Molecule Transport Review #2 REVIEW JEOPARDY S2C06 Jeopardy Review.
Chemistry Jeopardy Chapter 6 Review
description
Transcript of Chemistry Jeopardy Chapter 6 Review

Chemistry JeopardyChapter 6 Review
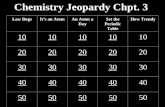
S superman block
S super
woman block
DFLeopar
dP Diddy block
P cock block
P soup block
Odds and Ends
200 200 200 200 200 200 200
400 400 400 400 400 400 400
600 600 600 600 600 600 600
800 800 800 800 800 800 800
1000 1000 1000 1000 1000 1000 1000

S superman block200
• Group 1 is called the – Alkali metals

S superman block 400
• Group 2 is called the ?– Alkaline earth metals

S superman block 600
• Which elements would you expect to behave similar to Mg?
• Ca and Be

S superman block 800
• Which block contains the most reactive metals family (group)?
• S block the Alkali metals

S superman block 1000
• Why are alkali and alkaline metals never found in their elemental state in nature? Which alkali metal is used to teat depression– They are too reactive– Li

S superwoman block 200
• What type of ions do alkali metals form?– 1+

S superwoman block 400
• Which alkali metal is the most reactive? Which alkali is produced commercially in large amounts for salt, bleach and baking soda– Fr– Na

S superwoman block 600
• Which alkaline earth metal is radioactive?– Radium

S superwoman block 800
• Why are the alkaline earth metals not as reactive as the alkali metals?– Smaller AR therefore higher IE

S superwoman block 1000
• The shells of marine animals and limestone is made up of what compound– Calcium carbonate

Blocks d & f 200
• Chromium is in the _________ metals? Why is it used to plate bumpers– Transition– Hard, resists corrosion and lustrous

Blocks d & f 400
• The artificial (synthesized) elements belong to which series?– Actinides

Blocks d & f 600
• Which element is used for paint pigments? Which element is used for electrical wires– Cr– Cu

Blocks d & f 800
• Which d block element is the best conductor of electricity? Why don’t we use it instead of Cu? Name the coinage metals– Ag…too expensive!– Cu, Ag and Au

Blocks d & f 1000
• How many karate is pure gold? Why don’t we use this for rings? What are the 4f and 5f called?– 24K..too soft– Lanthanides ad Actinides

P Diddy block200
• Elements of group 17 are called the? What type of ions do they form– Halogens– 1-

P Diddy block 400
• An element with an electron configuration of 1s2 2s2 2p2 belongs to what group?– Group 14

P Diddy block 600
• How many electrons do carbon, nitrogen and oxygen have in their outermost energy level? What ions do they form– 4; 5; and 6– +/-4: -3: -2

P Diddy block 800
• True or false? The p block contains metals and metalloids (semi metals) only– False it contains metals, semimetals
and non metals

P Diddy block 1000
• Who is the most reactive element on the PT? Compare the two forms of elemental oxygen– Fluorine– O2- odorless colorless, required for CR– O3-(ozone) strong odor, toxic. Ground
level=pollution, upper level protects against UV radiation

P cock block 200
• The major component of the atmosphere is?– Nitrogen

P cock block 400
• The most abundant element on earth is?– oxygen

P cock block 600
• The major component of glass is?– Si

P cock block 800
• The most abundant Noble Gas is? The most abundant element in the universe is?– Ar– H

P cock block 1000
• The chemical name for rust (Fe) is ?– Iron oxide

P soup block 200
• Steel is an alloy of carbon and?– Fe

P soup block 400
• Which group has the highest ionizing energy and why?– Noble Gases – Full valence shell

P soup block 600
• Most of earths hydrogen is combined with? – Oxygen in water

P soup block 800
• Which element is found in meteorites? Oh… and by the way it’s a d blocker– Ir

P soup block 1000
• Hemoglobin contains the element– Fe

Odds and Ends 200
• Naturally occurring elements are produced in the stars (our sun) by a process called?– Fusion, Hydrogen's are fused to
create He

Odds and Ends 400
• Define alloy– A mixture of metals

Odds and Ends 600
• The fizz in soft drinks comes from…hint they are called carbonated drinks!– Carbon dioxide

Odds and Ends 800
• What happens to the IE as you move down a group? As you move from L to R on the PT?– it decreases– It increases

Odds and Ends 1000
• State The Periodic Law– Chemical and physical properties of
elements are functions of their atomic numbers

Final Jeopardy• How is hydrogen obtained from
water? You must NAME the process