 2[WO(CN) 3L–L].3H 2O (Ph 3P) 2NH 2[WO(CN)](https://static.fdocuments.us/doc/165x107/60893efe64f7142ce10196ee/preparation-and-characterization-of-cyano-complexes-of-preparation-of-ph-3p.jpg)













![. Cdculate the equilibrium constant, K, for the following reacton at 25 cc, 1--12 (g) + 120 2 HI (g) if the equilibrium concerffabons are [1-12] = 0.106 M,](https://static.fdocuments.us/doc/165x107/5af4b31c7f8b9a9e598d14a6/-cdculate-the-equilibrium-constant-k-for-the-following-reacton-at-25-cc-1-12.jpg)

