Comparative Growth Behaviour and Leaf Nutrient Status of Native Trees Planted on Mine Spoil With and...
-
Upload
arvind-singh -
Category
Documents
-
view
214 -
download
0
Transcript of Comparative Growth Behaviour and Leaf Nutrient Status of Native Trees Planted on Mine Spoil With and...

Annals of Botany 87: 777±787, 2001doi:10.1006/anbo.2001.1414, available online at http://www.idealibrary.com on
Comparative Growth Behaviour and Leaf Nutrient Status of Native Trees Planted onMineSpoil With and Without Nutrient Amendment
ARVIND SINGH and J. S. SINGH*
Department of Botany, Banaras Hindu University, Varanasi 221 005, India
Received: 7 November 2000 Returned for revision: 13 December 2000 Accepted: 23 February 2001
trees raised
0305-7364/0
* For corrbanaras.ernet
The e�ect of nutrient amendment on growth of nine indigenous tree species planted on coal mine spoil was studied.Greater growth in fertilized plots was accompanied by greater foliar N and P concentrations in all species. Theresponse to fertilization varied among species and was greater in non-leguminous than in leguminous species.Furthermore, leguminous species exhibited higher growth rates compared to non-leguminous species. The log-transformed height-diameter relationships were signi®cant for all tree species and treatments. Acacia catechu,Dalbergia sissoo, Gmelina arborea and Azadirachta indica ®tted the elastic similarity model of tree growth; whereasPongamia pinnata and Phyllanthus emblica followed the constant stress model. Tectona grandis was the only specieswhich ®tted the geometric similarity model. In Albizia lebbeck and Terminalia bellirica, the b-values (the gradients ofthe log-transformed height to diameter relationships) were considerably lower (50.5), and these two species did not®t any model of tree growth. In several cases, the b-values were considerably in¯uenced by nutrient amendment. Thelog-transformed crown mass and trunk mass relationships were signi®cant for all treatments and species. The slope ofthe crown mass:trunk mass relationship was near unity in A. indica, D. sissoo, G. arborea, P. emblica, P. pinnata,T. grandis, and T. bellirica. However, in A. catechu and A. lebbeck, this slope was well below unity suggesting a greaterallocation to non-photosynthetic tissue. Fertilizer amendment resulted in a heavier crown relative to trunk in A. indica,T. grandis and T. bellirica. # 2001 Annals of Botany Company
Key words: Diameter increment, fertilizer application, foliar N, foliar P, height increment, tree growth, volume
increment.growth on mine spoil?
INTRODUCTION
During mining, overburden material (spoil) overlying thecoal seam is often removed and dumped in a haphazardmanner. Mine spoil is nutritionally and microbiologicallypoor (Wali, 1975; Singh and Jha, 1993), and it needs to bestabilized to prevent erosion and contamination of riversand adjoining agricultural lands from harmful leachates.Natural restoration is a slow process (Iverson and Wali,1982; Jha and Singh, 1991, 1992), but it can be acceleratedby planting trees and herbaceous species. This two-tieredvegetation increases the biological fertility and biodiversityof the mine spoil (JS Singh et al., 1996). To overcome theproblem of nutritional de®ciency, fertilizers are oftenapplied. Several studies have demonstrated that fertilizationpromotes establishment and increases biomass productionof herbaceous species (Schoenholtz et al., 1992; Piha et al.,1995; A Singh et al., 1996). However, fertilization of woodyspecies planted on mined land has a variable e�ect ongrowth (Vogel, 1981). A number of fertilization studies onnutrient-poor habitats other than mine spoil have suggestedincreased growth rates in woody species (Vitousek et al.,1987, 1993; Gerrish et al., 1988; Tanner et al., 1990, 1992;Raich et al., 1996; Vitousek and Farrington, 1997).
In the absence of shelter from existing vegetation, young
on mine spoil face pronounced wind stress.1/060777+11 $35.00/00
espondence. Fax (91) 542 368174, e-mail [email protected]
Architectural traits are important for wind stability(McMahon, 1973; King, 1986; Niklas, 1992). Trees di�erin relative allocation of resources to supporting vs.photosynthetic tissues, and thus has implications forgrowth rates and the ability to resist buckling or breakingunder wind loads (King, 1981; Niklas, 1992; O'Brien et al.,1995). Nutrient amendments are likely to in¯uence theallocation patterns and hence the wind stability.
The objective of the present study was to evaluate thein¯uence of nutrient amendment on growth of nine nativetree species planted on mine spoil. The following questionswere addressed: (1) do woody species di�er in the degree oftheir response to fertilization of mine spoil; (2) doleguminous species and non-leguminous species di�er intheir response; (3) does fertilization alter tree architecturaltraits; and (4) does leaf nutrient status act as an indicator of
MATERIALS AND METHODS
Site description
The study was conducted at the Jayant coal mine in theSingrauli Coal®elds, India. Singrauli extends over 2200 km2
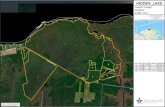
(238470±248120 N, 818480±828520 E and elevations of 280±519 m above mean sea level), of which 80 km2 lie in UttarPradesh and the rest in Madhya Pradesh (Fig. 1). Theclimate is tropical monsoonal and the year is divisible
into a mild winter (November±February), a hot summer# 2001 Annals of Botany Company

INDIA
N
82° 35' 82° 45'
82° 35' 82° 45'
24°
10'
24°
10'
TOOBRA
Revegetation site
Coal mines
Road
MOHER
AMLOHRI
NIGAHI
KHADIA MARRAK
BINA
TO RENUKOOT
JHINGURDA
M.P. U.P.
KAKRI
DUDHICHUAJAYANT
GORBI
FIG. 1. Location of study site (revegetation site) within Jayant coal mine. Jayant coal mine is one of the 11 coal mine blocks of the Singraulira
778 Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil
(April±June) and a warm rainy season (July±September). March and October are transitional months.Mean monthly minimum temperature within an annualcycle ranges from 6.4 to 28 8C and mean monthlymaximum from 20 to 42 8C. The annual rainfall averages1069 mm, 90% of which occurs during the periodbetween June and September. The texture of the spoilmaterial was 80% sand, 10% silt, and 10% clay, with apH of 7.4, total N 0.018% and total P 0.01% (Singh,1999). Soil cores to a depth of 10 cm consisted of 75% ofparticles greater than 2 mm in diameter. The potential
Coal®elds. M.P., Madhya P
natural vegetation is a tropical dry deciduous forest.
Experimental design and methods
Nursery-raised 1-year-old individuals of the followingnine tree species were planted on fresh ¯at mine spoil in July1993: Albizia lebbeck, Acacia catechu, Azadirachta indica,Dalbergia sissoo, Gmelina arborea, Phyllanthus emblica,Pongamia pinnata, Tectona grandis and Terminalia bellirica.All species are native, and are natural components of a dry
tropical deciduous forest. The seedlings were planted in20 m � 20 m plots with a spacing of 2 m � 2 m. Plots wereseparated from each other by a 5 m strip. Fertilizertreatment was applied annually in July in 1994±1996, andconsisted of a full dose of NPK, a half dose of NPK, and acontrol without fertilizer amendment. Full dose fertilizertreatment was 60 kg haÿ1 N as urea, 30 kg haÿ1 P as singlesuper phosphate, and 40 kg haÿ1 K as muriate of potash.Urea and single super phosphate were applied in granularform and muriate of potash in powder form. Three replicateplots were maintained for each treatment. Thus, at the rateof 7 kg haÿ1 for all nine species there was a total of 81plots. From July 1994, the grass Pennisetum pedicellatumwas seeded in 20 cm wide belts between rows of trees toavoid root competition with trees. The aim behind grassseeding was to increase organic matter input into the minespoil. Besides this, grass cover would serve as a mulch,nurse crop, and a trap for wind-blown seeds and othermaterials. Furthermore, ground cover decreases leachinglosses of nutrients in young plantations (McLaughlin et al.,1985).
A total of 15 individuals for each species, distributed
desh; U.P., Uttar Pradesh.
equally between the three replicate plots, and selected

for signi®cance through a two-tailed Student's t-test.
a
at random avoiding edge-rows, was used for growthmeasurements for each treatment. Height and diametermeasurements were made in April 1996 (33 months afterplantation) and in December 1997 (53 months afterplantation). Diameter (d) was measured at 20 cm abovethe ground surface. Height (h) was measured using a scaledbamboo stick. During the second sampling (53 monthsafter plantation) crown diameter and area were alsomeasured. Crown area (CA) was estimated asCA � ��Cd1 � Cd2�=4�2p, where Cd1 and Cd2 are crowndiameters at two perpendicular axes beneath the canopy atground surface.
Volumes of trees (V) were calculated as a cone (V � d 2h).Two architectural traits, viz. slopes (b) of the relationshipsbetween the log-transformed height and diameter values,and between log-transformed crown mass and trunk massvalues were determined through regression analysis. Har-vesting of trees from the restoration site was prohibited,hence we used proxy variables for crown mass and trunkmass. For a variety of tropical trees, ranging from under-storey to emergent, the square of canopy area is nearly equalto the crown mass and d 2h approximates the trunk mass(O'Brien et al., 1995). Therefore, in our study, the square ofcrown area served as a proxy variable for crown mass.
Mature, healthy leaves were collected from exposed, mid-canopy positions of the tagged trees in September 1997,dried at 80 8C and powdered for chemical analysis. FoliageN was estimated with the microkjeldahl method of Jackson(1958). Phosphorus was analysed after digestion in amixture of HClO4 , HNO3 and H2SO4 (1 :5 :1), using thephosphomolybdic acid blue method of Jackson (1958).
Tree growth was assessed as increments in height,diameter and volume from the values measured in April1996 and December 1997. Annual increments of the abovevariables were calculated from the di�erences between thetwo measurements. The in¯uence of height and diameterattained by the trees at the ®rst sampling (33 months after
Singh and SinghÐE�ect of Fertiliz
planting) on subsequent annual growth, and the in¯uence
TABLE 1. Summary of ANOVA on height diameter and volumefor nine tree species grown with t
Source of variation d.f.
Hei
F
DimensionSpecies 8 120.07Sampling date 1 563.48Treatment 2 59.48Species � sampling date 8 7.25Species � treatment 16 4.41Sampling date � treatment 2 2.68Species � sampling date � treatment 16 0.47Residual 756
Growth ratesSpecies 8 132.14Treatment 2 48.94Species � treatment 16 8.66Residual 378
of leaf nutrient status on annual growth were exploredthrough regression analyses.
Data from individual trees were subjected to analysis ofvariance using the General Linear Model of the SPSSpackage (SPSS/PC, 1993). The overall restoration plan forthe site did not permit mixing of plots of di�erent species ina block. Therefore, the plots of the nine species were in e�ectblocks, and the treatments were randomized within theblocks. Because of this, plot was not included as a source ofvariation in the analysis and 15 individuals were treated asreplicates. Di�erences between treatment means were tested
tion on Tree Growth on Mine Spoil 779
RESULTS
Height, diameter and volume
Analysis of variance revealed signi®cant e�ects of treatmentand sampling date on height, diameter and tree volume.Species � treatment and species � sampling date inter-actions were signi®cant for height, diameter and volume,but the sampling date � treatment interaction was signi®-cant only for volume (Table 1).
Leguminous species. The leguminous trees in fertilizedplots were 7±15% taller than control trees on the ®rst samp-ling date (33 months after plantation) and 4±16% tallerthan controls on the second sampling date (53 months afterplantation) (Table 2). Compared to controls, the treediameters were 4±27% greater at the ®rst and 4±31%greater at the second sampling in fertilized plots. A. catechudid not show a signi®cant response to fertilization, whereasthe full NPK dose had a signi®cant e�ect on diameter andvolume of D. sissoo at the ®rst sampling only. In A. lebbeck,full NPK treatment had a signi®cant e�ect on height,diameter and volume. In P. pinnata, diameter and volumewere signi®cantly increased by full NPK treatment. Half-strength NPK treatment had no signi®cant e�ect on any of
the species., and growth rates (height, diameter and volume increments)wo levels of NPK and a control
ght Diameter Volume
P F P F P
0.000 58.76 0.000 60.46 0.0000.000 251.36 0.000 250.20 0.0000.000 74.34 0.000 53.10 0.0000.000 3.07 0.002 14.41 0.0000.000 2.43 0.001 6.05 0.0000.069 2.87 0.057 12.34 0.0000.959 0.28 0.998 2.23 0.004
0.000 75.74 0.000 46.15 0.0000.000 70.81 0.000 39.53 0.0000.000 6.98 0.000 7.15 0.000

TABLE 2. Height, diameter, volume and crown cover for nine tree species of two di�erent ages grown on mine spoil with twolevels of NPK and a control
Species Treatment
33 months after plantation 53 months after plantation
Height (m)Diameter
(cm)Tree volume(d 2h) (cm3) Height (m)
Diameter(cm)
Tree volume(d 2h) (cm3)
Crowncover (m2)
LegumesAcacia catechu Control 2.49a 5.14a 8460a 3.74a 6.89a 22 825a 4.58a
Half NPK 2.74a 5.68a 9727a 4.33a 8.34a 33 086a 5.64a
Full NPK 2.73a 5.50a 9699a 4.21a 7.81a 30 092a 5.87a
Albizia lebbeck Control 3.24a 6.01a 13 024a 4.32a 7.66a 28 107a 5.91a
Half NPK 3.46ab 6.73ab 18 504ab 4.58ab 8.78ab 41 664ab 5.93a
Full NPK 3.74b 7.66b 24 061b 5.01b 10.01b 54 964b 7.15a
Dalbergia sissoo Control 3.21a 4.84a 7985a 4.67a 6.48a 20 320a 7.30a
Half NPK 3.67a 5.03ab 10 081ab 5.3a 6.73a 26 060a 8.08a
Full NPK 3.42a 5.65b 12 171b 4.85a 7.42a 29 310a 8.69a
Pongamia pinnata Control 2.67a 4.11a 4919a 3.92a 5.63a 13 389a 5.57a
Half NPK 2.92a 4.49a 6468ab 4.09a 6.23ab 17 098a 6.69a
Full NPK 2.94a 5.92b 8877b 4.24a 7.55b 25 685b 7.30a
Non-legumesAzadirachta indica Control 2.47a 4.44a 5304a 3.23a 5.86a 11 903a 3.85a
Half NPK 2.95b 5.42b 9180b 3.97b 7.11b 20 660b 5.60b
Full NPK 3.05b 6.23b 13 433b 4.05b 7.88b 27 823b 6.74b
Gmelina arborea Control 3.05a 6.28a 15 799a 4.08a 8.35a 36 754a 7.19a
Half NPK 3.15a 6.90a 17 137a 4.55a 9.91a 49 950a 7.69a
Full NPK 4.03b 8.59b 31 533b 6.05b 12.52b 99 741b 11.26b
Phyllanthus emblica Control 2.21a 3.49a 3505a 2.77a 4.41a 6995a 4.74a
Half NPK 2.54b 4.63b 6050a 3.35b 6.17b 12 152ab 6.42b
Full NPK 2.48ab 4.72b 6644a 3.25ab 6.21b 14 963b 6.63b
Tectona grandis Control 1.63a 3.67a 2486a 2.13a 4.41a 4677a 1.18a
Half NPK 2.07b 4.22a 4207b 2.8b 5.12ab 8336b 1.85b
Full NPK 2.45b 5.09b 7728c 3.17b 6.17b 14 737b 2.69b
Terminalia bellirica Control 1.70a 4.27a 3484a 2.25a 5.53a 7656a 1.48a
Half NPK 1.92ab 4.66a 4741a 2.52ab 5.96a 10 137a 1.73ab
Full NPK 2.07b 5.77b 7462b 2.78b 7.96ab 18 860b 2.0b
Values in a column for each species su�xed with di�erent letters are signi®cantly di�erent from each other at P 5 0.05.
780 Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil
Non-leguminous species. Trees in fertilized plots were 3±50% taller at the ®rst sampling and 12±49% taller at thesecond sampling compared to controls. In fertilizeramended plots the diameters were 9±40% greater at the®rst sampling and 8±50% greater than controls at thesecond sampling. The full NPK treatment had a signi®cantimpact on height, diameter and volume in all non-leguminous species (Table 2). The e�ect of half-strengthNPK treatment was signi®cant on height, diameter andvolume in A. indica, on height and volume in T. grandis,
and on height and diameter in P. emblica.reduced the slope whereas the full NPK dose increased it.
Height :diameter relationships
Leguminous species. The log-transformed height:dia-meter relationships were signi®cant for all three nutrienttreatments in leguminous tree species for both samplingperiods (Table 3), and slopes (b-values) were consistentbetween the sampling periods. In P. pinnata the slope wasaround 0.50, in D. sissoo and A. catechu it was nearer to0.67, and in A. lebbeck it was less than 0.50. Compared tocontrols, application of half-strength NPK resulted in
increased b-values for all the leguminous species exceptP. pinnata. In this species full strength NPK resulted in anincrease in b-value from around 0.5 to 0.7.
Non-leguminous species. The log-transformed height :dia-meter relationships were signi®cant for all three treatmentsin all non-leguminous species (Table 3). The b-values incontrol trees were 0.67 for A. indica and G. arborea,between 0.4 and 0.57 for T. bellirica and P. emblica, andalmost 1.0 for T. grandis. Application of NPK resulted in adecrease in b-values in A. indica, G. arborea and P. emblica.But in T. bellirica, fertilizer treatment increased the slopefrom 0.4 to around 1. In T. grandis, half-strength NPK
Crown mass : trunk mass relationships
Leguminous species. The log-transformed crown massand trunk mass relationships were signi®cant for all threenutrient treatments in all leguminous species (Table 4). Theslope of the relationship increased from control to full NPKtreatment in A. catechu, A. lebbeck and D. sissoo. InP. pinnata, the slope was greater in the full NPK treatment
than in half-strength NPK as compared to the control.
TABLE3.Regressionoflog10height(m)andlog10diameter
(cm
)forninetree
speciesoftwodi�erentages
grownonminespoilunder
twolevelsofNPKandacontrol
Species
Treatment
33monthsafterplantation
53monthsafterplantation
Intercept
Slope
r2P
Intercept
Slope
r2P
Legumes
Acaciacatechu
Control
ÿ0. 070+
0. 16
0. 605+
0. 10
0. 73
0. 000
0. 145+
0. 19
0. 612+
0. 10
0. 74
0. 000
HalfNPK
ÿ0. 070+
0. 25
0. 619+
0. 15
0. 57
0. 000
0. 170+
0. 31
0. 609+
0. 14
0. 56
0. 001
FullNPK
ÿ0. 224+
0. 32
0. 717+
0. 19
0. 52
0. 002
ÿ0. 011+
0. 37
0. 702+
0. 18
0. 53
0. 002
Albizia
lebbeck
Control
0. 522+
0. 18
0. 365+
0. 10
0. 49
0. 003
0. 741+
0. 20
0. 355+
0. 10
0. 47
0. 004
HalfNPK
0. 310+
0. 16
0. 493+
0. 09
0. 71
0. 000
0. 470+
0. 18
0. 487+
0. 08
0. 71
0. 000
FullNPK
0. 350+
0. 22
0. 477+
0. 11
0. 59
0. 000
0. 530+
0. 11
0. 470+
0. 11
0. 57
0. 001
Dalbergia
sissoo
Control
ÿ0. 010+
0. 23
0. 745+
0. 14
0. 67
0. 000
0. 168+
0. 26
0. 734+
0. 13
0. 68
0. 000
HalfNPK
ÿ4. 480+
0. 34
0. 800+
0. 21
0. 53
0. 002
0. 118+
0. 39
0. 807+
0. 20
0. 53
0. 001
FullNPK
ÿ0. 075+
0. 29
0. 751+
0. 17
0. 60
0. 000
0. 089+
0. 33
0. 742+
0. 16
0. 60
0. 000
Pongamia
pinnata
Control
0. 255+
0. 13
0. 517+
0. 09
0. 70
0. 000
0. 495+
0. 16
0. 506+
0. 09
0. 68
0. 000
HalfNPK
0. 355+
0. 15
0. 478+
0. 10
0. 62
0. 000
0. 561+
0. 19
0. 464+
0. 11
0. 60
0. 000
FullNPK
ÿ0. 130+
0. 21
0. 726+
0. 13
0. 72
0. 000
ÿ0. 049+
0. 26
0. 739+
0. 12
0. 71
0. 000
Non-legumes
Azadirachta
indica
Control
ÿ0. 038+
0. 22
0. 632+
0. 15
0. 57
0. 001
0. 0641+
0. 26
0. 626+
0. 15
0. 57
0. 001
HalfNPK
0. 104+
0. 27
0. 578+
0. 16
0. 50
0. 003
0. 266+
0. 31
0. 567+
0. 17
0. 48
0. 003
FullNPK
0. 069+
0. 23
0. 572+
0. 12
0. 61
0. 000
0. 228+
0. 25
0. 571+
0. 12
0. 61
0. 000
Gmelinaarborea
Control
ÿ0. 212+
0. 27
0. 720+
0. 15
0. 65
0. 000
ÿ0. 155+
0. 31
0. 733+
0. 15
0. 64
0. 000
HalfNPK
ÿ4. 066+
0. 18
0. 598+
0. 09
0. 75
0. 000
0. 153+
0. 22
0. 595+
0. 09
0. 74
0. 000
FullNPK
0. 606+
0. 19
0. 367+
0. 09
0. 56
0. 001
0. 897+
0. 23
0. 358+
0. 09
0. 54
0. 001
Phyllanthusem
blica
Control
0. 091+
0. 11
0. 568+
0. 09
0. 75
0. 000
0. 201+
0. 13
0. 556+
0. 10
0. 73
0. 000
HalfNPK
0. 108+
0. 18
0. 539+
0. 11
0. 62
0. 000
0. 232+
0. 21
0. 537+
0. 11
0. 62
0. 000
FullNPK
0. 111+
0. 16
0. 518+
0. 10
0. 65
0. 000
0. 277+
0. 17
0. 496+
0. 12
0. 66
0. 000
Tectonagrandis
Control
ÿ0. 698+
0. 26
0. 910+
0. 20
0. 60
0. 000
ÿ0. 592+
0. 29
0. 903+
0. 20
0. 60
0. 000
HalfNPK
ÿ0. 377+
0. 17
0. 768+
0. 12
0. 76
0. 000
ÿ0. 214+
0. 19
0. 762+
0. 12
0. 75
0. 000
FullNPK
ÿ0. 721+
0. 21
0. 989+
0. 13
0. 81
0. 000
ÿ0. 623+
0. 24
0. 972+
0. 14
0. 80
0. 000
Terminaliabellirica
Control
ÿ0. 054+
0. 14
0. 406+
0. 10
0. 57
0. 001
0. 110+
0. 16
0. 410+
0. 10
0. 57
0. 001
HalfNPK
ÿ0. 786+
0. 20
0. 932+
0. 13
0. 79
0. 000
ÿ0. 751+
0. 23
0. 935+
0. 13
0. 79
0. 000
FullNPK
ÿ0. 938+
0. 35
0. 948+
0. 20
0. 63
0. 000
ÿ0. 962+
0. 42
0. 954+
0. 20
0. 62
0. 000
Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil 781

TABLE 4. Regression of crown mass (proxy variable: log10 crown area in m2) to trunk mass [proxy variable: (d2 in cm2)(h in cm)] for nine tree species grown on mine spoil under two levels of NPK and a control
Treatment Intercept Slope r2 P
LegumesAcacia catechuControl ÿ4.83+ 1.16 0.79+ 0.12 0.77 0.000Half NPK ÿ7.47+ 2.02 1.05+ 0.19 0.69 0.000Full NPK ÿ7.84+ 1.8 1.11+ 0.17 0.75 0.000
Albizia lebbeckControl ÿ3.23+ 1.55 0.67+ 0.15 0.59 0.000Half NPK ÿ4.87+ 1.41 0.80+ 0.13 0.73 0.000Full NPK ÿ6.63+ 1.63 0.97+ 0.15 0.76 0.000
Dalbergia sissooControl ÿ4.94+ 0.29 0.89+ 0.29 0.42 0.009Half NPK ÿ7.07+ 2.19 1.10+ 0.21 0.66 0.000Full NPK ÿ7.93+ 2.67 1.19+ 0.26 0.61 0.000
Pongamia pinnataControl ÿ5.85+ 2.58 0.98+ 0.27 0.49 0.003Half NPK ÿ3.34+ 2.94 0.73+ 0.30 0.30 0.032Full NPK ÿ9.82+ 2.96 1.35+ 0.29 0.62 0.000
Non-legumesAzadirachta indicaControl ÿ8.19+ 2.13 1.16+ 0.22 0.66 0.000Half NPK ÿ10.91+ 1.77 1.44+ 0.18 0.82 0.000Full NPK ÿ10.42+ 1.22 1.39+ 0.12 0.91 0.000
Gmelina arboreaControl ÿ6.99+ 1.81 1.05+ 0.17 0.73 0.000Half NPK ÿ5.79+ 1.91 0.91+ 0.18 0.66 0.000Full NPK ÿ5.03+ 2.12 0.86+ 0.18 0.62 0.000
Phyllanthus emblicaControl ÿ4.55+ 1.24 0.88+ 0.14 0.74 0.000Half NPK ÿ2.78+ 2.38 0.68+ 0.25 0.36 0.018Full NPK ÿ5.06+ 1.22 0.93+ 0.13 0.79 0.000
Tectona grandisControl ÿ9.61+ 1.42 1.18+ 0.17 0.79 0.000Half NPK ÿ10.29+ 1.44 1.28+ 0.16 0.83 0.000Full NPK ÿ12.26+ 1.34 1.49+ 0.14 0.89 0.000
Terminalia belliricaControl ÿ6.54+ 1.71 0.84+ 0.19 0.59 0.000Half NPK ÿ11.21+ 1.42 1.34+ 0.15 0.85 0.000Full NPK ÿ13.18+ 2.22 1.48+ 0.23 0.75 0.000
782 Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil
Non-leguminous species. The log-transformed crown massand trunk mass relationships were signi®cant for all threenutrient treatments in all non-leguminous species (Table 4).The slope of the relationship increased from control to fullNPK treatment in T. grandis and T. bellirica, whereas itdecreased from control to full NPK treatment in G. arborea.In A. indica, fertilizer application resulted in an increase inslope but the value was only slightly higher in half-strengthNPK compared to full NPK treatment. In P. emblica, theslope was greater in full NPK but lower in half NPK in
comparison to the control.signi®cant e�ect on foliar P concentration in all species.
Foliar N and P concentrationsLeguminous species. The foliar N and P concentrationsincreased from control to full NPK treatment in allleguminous species (Table 5). The e�ect of both NPKtreatments was signi®cant on foliar N concentration inA. lebbeck and D. sissoo, whereas in A. catechu andP. pinnata the e�ect of half-strength NPK was not
signi®cant. The e�ect of both NPK doses was signi®canton foliar P concentration in all leguminous species exceptfor D. sissoo, whereas half-strength NPK treatment did nothave a signi®cant e�ect and di�erences between half-strength NPK and full NPK treatments were generallynot signi®cant.
Non-leguminous species. Foliar N and P concentrationsincreased from control to full NPK treatment in all non-leguminous species (Table 5). The e�ect of both half- andfull-strength NPK treatments was signi®cant on foliar Nconcentration in all species. Application of half-strengthNPK fertilizer did not have a signi®cant e�ect on foliar Pconcentration in any species, whereas full NPK dose had a
Tree growth
Analysis of variance indicated a signi®cant e�ect oftreatment on height, diameter and volume growth (annualincrements). The species � treatment interaction was also
signi®cant for height, diameter and volume growth
(no e�ect on T. bellirica).
TABLE 5. N and P concentrations in mature foliage for ninetree species grown on mine spoil under two levels of NPK and
a control
Species Treatment N (%) P (%)
LegumesA. catechu Control 2.19a 0.134a
Half NPK 2.28ab 0.178b
Full NPK 2.34b 0.196b
A. lebbeck Control 2.05a 0.143a
Half NPK 2.13b 0.183b
Full NPK 2.19b 0.201b
D. sissoo Control 1.95a 0.160a
Half NPK 2.04b 0.183ab
Full NPK 2.12c 0.205b
P. pinnata Control 2.14a 0.138a
Half NPK 2.20ab 0.183b
Full NPK 2.27b 0.196b
Non-legumesA. indica Control 1.76a 0.165a
Half NPK 1.88b 0.178ab
Full NPK 1.98b 0.196b
G. arborea Control 1.82a 0.165a
Half NPK 1.91b 0.174ab
Full NPK 2.05c 0.192b
P. emblica Control 1.20a 0.111a
Half NPK 1.31b 0.134ab
Full NPK 1.39c 0.147b
T. grandis Control 1.01a 0.103a
Half NPK 1.19b 0.125ab
Full NPK 1.27b 0.134b
T. bellirica Control 1.10a 0.107a
Half NPK 1.25b 0.129ab
Full NPK 1.33b 0.138b
Values in a column for each species su�xed with di�erent letters aresigni®cantly di�erent from each other at P 5 0.05.
TABLE 6. Annual height, diameter and volume incrementsper tree species grown on mine spoil under two levels of NPK
and a control
Species TreatmentHeight(m)
Diameter(cm)
Volume(cm3)
LegumesA. catechu Control 0.74a 1.05a 8618a
Half NPK 0.95b 1.60b 14 016a
Full NPK 0.89ab 1.38b 12 236a
A. lebbeck Control 0.64a 0.98a 9020a
Half NPK 0.65a 1.22b 13 825ab
Full NPK 0.76b 1.4b 18518b
D. sissoo Control 0.87a 0.98a 7514a
Half NPK 0.98a 1.01a 9587a
Full NPK 0.86a 1.06a 10 404a
P. pinnata Control 0.75a 0.91a 5116a
Half NPK 0.70a 1.04a 6467a
Full NPK 0.78a 1.51b 10 149b
Non-LegumesA. indica Control 0.45a 0.85a 4004a
Half NPK 0.60b 1.01b 7012b
Full NPK 0.58b 0.93ab 8583b
G. arborea Control 0.61a 1.24a 12 525a
Half NPK 0.83b 1.8b 18 642a
Full NPK 1.19c 2.35b 40 930b
P. emblica Control 0.32a 0.55a 2086a
Half NPK 0.47b 0.92b 4756b
Full NPK 0.44b 0.89b 4967b
T. grandis Control 0.29a 0.44a 1314a
Half NPK 0.43b 0.53b 2478b
Full NPK 0.43b 0.65b 4205b
T. bellirica Control 0.32a 0.76a 2519a
Half NPK 0.35a 0.77a 3237a
Full NPK 0.42b 1.3b 6871b
Values in a column for each species su�xed with di�erent letters are.
Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil 783
suggesting that the e�ect of nutrient amendment dependedupon the species (Table 1).
Leguminous species. The height growth among legumes incontrol treatments ranged from 0.64 to 0.87 m per year( �x � 0.75 m per year per tree), diameter growth from 0.91to 1.05 cm per year ( �x � 0.98 cm per year per tree) andvolume increment from 5116 to 9020 cm3 per year( �x � 7567 cm3 per year per tree) (Table 6). Half-strengthNPK treatment increased the annual height increment by1.6±28.4%, diameter increment by 3.10±52.4% andvolume increment by 26.4±62.6%, whereas full NPKtreatment increased the height increment by 4.0±20.3%,the diameter increment by 8.2±65.9% and the volumeincrement by 38.5±105.3% over controls. Height, diameterand volume increments in D. sissoo, height increment inP. pinnata and volume increment in A. catechu were notsigni®cantly a�ected by fertilization. In A. lebbeck, half-strength NPK treatment did not have a signi®cant e�ect onheight and volume increments but signi®cantly a�ected thediameter increment. In P. pinnata, only full NPK treatmenthad a signi®cant e�ect on diameter and volume increments.Only half-strength NPK treatment had a signi®cant e�ecton height increment in A. catechu, whereas both levels of
NPK had a signi®cant e�ect on diameter increment.Non-leguminous species. The height increment amongnon-legumes in the control treatment ranged from 0.29 to0.61 m per year ( �x � 0.39 m per year per tree), diameterincrement from 0.44 to 1.24 cm per year ( �x � 0.76 cm peryear per tree) and volume increment from 1314 to12 525 cm3 per year ( �x � 4490 cm3 per year per tree).Compared to controls, the height increment was 9.4±48.3%greater, the diameter increment 1.3±67.3% greater and thevolume increment 28.5±128.0% greater under half-strengthNPK treatment. Full NPK treatment increased the heightincrement by 29.9±95.0%, the diameter increment by 9.4±89.5% and the volume increment by 114.4±226.8% overcontrols (Table 6). The full NPK treatment had a signi®-cant e�ect on height, diameter and volume increments in allthe non-leguminous species (except for diameter incrementin A. indica). Height, diameter and volume increments inP. emblica and T. grandis, height and diameter incrementsin G. arborea and height and volume increments in A. indicawere signi®cantly a�ected by half-strength NPK treatment
signi®cantly di�erent from each other at P5 0 05.
E�ect of tree dimensions on subsequent growth
Signi®cant positive relationships occurred between height
and subsequent height growth and between diameter and
0.5
1.0
1.5
0.01 2 3
Height (m)
4 5
Heightincrement(m
peryearpertree)
B Fertilized
1
2
3
03
Diameter (cm)
6 9 12
Diameterincrement(cm
peryearpertree)
D Fertilized
0.5
1.0
1.5
0.01 2 3 4 5
Heightincrement(m
peryearpertree)
A Control
1
2
3
01 2 3 4 5
Diameterincrement(cm
peryearpertree)
C Control
FIG. 2. Relationships between initial height (33 months after plantation) and subsequent annual height increment, and between initial diameterand subsequent annual diameter increment for trees planted on mine spoil with and without fertilizer ( full dose) application. Regression equationsare (A) y � 0.113 � 0.177x (r2 � 0.33, P 5 0.01); (B) y � 0.353 � 0.119x (r2 � 0.121, P 5 0.01); (C) y � 0.688 � 0.038x (r2 � 0.033, P 5 0.05);and (D) y � 1.134 � 0.021x (r2 � 0.005, P 4 0.05). Data are for 135 (nine species � 15 individuals) trees each from control and fertilized ( full
dose) plots.
784 Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil
subsequent diameter increment among trees in unfertilizedplots across all species (Fig. 2). These relationships wereweaker or not signi®cant for plants receiving the full NPK
dose (Fig. 2).Relationships between leaf nutrient status and tree growth
Height, diameter and volume increments had signi®cantpositive relationships with foliar N and P concentrations
(Fig. 3).DISCUSSION
Self supporting plants that allocate too little biomass tostems may buckle under their own mass or break, due to
wind stress or other loads (O'Brien et al., 1995). Treearchitecture (height-diameter, crown mass-trunk relation-ships) is therefore of considerable importance in selectingspecies for plantation on mine spoils. The present speciesdi�ered considerably in architectural traits, which were alsoin¯uenced by nutrient amendment. The slope (G) of log-transformed height :diameter relationships at the ®rstsampling varied from 0.365 to 0.989 and for the secondsampling from 0.355 to 0.972, compared to the range 0.597to 0.736 recorded for mature trees of eight neotropical treespecies of Barro Colorado Island, Panama (O'Brien et al.,1995). The present analysis suggested that the legumesA. catechu and D. sissoo and the non-legumes A. indica andG. arborea followed the elastic similarity model of treegrowth.
According to Rich (1986), when the scaling exponent b
between diameter and height is equal to 0.67, then the tree
15000
30000
45000
00.8 1.6
Leaf N (%)
2.4
Volumeincrement
(cm
3peryearpertree)
C
0.08 0.16
Leaf P (%)
0.2
F
0.8
1.6
2.4
Diameterincrement
(cm
peryearpertree)
B E
0.5
1.0
1.5
Heightincrement
(mperyearpertree)
A D
FIG. 3. Relationships between leaf nutrient status and growth parameters for trees planted on mine spoil. Regression equations are (A)y � ÿ1.73 � 0.554x (r2 � 0.697, P 5 0.01); (B) y � 0.027 � 0.587x (r2 � 0.376, P 5 0.01); (C) y � ÿ7823.003 � 9530.92x (r2 � 0.273, P 50.01); (D) y � ÿ2.55 � 5.625x (r2 � 0.554, P 5 0.01); (E) y � ÿ0.31 � 8.725x (r2 � 0.431, P 5 0.01); and (F) y � ÿ14 958 � 152 040x (r2 �
0.360, P 5 0.01). Data are treatment means for the nine species (n � 9 � 3 � 27).
Singh and SinghÐE�ect of Fertilization on Tree Growth on Mine Spoil 785
is said to maintain elastic similarity through ontogeny.Large arborescent growth forms usually follow an elasticsimilarity model (Norberg, 1988). An alternative model oftree growth is constant stress, where b � 0.5. This model isbased on the assumption that trunk taper is such that stressproduced by wind pressure along the stem is equalized(Dean and Long, 1986). The legume P. pinnata and thenon-legume P. emblica were found to follow this constantstress model of growth. According to Niklas (1992), the
constant stress model is the most generally applicable modelin a windy habitat. A third model of geometric similarity,where b � 1.0, presumes that the proportions of a treeremain constant through ontogeny. Smaller growth forms( from mosses to small trees) are best described by thismodel (Norberg, 1988). T. grandis was the only tree specieswhich exhibited a geometric similarity model.
In A. lebbeck and T. bellirica, b-values were 50.5, ®ttingnone of the above three models of tree growth. These twospecies were shorter relative to their diameter suggesting a
higher margin of safety against buckling than other species.
at
Nutrient enrichment appears to in¯uence the height-diameter relationship considerably in some species. Forexample, in T. bellirica b increased from 50.5 (constantstress) to around 1.0 (geometric similarity) and inG. arboreab declined from around 0.7 (elastic similarity) to 50.5.
As crown mass to trunk mass ratio is expected to remainconstant during tree growth, the slope of regression betweenthe proxy variables representing crown mass and trunkmass should not be signi®cantly di�erent from unity(O'Brien et al., 1995). In this study the slope value wasaround unity in A. indica, D. sissoo, G. arborea, P. emblica,T. grandis and T. bellirica suggesting a constant crown massto trunk mass ratio. In A. catechu and A. lebbeck, however,the slope value was well below unity suggesting that in thesespecies allocation favoured trunk mass over crown mass.These two species may be highly resistant to wind actionbut at the cost of photosynthetic tissue. Application ofNPK increased the slope value towards unity in A. catechuand A. lebbeck and beyond unity in A. indica, T. grandis andT. bellirica. Thus fertilizer amendment resulted in heaviercrowns in these species. Hulm and Killham (1990) observeda similar pattern following fertilizer application in Sitkaspruce. Trees with heavier crowns will be highly prone towind action, particularly on a loose substratum such asmine spoil. In D. sissoo and G. arborea, fertilizer amend-ment did not cause any major shift in allocation betweensupport and photosynthetic systems.
Average height, diameter and volume increments forleguminous species were greater than those for non-leguminous species, suggesting that the leguminous specieshave a greater capacity for growth in nutrient-poorhabitats. This may be due to the nitrogen ®xing ability ofleguminous species. Our study also suggested that the foliarN concentration in leguminous species was greater thanthat of non-leguminous species.
Fertilization of mine spoil promoted growth in all ninetree species, but the magnitude of the e�ect varied amongspecies and ranged from non-signi®cant to highly signi®-cant. The e�ect of fertilization on growth rates for height,diameter and volume was greatest in G. arborea and least inD. sissoo. Rates of growth under full NPK treatment inG. arborea were two±three times that of the control. Tanneret al. (1992) reported that trunk growth rate doubledfollowing fertilizer application for all tree species in aVenezuelan montane forest. Studies indicate that N, inparticular, limits production early in soil development(Vitousek et al., 1993) and that greatest growth occurs inplots receiving both N and P (Raich et al., 1996). Severalother studies have also suggested that the diameter growthis greater in fertilized plants compared to control plants(Gerrish and Bridges, 1984; Tanner et al., 1990, 1992;Herbert and Fownes, 1995; Vitousek and Farrington,1997). Hulm and Killham (1990) reported enhanced growthin trees of Sitka spruce fertilized with urea, as compared tocontrols over two growing seasons. In our study, theresponse to NPK fertilization was greater in non-leguminous than in leguminous species. Nitrogen ®xingspecies may overcome nitrogen de®ciency by virtue of their
786 Singh and SinghÐE�ect of Fertiliz
N2-®xing ability, hence they may remain una�ected by
N fertilization but they may respond to P fertilization(McMaster et al., 1982).
A large degree of variation occurred in tree volume (d 2h),which frequently ®gures as a proxy variable for biomass(Zavitkovski and Stevens, 1972; DeBell et al., 1989). In thisstudy, this variability was greater among legumes than non-legumes. Taking all species into consideration, full strengthNPK increased tree volume by 15±211% at the ®rstsampling and from 32 to 215% at the second yearsampling. Nambiar and Fife (1987) found that 2 yearsafter planting, stem volume of fertilized Pinus radiata treeswas 71% greater than that of control trees, and by 3.8 yearsof age, stem volume and biomass of N fertilized trees weretwice those of controls. In A. catechu and G. arborea, thegrowth response to fertilization for height, diameter andvolume was greater at the second compared to the ®rstsampling, suggesting an age interaction in growth response.Similarly, Nommik and Moller (1981) observed a maxi-mum growth response to fertilization 5 years after fertilizeramendment in Scots pine.
The greater growth in fertilized plots was accompaniedby increased N and P concentrations in the foliage. In ourstudy there was a positive relationship between growth rates(height, diameter and volume increments) and foliar N andP concentrations. Evidently, higher leaf nutrient statusincreases photosynthetic e�ciency, particularly in trees onnutrient-poor soils. N supply can a�ect plant growth andproductivity by altering both leaf area and photosyntheticcapacity (Sinclair, 1990; Frederick and Camberato, 1995).The photosynthetic capacity of leaves is reported to berelated to their N content primarily because enzymes of theCalvin cycle represent the majority of leaf N (Evans, 1989).Fahey et al. (1998) reported an increase in foliar nutrientconcentrations (N, P and K) in ®ve northern hardwoodforest tree species in response to NPK fertilization. Ourstudy clearly indicated that the tree species with lower foliarN concentration responded more to N fertilization in termsof increase in foliar N concentration compared to thosewith a higher foliar N concentration. A similar trend wasobserved for P concentration. This suggests that response offoliar N and P concentrations to fertilization depends onthe initial foliar nutrient status of the plants.
We found positive relationships between initial heightand the subsequent height increment, and initial diameterand the subsequent diameter increment for trees on controlplots. This demonstrates that plants that are able to attaingreater height and diameter initially will continue to growmore rapidly. The application of full dose NPK, however,weakened these relationships, suggesting that fertilizationtended to bring more uniformity among the growth ofindividuals (i.e. it eliminated the nutrient poverty ofsuppressed individuals). A positive relationship betweenheight and height increment has also been observed insaplings of Banksia grandis (Abbott, 1985).
We conclude that nutrient amendment promoted growthin all the tree species studied, increased N and P con-centrations in the foliage, and tended to bring moreuniformity among the growth of individuals, by eliminatingnutrient poverty of suppressed individuals. However, the
ion on Tree Growth on Mine Spoil
response to fertilizer application varied between species.

a
The species selected for this study also varied with respectto architectural traits and hence wind stability which wasa�ected by nutrient perturbation. Fertilizer amendmentmight make those trees with high crown mass:trunk massratios (e.g. Azadirachta indica, Tectona grandis and
Singh and SinghÐE�ect of Fertiliz
Terminalia bellirica) susceptible to wind load.
LITERATURE CITED
Abbott I. 1985. Rate of growth of Banksia grandis Willd. (Proteaceae)in western Australian forest. Australian Journal of Botany 33:381±391.
Dean TJ, Long JN. 1986. Validity of constant-stress and elastic-instability principles of stem formation in Pinus contorta andTrifolium pratense. Annals of Botany 58: 833±840.
DeBell DS, Whitesell CD, Schubert TH. 1989. Using N2-®xing Albiziato increase growth of Eucalyptus plantations in Hawaii. ForestScience 35: 64±75.
Evans JR. 1989. Photosynthesis and nitrogen relationships in leaves ofC3 plants. Oecologia 78: 9±19.
Fahey TJ, Battles JJ, Wilson GF. 1998. Responses of early successionalnorthern hardwood forests to changes in nutrient availability.Ecological Monographs 68: 183±212.
Frederick JR, Camberato JJ. 1995. Water and nitrogen e�ects onwinter-wheat in the south-eastern coastal plain. II. Physiologicalresponses. Agronomy Journal 87: 527±533.
Gerrish G, Bridges KW. 1984. A thinning and fertilizer experiment inMetrosideros dieback stands in Hawaii. Hawaii Botanical SciencePaper 43: 1±107.
Gerrish G, Mueller-Dombois D, Bridges KW. 1988. Nutrient limitationand Metrosideros dieback in Hawaii. Ecology 69: 723±727.
Herbert DA, Fownes JH. 1995. Phosphorus limitation of forest leafarea and net primary productivity on highly weathered tropicalmontane soils in Hawaii. Biogeochemistry 29: 223±235.
Hulm SC, Killham K. 1990. Response over two growing seasons of aSitka spruce stand to 15N-urea fertilizer. Plant and Soil 124:65±72.
Iverson LR, Wali MK. 1982. Reclamation of coal mined lands: the roleof Kochia scoparia and other pioneers in early succession.Reclamation and Revegetation Research 1: 123±160.
Jackson ML. 1958. Soil chemical analysis. Englewood Cli�s, NJ:Prentice-Hall.
Jha AK, Singh JS. 1991. Spoil characteristics and vegetationdevelopment of an age series of mine spoils in a dry tropicalenvironment. Vegetatio 97: 63±76.
Jha AK, Singh JS. 1992. In¯uence of microsites on redevelopment ofvegetation on coal mine spoils in a dry tropical environment.Journal of Environmental Management 36: 95±116.
King DA. 1981. Tree dimensions, maximizing the rate of height growthin dense stands. Oecologia 51: 351±356.
King DA. 1986. Tree form, height growth, and susceptibility to winddamage in Acer saccharum. Ecology 67: 980±990.
McLaughlin RA, Pope PE, Hansen EA. 1985.Nitrogen fertilization andground cover in a hybrid poplar plantation: e�ects on nitrateleaching. Journal of Environmental Quality 14: 241±245.
McMahon TA. 1973. Size and shape in biology. Science 179:1201±1204.
McMaster GS, Jow WM, Kummerow J. 1982. Response of Adenostomafasciculatum and Ceanothus greggii chaparral to nutrientadditions. Journal of Ecology 70: 745±756.
Nambiar EKS, Fife DN. 1987. Growth and nutrient retranslocation inneedles of radiata pine in relation to nitrogen supply. Annals ofBotany 60: 147±156.
Niklas KJ. 1992. Plant biomechanics, an engineering approach to plant
form and function. Chicago, IL: University of Chicago Press.Nommik H, Moller G. 1981. Nitrogen recovery in soil and needlebiomass after fertilization of a Scots pine stand, and growthresponses obtained. Studies in Forest Science 159: 5±37.
Norberg RA. 1988. Theory of growth geometry of plants and self-thinning of plant populations: geometric similarity, elasticsimilarity, and di�erent growth models of plants. AmericanNaturalist 131: 220±256.
O'Brien ST, Hubbell SP, Spiro P, Condit R, Foster RB. 1995.Diameter,height, crown and age relationships in eight neotropical treespecies. Ecology 76: 1926±1939.
Piha MI, Vallack HW, Reeler BM, Michael N. 1995. A low inputapproach to vegetation establishment on mine and coal ash wastesin semiarid regions. I. Tin mine tailings in Zimbabwe. Journal ofApplied Ecology 32: 372±381.
Raich JW, Russell AR, Crews TE, Farrington H, Vitousek PM. 1996.Both nitrogen and phosphorus limit plant production on youngHawaiian lava ¯ows. Biogeochemistry 32: 1±14.
Rich PM. 1986. Mechanical architecture of arborescent rain forestpalms. Principes 30: 117±131.
Schoenholtz SH, Burger JA, Kreh RE. 1992. Fertilizer and organicamendment e�ects on mine soil properties and revegetationsuccess. Soil Science Society of America Journal 56: 1177±1184.
Sinclair TR. 1990. Nitrogen in¯uence on the physiology of crop yield.In: Rabbinge R, Goudriaan J, van Keulen H, De Vries PenningFWT, van Laar HH, eds. Theoretical production ecology:re¯ections and prospects. Wageningen, Netherlands: Pudoc,41±55.
Singh A. 1999. Revegetation of coal mine spoil: in¯uence of nutrientamendment and neighbouring species on growth performance andfoliar nutrient dynamics of woody species. PhD Thesis, BanarasHindu University, India.
Singh A, Jha AK, Singh JS. 1996. In¯uence of NPK fertilization onbiomass production of Pennisetum pedicellatum seeded on coalmine spoil. Tropical Ecology 37: 285±287.
Singh JS, Jha AK. 1993. Restoration of degraded land: an overview. In:Singh JS, ed. Restoration of degraded land: concepts and strategies.Meerut, India: Rastogi Publications, 1±9.
Singh JS, Singh KP, Jha AK. 1996. An integrated ecological study onrevegetation of mine spoil. Final Technical Report submitted to theMinistry of Coal, Government of India, New Delhi.
SPSS/PC. 1993. SPSS/PC for the IBM PC/XT/AT. Chicago, IL: SPSSInc.
Tanner EVJ, Kapos V, Franco W. 1992. Nitrogen and phosphorusfertilization e�ects on Venezuelan montane forest trunk growthand litterfall. Ecology 73: 78±86.
Tanner EVJ, Kapos V, Freskos S, Healey JR, Theobald AM. 1990.Nitrogen and phosphorus fertilization of Jamaican montane foresttrees. Journal of Tropical Ecology 6: 231±238.
Vitousek PM, Farrington H. 1997. Nutrient limitation and soildevelopment: Experimental test of a biogeochemical theory.Biogeochemistry 37: 63±75.
Vitousek PM, Walker LR, Whiteaker LD, Matson PA. 1993. Nutrientlimitation to plant growth during primary succession in HawaiiVolcanoes National Park. Biogeochemistry 23: 197±215.
Vitousek PM, Walker LR, Whiteaker LD, Mueller-Dombois D, MatsonPA. 1987. Biological invasion by Myrica faya alters ecosystemdevelopment in Hawaii. Science 238: 802±804.
Vogel WG. 1981. A guide for revegetating coal mine soils in the easternUnited States. General Technical Report NE-68. United StatesDepartment of Agriculture Forest Service.
Wali MK. 1975. Practices and problems of land reclamation in westernNorth America. University Grand Forks: North Dakota Press,1±17.
Zavitkovski J, Stevens RD. 1972. Primary productivity of red alder
tion on Tree Growth on Mine Spoil 787
ecosystems. Ecology 53: 235±242.