Electron Orbital Angular Momentum and Spin- Orbit Coupling in
Electron Orbital Diagrams and Electron Configurations Labeling Electrons – Part 2 Supplement to...
-
Upload
damon-jefferson -
Category
Documents
-
view
219 -
download
0
description
Transcript of Electron Orbital Diagrams and Electron Configurations Labeling Electrons – Part 2 Supplement to...

Electron Orbital Diagrams and Electron Configurations
Labeling Electrons – Part 2Supplement to Book – Glencoe: Chapter 19-3

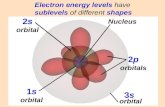
I. Orbitals in Energy Levels (L)
This is the 2nd quantum number The shape of the periodic table is a direct
result of the order in which electrons fill the energy sublevels and orbitals.

I. Orbitals in Energy Levels (L)
All electrons are found in specific orbitals within each energy level
Orbital – very specific space in which there is a high probability of finding an electron, basically a subshell within an energy level
There are four types of orbitals named by their shape

“s” – spherical like a ball
"s" orbital pictures
Click link above to see “s” orbitals
s-block = Groups 1 and 2, plus helium

“p” – dumbbell kind of like dumbbell weights
"p" orbital pictures
Click link above to see “p” orbitals
p-block = Groups 13 through 18, excluding helium

“d” – complex multiple combinations of shapes
"d" orbital pictures
Click link above to see “d” orbitals
d-block = Groups 3-12

“f” – complex multiple combinations of shapes
"f" orbital pictures
Click link above to see “f” orbitals
f-block = Elements #58-71 and #90-103 (pulled out at the bottom)

B. Each type of orbital has a specific number that exist
1. There is only 1 “s” orbital in each energy level, starting in the first energy level (n=1).
2. There are only 3 “p” orbitals in each energy level, starting with the second energy level (n =2).
3. There are only 5 “d” orbitals in each energy level, starting with the third energy level (n=3).
4. There are only 7 “f” orbitals in each energy level, starting with the fourth energy level (n=4).

C. “L” numbers
Each orbital shape has a “L” number associated with it:
s = 0 p = 1 d = 2f = 3

D. How many electrons fit?
Any single orbital may only hold 2 electrons maximum
“Rule of cups” When the cup is full, it will overflow
Can I sit down?
Dude, the couch is
full.
Hello McFly.

II. Electron Orbital Diagrams
A. Shows the locations of electrons in specific orbitals in specific energy levels
B. Electrons are represented as arrows
C. Orbitals are represented at a labeled line or box, in a stair step pattern
D. Electrons fill orbitals from lowest energy to highest energy, one electron at a time

II. Electron Orbital Diagrams
Examples: Hydrogen, Boron, Titanium Remember “Rule of Urinals”

Rule of Urinals
You never pee beside you buddy unless you have too!
If given a choice, you always choose a new, empty urinal.

III. Electron configurations
A. Electron configurations take the electron orbital diagram and condenses it down to letters and numbers to represent the location of electrons within the atom.

III. Electron configurations
Examples: Hydrogen, Boron, Titanium