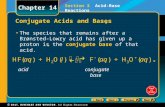
Entry Task: Jan 25 th Friday Question: Identify the acid, base and conjugate acid and conjugate base...
-
Upload
layla-hardick -
Category
Documents
-
view
236 -
download
0
Transcript of Entry Task: Jan 25 th Friday Question: Identify the acid, base and conjugate acid and conjugate base...

Entry Task: Jan 25th Friday
Question: Identify the acid, base and conjugate acid and conjugate base when nitric acid reacts with sodium hydroxide.
You have 5 minutes

Agenda:
• Discuss Acid Base properties & pH ws.• HW: Ch. 16 sec. 5-7 reading notes

I can…
• Distinguish an acid from a base by its properties.
• Explain the difference theories of acid and base behavior of an Arrhenius A/B, Bronsted-Lowry A/B and Lewis A/B.
• Use the concentrations of acids and bases to calculate pH and pOH.

Label the following as A- acid or B- base____ 1. KOH____ 2. covalently bonded____ 3. Red with litmus paper____ 4. negative charged solution____ 5. Hydrogen ions____ 6. pH 8-14____ 7. Reacts to carbonates____ 8. positive charged solutions____ 9. Reacts to metals____ 10. blue with litmus paper____ 11. H2SO4
____ 12. H in front of its formula____ 13. Hydroxide ions____ 14. Does not react with carbonates____ 15. pH 0-6____ 16. nonmetal-nonmetal____ 17. Does not react with metals____ 18. metal-nonmetal ____ 19. OH on the back of its formula____ 20. ionically bonded
BAABABAAABAABBAABBBB

Provide the name or formula the following acids and basesHF Phosphoric acidH2SO3 Carbonic acidZn(OH)2 Barium hydroxideKOH Sulfuric acidFe(OH)2 Hydrocyanic acid
Hydrofluoric acid H3PO4
Sulfurous acid H2CO3
Zinc II hydroxide Ba(OH)2
Potassium hydroxide H2SO4
Iron II hydroxide HCN

Identify the B-L acid, base, c-acid, and c-base for the followingH2SO4 + NH3 HSO4
- + NH4+
NaHCO3 + HCl NaCl + H2CO3
CO32– + HC2H3O2 C2H3O2
– + HCO3–
Acid C-baseBase C-acid
Acid C-baseBase C-acid
Acid C-baseBase C-acid

Identify the B-L acid, base, c-acid, and c-base for the following
HC2H3O2 + H2O H3O+ + C2H3O2-
HF + SO32– F– + HSO3
–
H3O+ + HSO3– H2O + H2SO3
Acid C-baseBase C-acid
Acid C-baseBase C-acid
Acid C-baseBase C-acid

Give the conjugate base for each of the following Brønsted-Lowry acids
1. HI
2. NH4+
3. H2CO3
4. HNO3
I-
NH3
HCO3-
NO3-

Give the conjugate acid for each of the following Brønsted-Lowry bases.
1. CN–
2. O2–
3. CH3COO–
4. NH3
HCN
OH-
CH3COOH
NH4+

What is the difference between a strong acid and a weak acid?
Strong acids completely ionize in water making them strong conductors of electricity.
Weak acids partially ionize in water making them weak conductors of electricity.

Exercise Questions 16.8Designate the BL acid and BL base on the left side of each reaction and its conjugates on the right. HCHO2 + H2O CHO2
- + H3O+
HSO4
- + CO32- SO4
2- + HCO3 –
H3O+ + HPO4
2- H2PO4- + H2O
Acid C-baseBase C-acid
Acid C-baseBase C-acid
Acid C-baseBase C-acid

Exercise Questions 16.11Label each of the following as being a strong acid, weak acid or a species with negligible acidity AND provide its conjugate baseHNO2 ________________________________H2SO4 _______________________________HPO4 2-______________________________CH4_________________________________CH3NH3
+_____________________________
Weak Acid
Weak acidStrong Acid
negligibleWeak acid
NO2+1
HSO4+1
PO4+3
CH3+1
CH3NH2+2

Exercise Questions 16.16Predict the products of the following acid-base reactions also predict whether the equilibrium lies to the right or left. NH2 + H2O
HClO2 + H2O
H3O+ + F-
NH3+ + OH-
Which scenario would likely happen?
Water is a stronger acid than OH- so the equilibrium will shift to the product
ClO2- + H3O+
HClO2 is a stronger acid than H3O+ so the equilibrium will shift to the product
H2O + HFHF is a stronger acid than F- so the equilibrium will
shift to the reactant

Exercise Questions 16.19Calculate [H+] for each of the following solutions and indicate whether the solution is acidic, basic or neutral: SHOW WORK!!
[OH-] = 0.00005M
[OH-] = 3.2 x10-9
Solution with 100 times greater than [H+]
[H+] = 1.0x 10-14
[OH-][H+] = 1.0x 10-14
[0.00005][H+]= 2.0 x 10-10 this is a
basic solution
[H+] = 1.0x 10-14
[3.2 x10-9][H+]= 3.1x 10-6 this is a
acidic solution
[H+] = 1.0x 10-14
[100] [1.0 x10-7][H+]= 1.0 x 10-9 this is a
basic solution

Exercise Questions 16.25 If NaOH is added to water, how does [H+] change?
If [H+] = 0.0003M. What is the pH?
If pH is 7.8, what is the molar concentration of H+ and OH- in solution?
NaOH + H2O Na+ + OH- + H3O+
This just dilute the solution doing nothing to the H+ level
pH = -log (0.0003) = pH = -(-3.52) = 3.52
[H+]= 1.6 x10-8
[H+] = -7.8

[H+] [OH-] pH pOH Acid/Base
7.5 x 10-3M
3.6 x 10-10M
8.25
5.70
Exercise Questions 16.27Fill in the missing information
1.3 x10-12 11.92.1 Acid
9.42.8 x10-5 4.6 Acid
5.6 x10-9 1.8 x10-6 5.7 Base
2.0 x10-65.0 x10-9 8.3 Base

Exercise Questions 16.28Fill in the missing information
[H+] [OH-] pH pOH Acid/Base
5.15
7.85
1.8 x 10-6M
9.2 x 10-12M
7.1 x10-6 8.85 Acid1.4 x10-9
1.4 x10-87.1 x10-7 6.15 Acid
5.6 x10-9 8.255.74 Acid
1.1 x10-3 2.96 11.0 Acid


















![pH = -log [H + ]. Strong Acid. Strong base. Weak Acid. Weak Base. “Conjugate Acid – Base Pair” a proton donor & its corresponding proton.](https://static.fdocuments.us/doc/165x107/56649e215503460f94b0d171/-ph-log-h-strong-acid-strong-base-weak-acid-.jpg)

