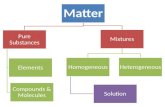
Elements, Compounds and Mixtures. Pure Substances: Elements and Compounds.
Chapter 3Atoms and Elements 3.1 Elements and Symbols Elements are pure substances that cannot be...
-
Upload
marlene-lamb -
Category
Documents
-
view
223 -
download
0
Transcript of Chapter 3Atoms and Elements 3.1 Elements and Symbols Elements are pure substances that cannot be...

Chapter 3Chapter 3 Atoms and Atoms and ElementsElements
3.1 Elements and Symbols Elements are
• pure substances that cannot be separated into simpler substances by ordinary laboratory processes.
• the building blocks of matter.
gold carbon aluminum
1

Sources of Some Element Sources of Some Element NamesNamesSome elements are named for planets, mythological figures, minerals, colors, scientists, and places.
A symbol
•represents the name of an element.
•consists of 1 or 2 letters.
•starts with a capital letter
•With few elements use their own special symbols
2
1-Letter Symbols 2-Letter SymbolsC carbon Co cobalt N nitrogen Ca calciumS sulfur Au gold
Mg magnesium

Physical Properties of Physical Properties of ElementsElements
Some physical properties ofcopper are:
Color Red-orangeLuster Very shinyMelting point 1083 °CBoiling point 2567 °CConduction of electricity ExcellentConduction of heat Excellent
3

ExampleExample
Select the correct symbol for each.A. Phosphorous
1) K 2) P 3) Ph
Select the correct name for each symbol.A. Ne
1) neon 2) nitrogen 3) nickel
B. K 1) potassium 2) phlogiston 3) phosphorus
4

Development of the Periodic TableDevelopment of the Periodic TableMendeleev’s Periodic Table (1871)
Until the discovery of the proton, the elements were typically organized by increasing atomic weight.The modern organization is by increasing atomic number.
3.3The Periodic Table3.3The Periodic Table

Groups and PeriodsGroups and PeriodsOn the periodic table,
• elements are arranged according to similar properties.
• groups contain elements with similar properties in vertical columns.
• periods are horizontal rows of elements.
6

Names of Some Representative Names of Some Representative ElementsElements
Group Numbers
•use the letter A for the representative elements (1A to 8A)
•and the letter B for the transition elements.
•also use numbers 1-18 to the columns from left to right.

Elements and the Periodic Elements and the Periodic TableTable

ExamplesExamples
Identify the element described by the following:
A. Group 7A (17), Period 4 1) Br 2) Cl 3) Mn
B. Group 2A (2), Period 3 1) beryllium 2) boron 3)
magnesium
C. Group 5A (15), Period 2 1) phosphorus 2) arsenic 3) nitrogen
9

Metals, Nonmetals, and Metals, Nonmetals, and MetalloidsMetalloids
The heavy zigzag line separates metals and
nonmetals.
• Metals are located to the left.
• Nonmetals are located to the right.
• Metalloids are located along the heavy zigzag line between the metals and nonmetals.
10

Metals, Nonmetals, and Metals, Nonmetals, and Metalloids on the Periodic TableMetalloids on the Periodic Table
11
The heavy zigzag line separates metals and nonmetals.
• Metals are located to the left.
• Nonmetals are located to the right.
• Metalloids are located along the heavy zigzag line between the metals and nonmetals.

Physical properties Physical properties
Metals
• are shiny and ductile.
• are good conductors of heat and electricity. Nonmetals
• are dull, brittle, and poor conductors.
• are good insulators.Metalloids
• are better conductors than nonmetals, but not as good as metals.
• are used as semiconductors and insulators.

Periods: 7 horizontal rows.
Groups: 18 vertical columns. International standard: 1-18US system: 1A-8A, 1B-8B
SUMMARY

ExamplesExamples
Identify each of the following elements as 1) metal, 2) nonmetal, or 3) metalloid.
A. sodium ____B. iodine ____C. Argon ____D. iron ____E. Silicon ____
14

The Law of Multiple Proportions and Dalton’s The Law of Multiple Proportions and Dalton’s Atomic TheoryAtomic Theory
• Elements are made up of tiny particles called atoms.
• Each element is characterized by the mass of its atoms. Atoms of the same element have the same mass, but atoms of different elements have different masses.
• The chemical combination of elements to make different chemical compounds occurs when atoms join in small whole-number ratios.
• Chemical reactions only rearrange how atoms are combined in chemical compounds; the atoms themselves don’t change.
3.4 The Atom

Atomic Structure: ElectronsAtomic Structure: Electrons
Cathode-Ray Tubes: J. J. Thomson (1856-1940) proposed that cathode rays must consist of tiny negatively charged particles. We now call them electrons.

Rutherford’s Gold Foil Rutherford’s Gold Foil ExperimentExperimentIn Rutherford’s gold foil experiment, positively charged particles • were aimed at atoms of gold.• mostly went straight through the atoms. • were deflected only occasionally.Conclusion:
There must be a small, dense, positively charged nucleus in the atom that deflects positive particles that come close.

Subatomic ParticlesSubatomic Particles
Atoms contain subatomic particles.
• Protons have a positive (+) charge.
• Electrons have a negative (-) charge.
• Neutrons are neutral.
• Like charges repel and unlike charges attract.

Structure of the AtomStructure of the Atom
An atom consists
• of a nucleus that contains protons and neutrons.
• of electrons in a large, empty space around the nucleus.

Atomic Mass ScaleAtomic Mass Scale
On the atomic mass scale,
• 1 atomic mass unit (amu) has a mass equal to 1/12 of the mass of the carbon-12 atom.
• a proton has a mass of about 1 (1.007) amu.
• a neutron has a mass of about 1 (1.008) amu.
• an electron has a very small mass, 0.000 549 amu.
20

ExamplesExamples
Identify each statement as describing a1) proton, 2) neutron, or 3) electron.
A. found outside the nucleusB. has a positive chargeC. is neutralD. found in the nucleus
21

ExamplesExamples Is each of the following statements true or
false?◦ Protons are heavier than electrons◦ Protons are attracted to neutrons◦ Electrons are small that they have no
electrical charge◦ The nucleus contains all the protons and
neutrons of an atom
22

3.5 Atomic Number and Mass 3.5 Atomic Number and Mass NumberNumber
The atomic number
• is specific for each element.
• is the same for all atoms of an element.
• is equal to the number of protons in an atom.
• appears above the symbol of an element.
23

Atomic Number and ProtonsAtomic Number and Protons
What is the atomic number, number of proton and electrons in
•H atom:
•Na atom:
•Co atom:
•Se atom:
24

Electrons in An AtomElectrons in An Atom
An atom
• of an element is electrically neutral; the net charge of an atom is zero.
• has an equal number of protons and electrons.
number of protons = number of electrons
Aluminum has 13 protons and 13 electrons. The net
(overall) charge is zero.
13 protons + 13 electrons = 0
(+13) + (-13) = 0 (neutral)
25

Mass NumberMass NumberThe mass number
• represents the number of particles in the nucleus.
• is equal to the number of protons + the number of neutrons.
26
Element Symbol Atomic number
Mass number
p+ e- n0
Carbon C 6 12 6 6 0
Chlorine 37
Gold 79

ExamplesExamples
An atom of zinc has a mass number of 65.A. How many protons are in this zinc atom?
1) 30 2) 35 3) 65
B. How many neutrons are in the zinc atom? 1) 30 2) 35 3) 65
C. What is the mass number of a zinc atom that has 37 neutrons?
1) 37 2) 65 3) 67
27

3.6 Isotopes and Atomic 3.6 Isotopes and Atomic MassMass
Isotopes
• are atoms of the same element that have different mass numbers.
• have the same number of protons, but different numbers of neutrons.
• Isoptic symbol
28
Z
A
X
X A
Atomic number
Mass number
or Element symbol
gives the mass number in the upper left corner and the atomic number in the lower left corner

Information from Atomic Information from Atomic SymbolsSymbolsExamples of number of subatomic particles for atoms
Atomic symbol
16 31 65
O P Zn 8 15 30
8 p+ 15 p+ …. p+
8 n 16 n ……n 8 e- 15 e- ….. e-
29

ExamplesExamples
Naturally occurring carbon consists of three isotopes: 12C, 13C, and 14C. State the number of protons, neutrons, and electrons in each of the following:
12C 13C 14C 6 6 6
protons ______ ______ ______
neutrons ______ ______ ______
electrons ______ ______ ______
30

ExamplesExamples
Write the atomic symbols for atoms with the following subatomic particles:
A. 8 p+, 8 n, 8 e- ___________
B. 17p+, 20n, 17e- ___________
C. 47p+, 60 n, 47 e- ___________
31

Atomic MassAtomic Mass
The atomic mass of an element
• is listed below the symbol of each element on the periodic table.
• gives the mass of an “average” atom of each element compared to 12C.
• is not the same as the mass number.
• has the smallest unit of amu
32
Na22.99

ExamplesExamples
Using the periodic table, specify the atomic mass of
each element.
A.calcium __________
B. aluminum __________
C. lead __________
D. barium __________
E. iron __________
33

Isotopes of SulfurIsotopes of Sulfur
A sample of naturallyoccurring sulfur containsseveral isotopes with thefollowing abundancesIsotope %
abundance 32S 95.02 33S 0.75 34S 4.21 36S 0.02
34
32S, 33S, 34S, 36S16 16 16 16
Average mass of S is 32.07 amu

Calculating Atomic MassCalculating Atomic Mass
The calculation for atomic mass requires the
• percent(%) abundance of each isotope.
• atomic mass of each isotope of that element.
• sum of the weighted averages.
35
Average massor Atomic mass(amu)
= isotoptic mass 1 x % abundance + isotopic mass 2 x % abundance +......isotopic mass n x % abundance
Report your answer to two decimal places

Atomic MassesAtomic Masses
carbon-12: 98.89 % natural abundance 12.00 amu
carbon-13: 1.11 % natural abundance 13.0034 amu
Why is the atomic mass of the element carbon 12.01 amu?
= 12.01 amu
mass of carbon = (12 amu)(0.9889) + (13.0034 amu)(0.0111)
= 11.87 amu + 0.144 amu

Atomic Mass for ClAtomic Mass for Cl
The atomic mass of chlorine is
• due to all the Cl isotopes.
• not a whole number.• the average of two
isotopes: 35Cl and 37Cl.
37

Calculating Atomic Mass for Calculating Atomic Mass for ClCl
35Cl has atomic mass 34.97 amu (75.76%) and 37Cl has atomic mass 36.97 amu (24.24%).
• Use atomic mass and percent of each isotope to calculate the contribution of each isotope to the weighted average. Atomic mass 35Cl x % abundance =Atomic mass 37Cl x % abundance =
• Sum is atomic mass of Cl is
38