Chapter 13 Electrons Models of the Atom Electron Arrangement Physics and the Quantum Mechanical...
-
Upload
karl-keniston -
Category
Documents
-
view
222 -
download
1
Transcript of Chapter 13 Electrons Models of the Atom Electron Arrangement Physics and the Quantum Mechanical...

Chapter 13 Electrons
• Models of the Atom• Electron Arrangement• Physics and the Quantum Mechanical
Model

Chapter 13.1 Models of the Atom
• The atom was considered a solid indivisible mass originally
http://www.youtube.com/watch?v=uhgd9VM9rsM

Thomson Model
• Plum – Pudding Atom: Negatively charged electrons stuck into a lump of positively charged material.

Rutherford Model
• Electrons surround a dense nucleus made of protons and neutrons. The rest of the atom was empty space

Bohr Model (Planetary Model)
• Electrons are arranged in concentric circular paths (orbits) around the nucleus.
• Patterned after the motion of the planets • Electrons have a fixed energy



Quantum Mechanical Model
• Primarily a mathematical model • Restricts electron to certain energy levels• No defined path for electrons• Gives a percent chance of finding an
electron in a certain space

http://www.youtube.com/watch?v=xqNSQ3OQMGI

Quantum
• Amount of energy required to move an electron from its current energy level to the next highest energy level


Fixed Energy Levels
• Rungs of a Ladder• Garden Steps



Orbits
• S orbital• P orbital• D orbital • F orbital




Chapter 13.2 Electron Arrangement
• Electron Configurations• Exceptional Electron Configurations

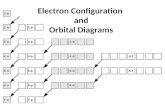
Electron Configurations
• The way in which electrons are arranged around the nuclei of an atom.
• Three Rules:– Aufbau Principle– The Pauli Exclusion Principle– Hund’s Rule

Aufbau Principle
• Electrons enter orbitals of lowest energy first.
• Some orbitals overlap between different energy levels

The Pauli Exclusion Principle
• An atomic orbital may describe at most two electrons
• One or two electrons may occupy one orbital
• They will have opposite spins (shown by opposite arrows)

Hund’s Rule
• When electrons occupy orbitals of equal energy, one electron enters each orbital until all the orbitals contain one electron with parallel spins.