Chapter 4: Section 1. The Atom is the smallest particle of an element.
Definitions Atom: smallest particle of an element that can exist and still have the properties of...
-
Upload
edwina-hancock -
Category
Documents
-
view
223 -
download
4
Transcript of Definitions Atom: smallest particle of an element that can exist and still have the properties of...

Definitions
• Atom: smallest particle of an element that can exist and still have the properties of the element.
• Compound: A pure substance that can be broken down into two or more simpler pure substances by chemical means
• Molecule: group of two or more atoms that function as a unit because the atoms are tightly bound together

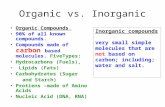
• MOLECULAR COMPOUNDS
• All atoms are nonmetals• Electrons are shared to
form a bond
• IONIC COMPOUNDS• Metal and a non metal• Electrons are
transferred to form cations and anions

Ionic vs molecular compounds

Molecular orCovalent bond
ValenceElectrons areshared

Homonuclear molecules (diatoms)and Heteronuclear molecules(polyatomic
molecules)

Homonuclear molecules
HONFIBrClI never fly high over brown clouds

Molecule is the smallest particle of a compound capable of a stable, independent
existence
Phosphorus exists in a stable form of P-P-P-P
Sulfur exists in a stable form of S-S-S-S-S-S-S-S
What would be an easier way to write these symbols?

Compounds
• H2O # H ____, # O ___
• C6H12O6 # C______, # H _____, # O____
• Mg(OH)2 # Mg_____, # H_____, # O____
• Ca10(PO4)6(OH)10
• 2 H2O #H in each molecule? _____________

Tell whether the following have ionic or covalent bonds
• CaCl2 MgO
• CO2 NH4Cl
• H2O HCl
• BaSO4 KI
• K2O NaOH
• NaF NO2
• Na2CO3 AlPO4
• CH4 FeCl3
• SO2 P2O5
• LiBr N2O5

Naming Molecular compounds
• Binary compounds (not hydrocarbons)• Two nonmetals• Shared electrons• Name ends in ide• No ions formed• Use Greek prefixes• Mono*, di, tri, tetra, penta, hexa, hepta, octa,
nona, deca

Naming molecular compounds
formula nameP2O5
Dinitrogen trioxideNO
Sulfur trioxidePI3
SCl6
Silicon dioxide

Lewis structures
• Atoms combine to form molecules so they will have lower energy
• Using only valence electrons try and give each atom a complete valence shell (complete octet except Hydrogen)
• Then try and give them the number of bonds they want : called covalency

Lewis structuresgroup 1 simple structures
Formula Lewis structureNH3
H2O
N2H4
CH4O
PF3

Lewis structuresgroup 2: polyatomic ions
formula Lewis structure
OH-
NH4+
NO3-

Lewis structuresgroup 3: multiple bonds
Formula Lewis structure
H2CO
HCN
CO
CO2

Molecular geometryis not shown by Lewis structures

VSEPR
• Valence Shell Electron Pair Repulsion








Give the molecular geometry and bond angles of each structure